Scalable Synthesis of High-Purity (E)-1-Chloro-2-Iodoethylene Intermediates for Global Pharma
The pharmaceutical and fine chemical industries continuously seek robust methodologies for constructing complex halogenated building blocks, which serve as critical precursors in the synthesis of active pharmaceutical ingredients (APIs). Patent CN111807938B introduces a groundbreaking approach for the preparation of (E)-1-chloro-2-iodoethylene compounds, addressing long-standing challenges associated with traditional halogenation techniques. This innovation leverages a transition metal-free catalytic system that utilizes inexpensive lithium salts and organic oxidants to achieve high-efficiency difunctionalization of alkynes. The significance of this technology lies in its ability to produce high-purity intermediates under mild reaction conditions, thereby enhancing the safety profile and economic viability of the manufacturing process. By eliminating the need for hazardous reagents like iodine chloride, this method aligns perfectly with modern green chemistry principles while maintaining exceptional yield and selectivity standards required by global regulatory bodies. 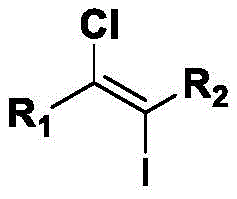
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of vicinal dihaloalkenes has relied heavily on the direct addition of molecular halogens or interhalogen compounds such as iodine chloride (ICl) to alkyne substrates. While chemically effective, these conventional reagents pose severe safety and handling challenges due to their high toxicity, corrosivity, and volatility, which complicate storage and transportation logistics for large-scale operations. Furthermore, reactions involving molecular halogens often suffer from poor regioselectivity and stereoselectivity, leading to complex mixtures of isomers that require energy-intensive and costly purification steps to isolate the desired (E)-isomer. The generation of hazardous waste streams associated with these traditional methods also imposes a significant environmental burden, increasing the overall cost of compliance and waste disposal for chemical manufacturers. Consequently, there is an urgent industrial demand for safer, more selective, and environmentally benign alternatives that can deliver consistent quality without compromising operational safety.
The Novel Approach
The methodology disclosed in patent CN111807938B represents a paradigm shift by employing a combination of lithium chloride, lithium iodide, and an organic oxidant in a mixed solvent system to achieve the desired transformation. This novel approach operates under remarkably mild conditions, typically initiating at 0°C and warming to room temperature, which significantly reduces energy consumption compared to high-temperature processes. The use of stable solid salts instead of volatile liquids simplifies the dosing and handling procedures, thereby minimizing the risk of accidental exposure and improving workplace safety standards. Moreover, this system demonstrates excellent functional group tolerance, allowing for the successful halogenation of substrates containing sensitive moieties such as esters, ketones, and trifluoromethoxy groups without degradation. The one-pot nature of the reaction streamlines the workflow, reducing the number of unit operations and facilitating a more efficient path from raw materials to high-value intermediates.
Mechanistic Insights into Oxidative Iodochlorination of Alkynes
The core of this technological advancement lies in the oxidative generation of electrophilic halogen species in situ, which then react with the alkyne triple bond to form the vinyl halide product. The mechanism likely involves the oxidation of iodide ions by the organic oxidant to generate an electrophilic iodine species, which attacks the alkyne to form a cyclic iodonium intermediate. Subsequent nucleophilic attack by the chloride ion occurs in an anti-addition fashion, resulting in the exclusive formation of the (E)-isomer with high stereochemical fidelity. This controlled pathway ensures that the chlorine and iodine atoms are installed at specific positions on the double bond, providing the precise structural architecture needed for downstream cross-coupling reactions like Suzuki or Sonogashira couplings. The ability to tune the reaction outcome by adjusting the molar ratios of the halide salts and oxidant offers chemists a powerful tool for optimizing yields across a diverse range of substrate classes. 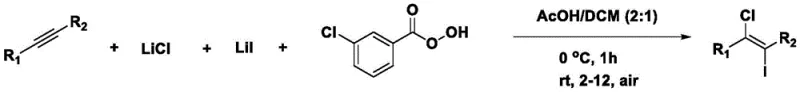
Impurity control is another critical aspect where this mechanism excels, as the mild oxidative conditions prevent the over-oxidation or polymerization of the alkyne starting material. The specific choice of solvent system, typically a mixture of acetic acid and dichloromethane, plays a vital role in stabilizing the reactive intermediates and ensuring homogeneous reaction conditions. By avoiding strong acids or harsh Lewis acids often required in traditional methods, the process minimizes the formation of side products such as trihalogenated species or hydration byproducts. This high level of chemical selectivity translates directly into a cleaner crude reaction profile, which simplifies the subsequent isolation and purification steps. For R&D teams, understanding this mechanistic nuance is essential for scaling the process, as it highlights the importance of maintaining strict temperature control and reagent addition rates to preserve the integrity of the catalytic cycle.
How to Synthesize (E)-1-Chloro-2-Iodoethylene Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry of the halide salts and the rate of oxidant addition to ensure optimal conversion. The standard protocol involves dissolving the alkyne substrate along with lithium chloride and lithium iodide in the organic solvent mixture before cooling the solution to initiate the reaction. Detailed standard operating procedures regarding specific molar ratios, solvent volumes, and workup techniques are critical for reproducing the high yields reported in the patent examples. ![Specific chemical structure of (E)-1-(1-chloro-2-iodo)-1-[4-(trifluoromethoxy)]styrene example compound](/insights/img/e-1-chloro-2-iodoethylene-synthesis-pharma-supplier-20260306172641-04.png)
- Dissolve alkyne substrate, lithium chloride, and lithium iodide in a mixed organic solvent system containing acetic acid.
- Cool the reaction mixture to 0°C and slowly add an organic oxidant such as m-CPBA while maintaining temperature control.
- Allow the reaction to warm to room temperature and stir for 6 to 12 hours, followed by extraction and purification to isolate the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented technology offers substantial strategic advantages by fundamentally altering the cost structure and risk profile of intermediate manufacturing. The substitution of expensive and hazardous interhalogen reagents with commodity lithium salts results in a drastic reduction in raw material costs, while simultaneously simplifying the logistics of chemical sourcing and storage. The mild reaction conditions reduce the demand for specialized high-pressure or high-temperature reactor equipment, allowing for production in standard glass-lined or stainless steel vessels that are readily available in most multipurpose chemical plants. This flexibility enhances supply chain resilience by enabling production across a wider network of manufacturing sites, thereby reducing the risk of bottlenecks or single-point failures in the supply of critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of toxic iodine chloride reagents removes the need for expensive scrubbing systems and specialized containment infrastructure, leading to significant capital expenditure savings. Furthermore, the high atom economy of the reaction ensures that a greater proportion of raw materials are converted into the final product, minimizing waste disposal costs and maximizing overall process efficiency. The use of stable, non-volatile salts also reduces losses due to evaporation or degradation during storage, contributing to a more predictable and lower cost of goods sold over the long term.
- Enhanced Supply Chain Reliability: Sourcing stable solid salts like lithium chloride and lithium iodide is far more reliable than procuring hazardous volatile liquids, which are often subject to strict transportation regulations and supply constraints. This stability ensures a continuous flow of materials to the production line, reducing the likelihood of shutdowns due to reagent shortages or delivery delays. Additionally, the robustness of the reaction against variations in substrate quality means that the process can tolerate a wider range of raw material specifications, further securing the supply chain against upstream fluctuations.
- Scalability and Environmental Compliance: The generation of less hazardous waste streams simplifies the environmental permitting process and reduces the ongoing costs associated with waste treatment and disposal. The ability to run the reaction at near-ambient temperatures lowers energy consumption, aligning with corporate sustainability goals and reducing the carbon footprint of the manufacturing process. These factors combined make the technology highly scalable, allowing for seamless transition from pilot plant trials to full commercial production without the need for major process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this halogenation technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the process capabilities. ![Specific chemical structure of (E)-1-(1-chloro-2-iodo)-1-[4-(tert-butyl)]styrene example compound](/insights/img/e-1-chloro-2-iodoethylene-synthesis-pharma-supplier-20260306172641-011.png)
Q: What are the primary advantages of this halogenation method over traditional ICl reagents?
A: This method avoids the use of highly toxic and volatile iodine chloride (ICl) reagents, utilizing stable and inexpensive lithium salts instead, which significantly improves operational safety and reduces hazardous waste.
Q: What is the regioselectivity of the iodochlorination reaction described in the patent?
A: The process achieves high regioselectivity for the (E)-1-chloro-2-iodoethylene configuration, ensuring consistent stereochemistry crucial for downstream cross-coupling reactions in drug synthesis.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the reaction operates under mild conditions (0°C to room temperature) with simple workup procedures, making it highly adaptable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (E)-1-Chloro-2-Iodoethylene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality halogenated intermediates play in the development of next-generation therapeutics and agrochemicals. Our team of expert chemists has extensively evaluated the methodology described in CN111807938B and possesses the technical capability to adapt this route for diverse substrate classes to meet your specific project requirements. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of (E)-1-chloro-2-iodoethylene delivered meets the highest international standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced synthesis technology for your upcoming projects. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your development timeline and optimize your overall production costs.
