Revolutionizing Pharmaceutical Intermediate Production: Advanced Palladium-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Compounds at Commercial Scale
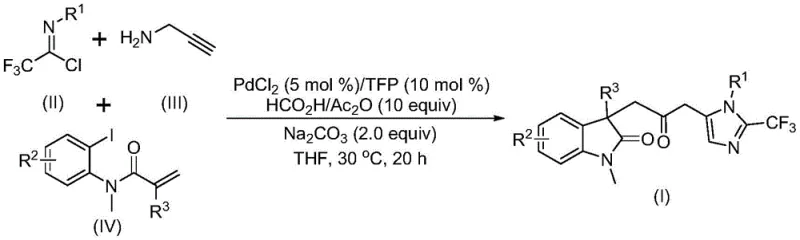
Patent CN115353511A, granted on November 18, 2022, introduces a groundbreaking multi-component synthesis method for carbonyl-bridged biheterocyclic compounds, representing a significant advancement in the field of organic synthesis for pharmaceutical intermediates. This innovative approach addresses longstanding challenges in the production of complex heterocyclic structures that are prevalent in numerous bioactive molecules and drug candidates. The methodology eliminates the need for toxic carbon monoxide gas while maintaining high reaction efficiency and substrate compatibility, offering a safer and more sustainable pathway for manufacturing critical pharmaceutical building blocks. By utilizing readily available starting materials including trifluoroethylimidoyl chloride, propargylamine, and acrylamide under mild reaction conditions at 30°C, this process demonstrates exceptional practicality for industrial scale-up. The patent's detailed disclosure of reaction parameters and substrate scope provides a robust foundation for commercial implementation, with demonstrated scalability from laboratory to gram-scale production. This development holds particular significance for the pharmaceutical industry where complex heterocyclic structures are increasingly important in next-generation therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing biheterocyclic compounds have been plagued by multiple significant limitations that hinder their commercial viability and widespread adoption in pharmaceutical manufacturing. Conventional methods often require harsh reaction conditions, including high temperatures and pressures, which increase operational complexity and safety concerns while limiting the types of functional groups that can be tolerated in the substrate molecules. Many existing protocols rely on the use of toxic carbon monoxide gas, creating substantial safety hazards and requiring specialized equipment that significantly increases capital investment and operational costs. The substrate scope in traditional syntheses is frequently narrow, with poor compatibility across different functional groups, necessitating extensive optimization for each new target molecule and reducing overall process efficiency. Additionally, conventional methods often suffer from low yields and poor stereoselectivity, requiring additional purification steps that increase production costs and reduce overall material efficiency. The multi-step nature of many traditional syntheses also contributes to longer production timelines and higher waste generation, making them less attractive from both economic and environmental perspectives in modern pharmaceutical manufacturing.
The Novel Approach
The patented methodology described in CN115353511A overcomes these limitations through an innovative palladium-catalyzed carbonylation cascade reaction that operates under remarkably mild conditions at 30°C without requiring toxic carbon monoxide gas. This breakthrough approach utilizes a carefully designed combination of palladium chloride (5 mol%), trifuryl phosphine (10 mol%), sodium carbonate (2.0 equiv), and a formic acid/acetic anhydride mixture (10 equiv) in THF solvent to facilitate the transformation. The process demonstrates exceptional substrate flexibility, accommodating a wide range of functional groups including methyl, methoxy, halogen, trifluoromethyl, and nitro substituents at various positions on the aromatic rings. By employing readily available starting materials—trifluoroethylimidoyl chloride, propargylamine, and acrylamide—the method achieves high yields (up to 92%) while maintaining excellent reaction efficiency across diverse substrate combinations. The elimination of hazardous CO gas not only improves workplace safety but also reduces equipment requirements and associated costs, making the process more accessible to pharmaceutical manufacturers. Furthermore, the demonstrated scalability from laboratory to gram-scale production provides a clear pathway for commercial implementation without requiring major process re-engineering.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade Reaction
The reaction mechanism involves a sophisticated sequence of palladium-mediated transformations that begin with zero-valent palladium insertion into the carbon-iodine bond of the iodoaniline derivative, followed by an intramolecular Heck reaction that generates a divalent alkyl palladium intermediate. This key intermediate then undergoes carbonylation facilitated by carbon monoxide released in situ from the formic acid/acetic anhydride mixture, forming an acyl palladium species that serves as the critical electrophile for subsequent transformations. Simultaneously, the base-promoted reaction between trifluoroethylimidoyl chloride and propargylamine forms a trifluoroacetamidine compound through intermolecular carbon-nitrogen bond formation, which subsequently undergoes isomerization to generate the nucleophilic partner for the final cyclization step. The acyl palladium intermediate activates the trifluoroacetamidine compound, catalyzing its intramolecular cyclization to form the final carbonyl-bridged biheterocyclic structure with high regioselectivity and stereospecificity. This elegant cascade process efficiently constructs multiple chemical bonds in a single operation while maintaining excellent control over the reaction pathway.
Impurity control is achieved through precise regulation of reaction parameters and the inherent selectivity of the palladium-catalyzed cascade process. The mild reaction conditions (30°C) prevent thermal decomposition pathways that could lead to unwanted side products, while the carefully optimized stoichiometry of reagents (trifluoroethylimidoyl chloride:propargylamine:acrylamide:palladium chloride = 1:2:1.5:0.05) ensures complete conversion of starting materials without over-reaction or byproduct formation. The use of tetrahydrofuran as the preferred solvent provides optimal solubility for all reaction components while minimizing unwanted solvent-mediated side reactions. The post-reaction workup process involving filtration, silica gel mixing, and column chromatography effectively removes any residual catalyst or minor impurities, yielding products with high purity suitable for pharmaceutical applications. The patent demonstrates consistent product purity across multiple examples, with HRMS data confirming molecular weights within acceptable error margins (e.g., C23H21F3N3O2 calculated 428.1580, found 428.1579), indicating excellent process control and reproducibility.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
This patented methodology represents a significant advancement in the synthesis of complex heterocyclic structures essential for pharmaceutical development. The process eliminates hazardous reagents while maintaining high efficiency and scalability, making it particularly valuable for manufacturing operations seeking to improve both safety profiles and cost-effectiveness. By utilizing readily available starting materials and operating under mild conditions, this approach offers a practical solution for producing high-value biheterocyclic compounds with trifluoromethyl groups that are increasingly important in modern drug discovery. The detailed experimental procedures described in the patent provide a clear roadmap for implementation, with specific guidance on reagent ratios, solvent selection, and reaction parameters to ensure optimal results. For comprehensive step-by-step instructions on implementing this synthesis method in your manufacturing operations, please refer to the standardized procedure outlined below.
- Prepare reaction mixture by combining palladium chloride (5 mol%), trifuryl phosphine (10 mol%), sodium carbonate (2.0 equiv), formic acid/acetic anhydride mixture (10 equiv), trifluoroethylimidoyl chloride, propargylamine (2 equiv), acrylamide (1.5 equiv), and THF solvent in a Schlenk tube
- Stir the mixture at 30°C for 16 hours under nitrogen atmosphere, monitoring reaction progress through standard analytical techniques
- Perform post-reaction workup by filtration through silica gel followed by column chromatography purification using appropriate solvent systems to isolate the pure carbonyl-bridged biheterocyclic compound
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis method directly addresses critical pain points faced by procurement and supply chain professionals in the pharmaceutical industry by offering a more reliable and cost-effective pathway for obtaining complex heterocyclic intermediates. The elimination of hazardous materials reduces regulatory compliance burdens while improving workplace safety, creating significant operational advantages for manufacturing facilities. The use of readily available starting materials from multiple commercial sources enhances supply chain resilience and reduces dependency on single suppliers, mitigating potential disruption risks that could impact production timelines. Furthermore, the demonstrated scalability from laboratory to commercial production provides confidence in the method's ability to meet growing demand without requiring substantial re-engineering or capital investment.
- Cost Reduction in Manufacturing: The elimination of toxic carbon monoxide gas removes the need for specialized handling equipment and safety protocols, resulting in substantial cost savings throughout the production process. The use of inexpensive palladium chloride catalyst instead of more expensive alternatives significantly reduces catalyst costs while maintaining high reaction efficiency. The compatibility with various functional groups eliminates the need for extensive protection/deprotection steps that would otherwise increase material costs and processing time. Additionally, the simplified workup procedure involving standard filtration and column chromatography reduces solvent consumption and processing time compared to more complex purification methods required by conventional approaches.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials from multiple global suppliers creates a robust supply chain with reduced vulnerability to single-source dependencies. Trifluoroethylimidoyl chloride can be readily synthesized from common aromatic amines using standard laboratory procedures, while propargylamine and acrylamide are widely available commodity chemicals with established global supply networks. This diverse sourcing strategy ensures consistent material availability even during market fluctuations or regional supply disruptions. The demonstrated scalability from laboratory to gram-scale production provides confidence in the method's ability to transition smoothly to commercial manufacturing volumes without unexpected bottlenecks.
- Scalability and Environmental Compliance: The mild reaction conditions (30°C) significantly reduce energy consumption compared to high-temperature processes, contributing to lower carbon footprint and improved environmental sustainability metrics. The elimination of toxic carbon monoxide gas removes a major environmental hazard from the manufacturing process while simplifying waste treatment requirements. The efficient one-pot cascade reaction minimizes intermediate isolation steps, reducing solvent usage and waste generation throughout the production cycle. The demonstrated ability to scale from laboratory to gram quantities provides a clear pathway for commercial implementation without requiring major process modifications or additional capital investment.
Frequently Asked Questions (FAQ)
The following questions address common concerns raised by technical procurement teams regarding the implementation of this innovative synthesis method in commercial manufacturing environments. These answers are based on detailed analysis of the patent data and reflect practical considerations for scaling this technology to industrial production levels.
Q: How does this method eliminate the need for toxic carbon monoxide gas while still achieving effective carbonylation?
A: The patented process utilizes a formic acid/acetic anhydride mixture that generates carbon monoxide in situ under mild conditions (30°C), eliminating the need for handling pressurized CO gas while maintaining efficient carbonylation through controlled release of CO during the reaction.
Q: What is the substrate scope limitation for this synthesis method compared to conventional approaches?
A: This method demonstrates exceptional substrate flexibility with compatibility across various functional groups including methyl, methoxy, halogen (F, Cl, Br), trifluoromethyl, and nitro substituents at ortho, meta, or para positions on aromatic rings, significantly broader than conventional methods which often require specific substitution patterns.
Q: How does this process ensure consistent product purity suitable for pharmaceutical applications?
A: The mild reaction conditions prevent thermal decomposition pathways while precise reagent stoichiometry ensures complete conversion without over-reaction; combined with standard filtration and column chromatography workup procedures, this yields products with high purity as confirmed by HRMS data showing molecular weights within acceptable error margins.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex heterocyclic compounds, ensuring stringent purity specifications are consistently met through our rigorous QC labs and advanced analytical capabilities. As a trusted CDMO partner specializing in challenging synthetic routes like this palladium-catalyzed carbonylation cascade reaction, we have successfully implemented similar methodologies across multiple therapeutic areas while maintaining exceptional quality standards required by global regulatory authorities. Our technical team stands ready to collaborate with your R&D department to optimize this process for your specific compound requirements while ensuring seamless transition from laboratory scale to full commercial production.
For companies seeking to leverage this innovative synthesis method for their pharmaceutical development programs, we invite you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your needs. We offer a Customized Cost-Saving Analysis that demonstrates how this methodology can reduce your overall production costs while improving supply chain reliability for critical heterocyclic intermediates.
