Revolutionizing Triazole Manufacturing: A Metal-Free Route for High-Purity Pharmaceutical Intermediates
Revolutionizing Triazole Manufacturing: A Metal-Free Route for High-Purity Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and cost-effective methodologies for constructing nitrogen-containing heterocycles, particularly the versatile 1,2,4-triazole scaffold. Recent advancements detailed in patent CN113307790B introduce a groundbreaking preparation method for 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compounds that addresses critical bottlenecks in traditional synthesis. This technology leverages a metal-free oxidative cyclization strategy, utilizing inexpensive reagents like tetrabutylammonium iodide and tert-butyl peroxide to drive the reaction efficiently. The significance of this development cannot be overstated, as 1,2,4-triazoles serve as pivotal structural motifs in numerous bioactive molecules, ranging from antifungal agents like fluconazole to complex kinase inhibitors. 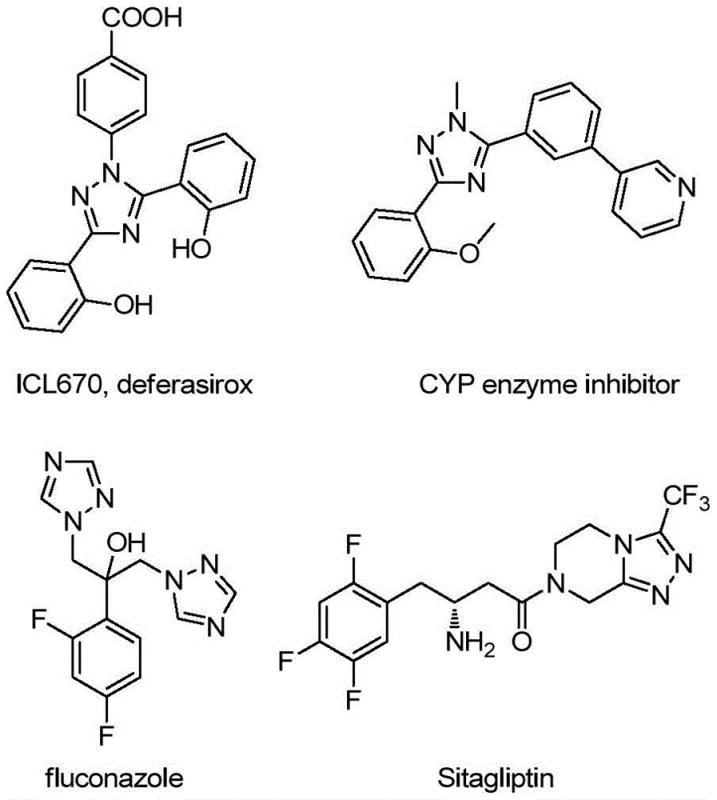
For R&D directors and process chemists, the ability to access these scaffolds without relying on precious metal catalysts represents a paradigm shift in process design. The patent outlines a procedure that operates under relatively mild thermal conditions, typically between 80°C and 100°C, and tolerates a wide array of functional groups. This robustness suggests that the method is not merely a laboratory curiosity but a viable pathway for the commercial scale-up of complex pharmaceutical intermediates. By eliminating the need for stringent anhydrous or oxygen-free environments, the process drastically simplifies reactor requirements and operational protocols, thereby enhancing the overall reliability of the supply chain for these high-value chemical entities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolyl-substituted 1,2,4-triazoles has been fraught with significant inefficiencies that hinder large-scale application. Traditional routes often rely on quinoline-2-carboxylic acid as a starting material, necessitating a cumbersome five-step reaction sequence to achieve the final target structure. This multi-step approach inherently suffers from cumulative yield losses, where the overall yield rarely exceeds 17%, rendering the process economically unviable for bulk manufacturing. Furthermore, these conventional methods frequently demand severe reaction conditions, including the use of toxic heavy metal catalysts and strict exclusion of moisture and air, which escalate both capital expenditure and operational complexity. The reliance on such harsh parameters not only increases the risk of safety incidents but also generates substantial volumes of hazardous waste, posing challenges for environmental compliance and waste management teams.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes a direct oxidative cyclization strategy that transforms readily available 2-methylquinoline derivatives and trifluoroethylimide hydrazides into the desired triazole products in a single pot. 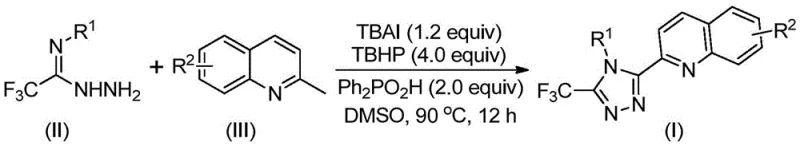
This streamlined approach bypasses the need for pre-functionalized carboxylic acid precursors, thereby shortening the synthetic timeline and improving atom economy. The reaction is promoted by a catalytic system comprising tetrabutylammonium iodide (TBAI) and tert-butyl peroxide (TBHP), which facilitates the oxidation of the methyl group to an aldehyde intermediate in situ, followed by condensation and cyclization. Experimental data from the patent indicates that this method can achieve impressive yields, with specific examples reporting conversion rates as high as 97% under optimized conditions. The operational simplicity is further enhanced by the use of common organic solvents like DMSO, which effectively dissolves the reactants and promotes the reaction without requiring exotic or highly specialized media.
Mechanistic Insights into TBAI/TBHP Promoted Oxidative Cyclization
The core of this technological breakthrough lies in the intricate interplay between the iodide catalyst and the peroxide oxidant, which drives the transformation through a radical-mediated pathway. The mechanism initiates with the oxidation of the methyl group on the 2-methylquinoline substrate (Formula III) to generate a reactive 2-quinoline carbaldehyde intermediate. This aldehyde then undergoes a condensation reaction with the trifluoroethylimide hydrazide (Formula II) to form a dehydrated hydrazone species. Subsequent oxidative iodination activates the hydrazone, triggering an intramolecular electrophilic substitution that closes the triazole ring. Finally, aromatization yields the stable 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compound (Formula I). 
From an impurity control perspective, this mechanism offers distinct advantages over transition-metal catalyzed couplings. The absence of palladium or copper eliminates the risk of metal leaching, a critical quality attribute for pharmaceutical intermediates intended for human consumption. Moreover, the mild reaction temperatures (80-100°C) minimize thermal degradation of sensitive functional groups, such as the trifluoromethyl moiety and various aryl substituents. The use of diphenylphosphoric acid as an additive further stabilizes the reaction environment, likely by modulating the acidity and preventing the formation of polymeric byproducts. This precise control over the reaction trajectory ensures a clean impurity profile, significantly reducing the burden on downstream purification processes like column chromatography or recrystallization.
How to Synthesize 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis in a production setting requires careful attention to reagent stoichiometry and thermal management to maximize yield and safety. The patent specifies a molar ratio of approximately 1.5:1:1.2:4:2 for the trifluoroethylimide hydrazide, 2-methylquinoline, TBAI, TBHP, and diphenylphosphoric acid, respectively. The reaction is typically conducted in dimethyl sulfoxide (DMSO) at a concentration that allows for effective mixing and heat transfer. While the laboratory scale uses Schlenk tubes, scaling this exothermic oxidative process requires robust temperature control systems to manage the decomposition of the peroxide safely. For detailed standardized operating procedures and safety protocols regarding this specific transformation, please refer to the technical guide below.
- Combine tetrabutylammonium iodide (TBAI), tert-butyl peroxide (TBHP), diphenylphosphoric acid, trifluoroethylimide hydrazide, and 2-methylquinoline derivative in an organic solvent such as DMSO.
- Heat the reaction mixture to a temperature between 80°C and 100°C and maintain stirring for 8 to 14 hours to ensure complete oxidative cyclization.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this metal-free synthesis route offers compelling economic and logistical benefits that directly impact the bottom line. The elimination of expensive transition metal catalysts, such as palladium or rhodium complexes, results in a drastic reduction in raw material costs. Furthermore, the removal of heavy metals from the process flow negates the need for specialized scavenging resins or complex filtration steps designed to meet strict residual metal limits, thereby simplifying the purification workflow and reducing processing time. The use of commodity chemicals like TBAI and aqueous TBHP ensures a stable and predictable supply chain, insulating manufacturers from the volatility often associated with precious metal markets.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of cheap and easily obtainable starting materials. 2-methylquinoline and aromatic amines used to prepare the hydrazide are bulk commodities available from multiple global suppliers, fostering competitive pricing. Additionally, the high atom efficiency of the one-pot reaction minimizes solvent usage and waste generation, leading to substantial cost savings in waste disposal and solvent recovery operations. The high yields reported, often exceeding 80% and reaching up to 97% for certain substrates, mean that less raw material is required to produce a kilogram of the final API intermediate, directly lowering the cost of goods sold (COGS).
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes significantly to supply chain resilience. Since the process does not require inert atmosphere techniques (e.g., nitrogen or argon blanketing) or strictly anhydrous solvents, it can be executed in standard glass-lined or stainless steel reactors found in most multipurpose chemical plants. This flexibility reduces the dependency on specialized equipment and allows for faster turnaround times between batches. The broad substrate tolerance means that a single manufacturing line can potentially produce a variety of analogues by simply swapping the starting quinoline or hydrazide, providing agility in responding to changing market demands for different drug candidates.
- Scalability and Environmental Compliance: Scaling this chemistry from gram to kilogram or tonne levels is facilitated by the homogeneous nature of the reaction mixture and the absence of heterogeneous metal catalysts that can cause fouling or filtration issues. The use of DMSO, a high-boiling polar aprotic solvent, allows for safe operation at the required temperatures while maintaining good solubility for all reagents. From an environmental standpoint, the avoidance of toxic heavy metals aligns with green chemistry principles and simplifies regulatory compliance regarding effluent discharge. The simplified post-treatment, involving filtration and standard chromatography or crystallization, ensures that the process remains efficient even at larger scales, supporting the commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on reaction parameters and substrate compatibility. Understanding these nuances is essential for process engineers evaluating the feasibility of integrating this technology into existing manufacturing portfolios.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, the patented method utilizes a metal-free catalytic system based on tetrabutylammonium iodide (TBAI) and tert-butyl peroxide (TBHP), significantly reducing raw material costs and eliminating the need for complex heavy metal removal processes.
Q: What is the substrate scope for the quinoline component?
A: The method demonstrates excellent tolerance for various substituents on the quinoline ring, including hydrogen, methyl, methoxy, halogens (Cl, Br), and nitro groups at positions 6, 7, or 8, allowing for diverse structural modifications.
Q: Is the reaction sensitive to air or moisture?
A: Unlike many traditional organometallic couplings, this oxidative cyclization does not require strict anhydrous or anaerobic conditions, making it highly suitable for large-scale industrial production with simplified operational requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free oxidative cyclization technology for the production of high-value heterocyclic intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of batch size. Our state-of-the-art facilities are equipped to handle oxidative reactions safely, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the demanding requirements of the global pharmaceutical industry. We are committed to delivering high-purity 3-quinolyl-5-trifluoromethyl-1,2,4-triazole derivatives that serve as critical building blocks for next-generation therapeutics.
We invite you to collaborate with us to leverage this advanced synthesis route for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this efficient process can optimize your budget. Please contact our technical procurement team today to request specific COA data for our available catalog compounds or to discuss route feasibility assessments for your proprietary molecules. Let us help you accelerate your drug development timeline with reliable, cost-effective, and scalable chemical solutions.
