Advanced Synthesis of 5-Trifluoromethyl-1,2,4-Triazole Derivatives for Commercial Scale-Up
The pharmaceutical and agrochemical industries continuously seek robust synthetic routes for nitrogen-containing heterocycles, particularly 1,2,4-triazole derivatives, which serve as critical scaffolds in numerous biologically active molecules. Patent CN111978265B discloses a groundbreaking preparation method for 5-trifluoromethyl substituted 1,2,4-triazole derivatives that addresses long-standing challenges in heterocyclic chemistry. The introduction of a trifluoromethyl group into these heterocyclic systems is known to significantly enhance electronegativity, metabolic stability, and lipophilicity, thereby improving the overall bioavailability of the final drug candidates. This specific patent technology leverages a ferric chloride-promoted strategy that operates under remarkably mild conditions, avoiding the stringent requirements for anhydrous or oxygen-free environments that typically plague traditional synthesis methods. By utilizing cheap and readily available starting materials such as hydrazides and trifluoroethylimide chlorides, this innovation opens new avenues for the cost-effective manufacturing of high-value intermediates used in drugs like Maraviroc and Sitagliptin. The ability to scale this process easily to the gram level and beyond provides a substantial foundation for industrial applications, ensuring that supply chains can meet the rigorous demands of global pharmaceutical production without compromising on quality or consistency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted 1,2,4-triazoles has been fraught with significant technical and economic hurdles that hinder efficient commercial production. Literature reports indicate that traditional methods often rely on the condensation of 3,5-ditrifluoromethyl-1,3,4-oxadiazoles with primary amines or the cyclization of trifluoromethyl hydrazides with amidines, processes that are frequently limited by harsh reaction conditions and lengthy procedural steps. These conventional pathways typically suffer from a narrow substrate range, meaning they cannot accommodate diverse functional groups without significant degradation or side reactions, leading to lower overall reaction yields and increased waste generation. Furthermore, some previously developed tandem cyclization reactions, while efficient for certain substrates, fail completely when alkyl hydrazones are involved, rendering them incapable of synthesizing 3-alkyl fluoro 1,2,4-triazoles which are essential for specific therapeutic applications. The requirement for specialized equipment to maintain inert atmospheres or extremely low temperatures further escalates the operational costs and complexity, making these methods less attractive for large-scale manufacturing where simplicity and reliability are paramount for maintaining supply chain continuity.
The Novel Approach
In stark contrast to the limitations of prior art, the novel approach detailed in patent CN111978265B introduces a simple yet highly efficient synthetic strategy promoted by ferric chloride that fundamentally reshapes the production landscape for these derivatives. This method utilizes inexpensive and commercially abundant acyl hydrazides and trifluoroethylimide chlorides as starting materials, drastically reducing the raw material costs associated with the synthesis of complex heterocyclic structures. The reaction proceeds smoothly in common organic solvents like 1,4-dioxane without the need for rigorous exclusion of air or moisture, which simplifies the operational workflow and reduces the burden on facility infrastructure and safety protocols. By enabling the easy scaling of the process to industrial levels, this technology provides unparalleled convenience for manufacturing applications, allowing producers to respond rapidly to market demands for high-purity pharmaceutical intermediates. The broad tolerance for various substituents on the aryl and alkyl groups ensures that a wide library of derivatives can be accessed from a single robust platform, enhancing the flexibility of research and development teams to explore new chemical spaces for drug discovery.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The core of this technological breakthrough lies in the sophisticated interplay between the base-promoted initial coupling and the subsequent Lewis acid-catalyzed cyclization, which together drive the formation of the 5-trifluoromethyl-1,2,4-triazole core with high precision. The reaction mechanism is believed to initiate with a base-promoted intermolecular carbon-nitrogen bond formation, where sodium bicarbonate facilitates the nucleophilic attack of the hydrazide on the imidoyl chloride to generate a trifluoroacetamidine derivative intermediate. Following this initial step, the addition of ferric chloride acts as a potent metal Lewis acid promoter that activates the intermediate for an intramolecular dehydration condensation reaction, effectively closing the triazole ring under thermal conditions. This dual-stage process ensures that the reaction proceeds with high selectivity and efficiency, minimizing the formation of unwanted byproducts that often complicate downstream purification processes in fine chemical manufacturing. The use of ferric chloride is particularly advantageous as it is an earth-abundant metal catalyst that avoids the toxicity and residual contamination issues associated with precious metal catalysts, aligning with modern green chemistry principles and regulatory standards for pharmaceutical ingredients.
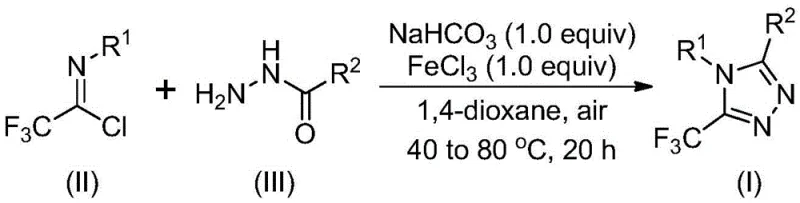
Furthermore, the impurity control mechanism inherent in this synthetic route is designed to maximize the purity profile of the final product, which is a critical parameter for R&D directors evaluating process feasibility for active pharmaceutical ingredients. The specific choice of aprotic solvents such as 1,4-dioxane, acetonitrile, or tetrahydrofuran plays a vital role in solubilizing the reactants effectively while promoting the desired reaction pathway over competing side reactions. The reaction conditions, typically ranging from 30 to 50 degrees Celsius for the initial step and 70 to 90 degrees Celsius for the cyclization, are optimized to balance reaction kinetics with thermal stability, ensuring that sensitive functional groups on the substrate remain intact throughout the transformation. Post-processing involves straightforward filtration and silica gel chromatography, techniques that are well-established in the industry and can be readily adapted for continuous manufacturing processes to further enhance throughput. This level of mechanistic control allows for the precise design of 3,4-disubstituted trifluoromethyl triazole derivatives, providing chemists with the tools to fine-tune the physicochemical properties of the molecule for optimal biological performance.
How to Synthesize 5-Trifluoromethyl-1,2,4-Triazole Derivatives Efficiently
To implement this synthesis route effectively, operators must adhere to a standardized protocol that leverages the specific molar ratios and temperature gradients defined in the patent to ensure consistent reproducibility and high yields. The process begins with the dissolution of sodium bicarbonate, trifluoroethylimide chloride, and hydrazide in an appropriate organic solvent, followed by a controlled heating phase to initiate the formation of the key intermediate species. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results accurately within their own laboratory or pilot plant facilities. Adhering to these guidelines ensures that the full potential of the ferric chloride promotion is realized, minimizing batch-to-batch variability and maximizing the efficiency of raw material conversion into the desired high-value triazole products.
- Mix sodium bicarbonate, trifluoroethylimide chloride, and hydrazide in an aprotic organic solvent such as 1,4-dioxane.
- Stir the mixture at a moderate temperature range of 30 to 50 degrees Celsius for 8 to 16 hours to facilitate initial bond formation.
- Add ferric chloride as a Lewis acid promoter and heat the system to 70 to 90 degrees Celsius for 6 to 10 hours to complete the cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers transformative benefits that directly impact the bottom line and operational resilience of the manufacturing organization. The elimination of expensive transition metal catalysts and the reliance on cheap, industrially available reagents like ferric chloride and sodium bicarbonate leads to substantial cost savings in raw material procurement without sacrificing reaction efficiency or product quality. Additionally, the removal of strict anhydrous and oxygen-free requirements significantly reduces the energy consumption and equipment maintenance costs associated with running specialized inert atmosphere reactors, thereby lowering the overall cost of goods sold. The robustness of the reaction conditions allows for greater flexibility in sourcing raw materials, as the process is tolerant to minor variations in reagent quality, which enhances supply chain reliability and reduces the risk of production delays due to material shortages. This scalability ensures that production can be ramped up quickly to meet surging market demand, providing a competitive edge in the fast-paced pharmaceutical intermediate sector where speed to market is often the deciding factor for commercial success.
- Cost Reduction in Manufacturing: The strategic replacement of precious metal catalysts with inexpensive ferric chloride results in a drastic simplification of the catalyst recovery and removal process, which traditionally accounts for a significant portion of downstream processing costs. By utilizing widely available commodity chemicals as promoters and bases, the dependency on volatile specialty chemical markets is minimized, stabilizing the cost structure of the manufacturing process against market fluctuations. The simplified post-processing workflow, which avoids complex extraction or distillation steps required by harsher methods, further reduces labor and utility expenses, contributing to a leaner and more profitable production model. These cumulative efficiencies translate into a more competitive pricing structure for the final intermediates, allowing downstream partners to optimize their own formulation costs while maintaining high margins.
- Enhanced Supply Chain Reliability: The use of commercially available starting materials that are widely produced in the global chemical industry ensures a stable and continuous supply stream, mitigating the risks associated with single-source dependencies or geopolitical disruptions. Since the reaction does not require specialized gases or ultra-dry solvents, the logistical burden on the supply chain is significantly reduced, allowing for faster turnaround times from order placement to delivery. The ability to store reagents under standard conditions without degradation simplifies inventory management and reduces warehousing costs, enabling manufacturers to maintain optimal stock levels to buffer against unexpected demand spikes. This reliability is crucial for long-term contracts with major pharmaceutical companies, where consistent delivery schedules are mandatory for maintaining regulatory compliance and production planning.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of toxic heavy metals align perfectly with increasingly stringent environmental regulations, reducing the cost and complexity of waste treatment and disposal procedures. The process is inherently designed for scale-up, with thermal parameters that are easily managed in large-scale reactors, ensuring that the transition from laboratory bench to commercial plant is smooth and predictable. By minimizing the generation of hazardous byproducts and utilizing greener solvent systems where possible, the manufacturing footprint is reduced, supporting corporate sustainability goals and enhancing the brand reputation of the supplier. This environmental compatibility future-proofs the production asset against tightening regulatory frameworks, ensuring long-term operational viability and reducing the risk of compliance-related shutdowns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, providing clarity for stakeholders evaluating its integration into their existing workflows. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details is essential for assessing the feasibility of adopting this method for specific project requirements and for anticipating the operational changes required to leverage its full potential.
Q: What are the advantages of the FeCl3-promoted method over traditional triazole synthesis?
A: The FeCl3-promoted method eliminates the need for harsh anhydrous or oxygen-free conditions, utilizes cheap and readily available starting materials, and offers a broader substrate scope compared to conventional methods that often suffer from low yields and narrow applicability.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the patent explicitly states that the method can be easily scaled up to the gram level and beyond, providing significant convenience for industrial scale production applications due to its simple operation and post-processing.
Q: What types of substituents are tolerated in this triazole synthesis?
A: The method demonstrates wide functional group tolerance, accommodating various substituted or unsubstituted aryl groups, alkyl, and alkenyl chains, allowing for the design of diverse 3,4-substituted trifluoromethyl triazole derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,4-Triazole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a manufacturing partner who can translate complex patent technologies into reliable commercial reality for your most demanding projects. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from process development to full-scale manufacturing is seamless and efficient. We are committed to meeting stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. Our expertise in handling fluorine chemistry and heterocyclic synthesis positions us as a leader in the field, capable of delivering the high-purity intermediates necessary for the next generation of life-saving medicines.
We invite you to contact our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific production needs and cost targets. Request a Customized Cost-Saving Analysis today to understand the potential economic benefits of switching to this superior manufacturing method for your supply chain. We are ready to provide specific COA data and route feasibility assessments to support your internal review processes and accelerate your project timelines. Partner with us to secure a stable, cost-effective, and high-quality supply of 5-trifluoromethyl-1,2,4-triazole derivatives that will empower your research and commercial success.
