Advanced Synthesis of (E)-1-Chloro-2-Iodoethylene Compounds for Global Pharma Supply Chains
Advanced Synthesis of (E)-1-Chloro-2-Iodoethylene Compounds for Global Pharma Supply Chains
The landscape of organic synthesis is constantly evolving to meet the rigorous demands of modern pharmaceutical and agrochemical manufacturing, where efficiency, safety, and cost-effectiveness are paramount. A significant breakthrough in this domain is documented in Chinese Patent CN111807938A, which discloses a novel and highly efficient preparation method for (E)-1-chloro-2-iodoethylene compounds. These halogenated olefins serve as critical synthetic building blocks, widely utilized in the construction of complex carbon-carbon and carbon-heteroatom bonds essential for active pharmaceutical ingredients (APIs). The innovation lies in its ability to bypass traditional hazardous reagents, offering a robust pathway for producing high-purity intermediates that are indispensable for downstream cross-coupling reactions such as Heck, Sonogashira, and Suzuki couplings.
This technology addresses a long-standing challenge in the industry: the safe and selective introduction of two different halogen atoms onto an unsaturated bond. By leveraging a unique combination of lithium halide salts and organic oxidants within a specific solvent system, the process achieves exceptional regioselectivity without the need for transition metal catalysts. For R&D directors and process chemists, this represents a substantial opportunity to streamline synthetic routes, reduce impurity profiles associated with metal contamination, and enhance the overall sustainability of the manufacturing process. The versatility of the method allows for a broad substrate scope, accommodating various functional groups that are often sensitive to harsher traditional conditions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of vicinal dihaloalkenes, particularly those containing both chlorine and iodine, has relied heavily on the direct addition of interhalogen compounds like iodine monochloride (ICl). While chemically effective, ICl is a highly corrosive, toxic, and volatile reagent that poses severe safety risks in both laboratory and industrial settings. The handling of such hazardous materials requires specialized equipment, stringent safety protocols, and expensive containment systems, all of which drive up operational costs and complicate regulatory compliance. Furthermore, reactions involving ICl often suffer from poor selectivity, leading to mixtures of stereoisomers or over-halogenated byproducts that are difficult to separate, thereby reducing the overall yield and purity of the desired intermediate.
Additionally, many existing alternative methods depend on transition metal catalysis or harsh acidic conditions that can degrade sensitive functional groups present in complex drug molecules. The use of heavy metals introduces another layer of complexity regarding purification, as residual metal levels must be strictly controlled to meet pharmacopeial standards. This necessitates additional downstream processing steps, such as scavenging or extensive chromatography, which not only increase production time but also generate significant amounts of chemical waste. Consequently, there is a pressing need for a greener, safer, and more economically viable alternative that can deliver high-quality halogenated building blocks without these inherent drawbacks.
The Novel Approach
The methodology described in patent CN111807938A offers a transformative solution by replacing dangerous interhalogen gases with stable, inexpensive, and easily handled lithium halide salts. This approach utilizes a "hydrogen bond network" strategy, where acetic acid acts as both a solvent component and a mediator to facilitate the activation of halide anions. By combining lithium chloride and lithium iodide with a mild organic oxidant, such as m-chloroperoxybenzoic acid (m-CPBA), the system generates the reactive halogenating species in situ under remarkably mild conditions. This eliminates the need for storing or transporting hazardous gases, significantly improving the safety profile of the operation while simplifying the logistical supply chain for raw materials.
Moreover, this transition-metal-free protocol exhibits outstanding functional group tolerance, allowing for the successful halogenation of alkynes bearing electron-withdrawing or electron-donating groups without side reactions. The reaction proceeds with high regioselectivity to exclusively form the (E)-isomer, which is often the thermodynamically preferred and biologically relevant configuration for subsequent coupling reactions. The simplicity of the one-pot procedure, combined with the ease of workup involving standard extraction and drying techniques, makes this method highly attractive for scaling up. It effectively bridges the gap between academic innovation and industrial practicality, providing a reliable route for the cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Regioselective Iodochlorination
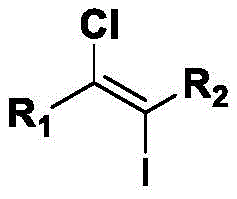
The core of this technological advancement lies in its unique mechanistic pathway, which diverges significantly from traditional electrophilic addition mechanisms. In this system, the organic oxidant activates the halide anions in the presence of the hydrogen-bonding solvent network, likely generating a reactive hypohalite or similar electrophilic species in situ. The difference in nucleophilicity between the chloride and iodide anions, modulated by the solvent environment, dictates the order of addition to the alkyne triple bond. This subtle control ensures that the iodine and chlorine atoms are installed at specific positions with high fidelity, avoiding the random distribution of halogens that plagues less controlled systems. The result is a clean conversion to the target (E)-1-chloro-2-iodoethylene scaffold with minimal formation of regioisomers.
From an impurity control perspective, the absence of transition metals is a critical advantage. Metal-catalyzed reactions often leave behind trace residues that can act as catalysts for decomposition or interfere with downstream biological assays. By utilizing simple lithium salts, the impurity profile is drastically simplified, consisting primarily of unreacted starting materials or benign salt byproducts that are easily removed during aqueous workup. The mild temperature profile, typically ranging from 0°C to room temperature, further suppresses thermal degradation pathways and polymerization side reactions. This level of control is essential for producing high-purity OLED material precursors or complex API intermediates where batch-to-batch consistency is non-negotiable for regulatory approval.
How to Synthesize (E)-1-Chloro-2-Iodoethylene Efficiently
The operational simplicity of this synthesis makes it accessible for both small-scale discovery chemistry and large-scale production environments. The process begins by dissolving the alkyne substrate along with stoichiometric amounts of lithium chloride and lithium iodide in a mixed solvent system comprising acetic acid and an organic co-solvent like dichloromethane. The mixture is cooled to 0°C to manage the exotherm upon the addition of the oxidant. Once the oxidant, such as m-CPBA, is introduced, the reaction is allowed to slowly warm to room temperature and stirred for a period of 6 to 12 hours. This gentle progression ensures complete conversion while maintaining the integrity of the stereochemistry. Detailed standardized synthesis steps are provided in the guide below.
- Dissolve the alkyne substrate, lithium chloride, and lithium iodide in a mixed solvent system of acetic acid and dichloromethane.
- Cool the reaction mixture to 0°C and add an organic oxidant such as m-chloroperoxybenzoic acid (m-CPBA).
- Slowly warm the mixture to room temperature and stir for 6-12 hours to complete the regioselective dihalogenation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthesis route offers tangible strategic benefits that extend beyond mere chemical efficiency. The substitution of volatile, regulated hazardous gases with stable solid salts fundamentally alters the risk profile of the manufacturing process. This shift not only reduces insurance and compliance costs associated with handling toxic substances but also mitigates the risk of supply disruptions caused by strict transportation regulations on hazardous chemicals. The raw materials required—lithium halides and common organic oxidants—are commodity chemicals with robust global supply chains, ensuring consistent availability and price stability compared to specialized interhalogen reagents.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the associated purification steps leads to significant operational savings. Traditional methods often require costly ligands and extensive chromatography to remove metal traces, whereas this method relies on cheap lithium salts that are easily washed away. Furthermore, the high atom economy of the reaction means that a greater proportion of the raw material mass is incorporated into the final product, reducing waste disposal costs. The simplified one-pot nature of the process also reduces labor hours and energy consumption, contributing to a lower overall cost of goods sold (COGS) for the final intermediate.
- Enhanced Supply Chain Reliability: By avoiding reagents that are subject to strict environmental and safety regulations, the supply chain becomes more resilient. The use of stable solids instead of pressurized gases simplifies storage and inventory management, reducing the need for specialized containment infrastructure. This flexibility allows for more agile production scheduling and faster response times to market demand fluctuations. Additionally, the broad substrate scope means that a single standardized protocol can be applied to produce a wide variety of derivatives, streamlining the procurement of diverse intermediates from a single reliable source.
- Scalability and Environmental Compliance: The mild reaction conditions and low toxicity profile make this process inherently scalable from gram to ton quantities without significant re-engineering. The reduction in hazardous waste generation aligns with increasingly stringent global environmental standards, facilitating smoother regulatory approvals in key markets. The ability to conduct the reaction in common solvents like dichloromethane and acetic acid, which are well-understood in industrial recovery systems, further enhances the sustainability of the operation. This ensures long-term viability and reduces the risk of production shutdowns due to environmental compliance issues.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this halogenation technology. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing workflows.
Q: What are the primary advantages of this halogenation method over traditional ICl reagents?
A: This method avoids the use of highly toxic and volatile iodine monochloride (ICl) gas, utilizing stable and cheap lithium halide salts instead. It offers superior safety profiles, easier handling, and excellent functional group tolerance under mild conditions.
Q: Does this process require expensive transition metal catalysts?
A: No, the process is entirely transition-metal-free. It relies on a hydrogen-bond network mediated by acetic acid and organic oxidants, which significantly reduces raw material costs and eliminates the need for complex heavy metal removal steps.
Q: What is the typical yield and scalability of this reaction?
A: The patent data demonstrates high yields ranging from 77% to 98% across various substrates. The one-pot nature, mild temperature profile (0°C to RT), and simple workup make it highly suitable for large-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (E)-1-Chloro-2-Iodoethylene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality halogenated intermediates play in the development of next-generation therapeutics and advanced materials. Our team of expert process chemists has thoroughly analyzed the potential of the technology described in CN111807938A and is fully equipped to translate this innovative methodology into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from the laboratory bench to full-scale manufacturing. Our state-of-the-art facilities are designed to handle complex chemistries with precision, adhering to stringent purity specifications and rigorous QC labs to guarantee batch-to-batch consistency.
We invite you to collaborate with us to leverage this advanced synthesis route for your specific project needs. Whether you require custom synthesis of novel derivatives or large-scale supply of standard intermediates, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can accelerate your supply chain and reduce your time to market.
