Advanced Synthesis of Tucatinib Intermediate: A Scalable Commercial Route for API Manufacturing
Advanced Synthesis of Tucatinib Intermediate: A Scalable Commercial Route for API Manufacturing
The rapid advancement of targeted cancer therapies has placed immense pressure on the supply chain for high-quality pharmaceutical intermediates. Specifically, the synthesis of Tucatinib (ONT-380), a potent HER2 inhibitor approved for breast cancer treatment, relies heavily on the efficient production of its key precursor, 4-([1,2,4]triazolo[1,5-a]pyridin-7-yloxy)-3-methylaniline. Recent intellectual property developments, specifically patent CN112898298A filed in June 2021, disclose a groundbreaking preparation method that addresses critical bottlenecks in existing manufacturing protocols. This technical insight report analyzes the novel synthetic pathway, highlighting its potential to redefine cost structures and supply reliability for global API manufacturers seeking a reliable pharmaceutical intermediate supplier.
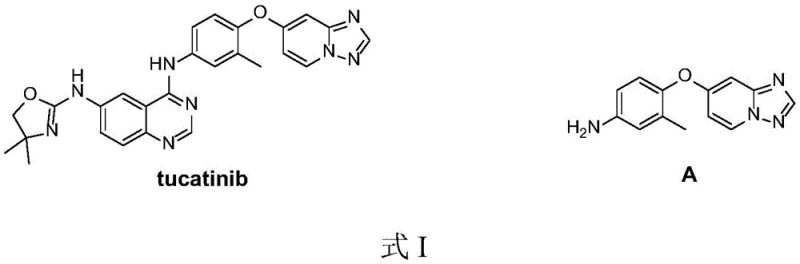
The structural complexity of Tucatinib necessitates a robust synthetic strategy for its intermediates to ensure consistent quality and availability. As illustrated in the chemical structures, the target molecule incorporates a fused triazolopyridine system linked via an ether bond to a substituted aniline moiety. The efficiency of constructing this specific heterocyclic scaffold directly impacts the commercial viability of the final drug substance. The disclosed patent provides a streamlined approach that bypasses the cumbersome multi-step sequences traditionally associated with this chemical class, offering a compelling value proposition for procurement teams focused on cost reduction in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the industrial preparation of the Tucatinib intermediate was plagued by significant inefficiencies and operational hazards. As detailed in the background art (referencing methods such as WO 2007059257), conventional routes often involved upwards of eight to ten synthetic steps, necessitating the use of sensitive protecting groups like tert-butyloxycarbonyl (Boc). These traditional pathways frequently relied on precious metal catalysis, specifically palladium-mediated cross-couplings and hydrogenolysis, which introduce heavy metal contamination risks and require expensive scavenging processes to meet regulatory limits. Furthermore, alternative direct substitution methods reported in other literature suffered from extremely harsh reaction conditions, requiring temperatures of 140°C for extended periods of 3 to 5 days, yet still delivering mediocre yields ranging from only 40% to 60%.
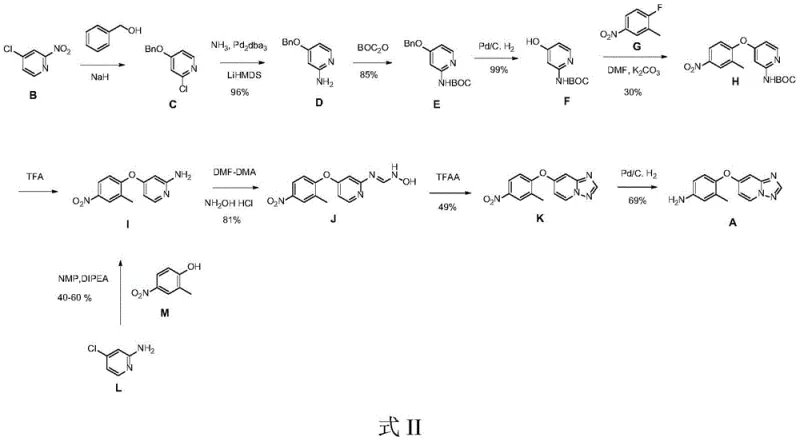
The reliance on column chromatography for purification in several of these legacy routes represents a major barrier to commercial scale-up. In a manufacturing environment, column chromatography is notoriously difficult to scale, consumes vast amounts of solvent, and drastically reduces throughput. The combination of low overall yields, high material costs due to protecting groups, and the safety risks associated with high-temperature, long-duration reactions created a fragile supply chain. For supply chain heads, these factors translated into unpredictable lead times and elevated inventory costs, underscoring the urgent need for a more robust and economically viable synthetic alternative.
The Novel Approach
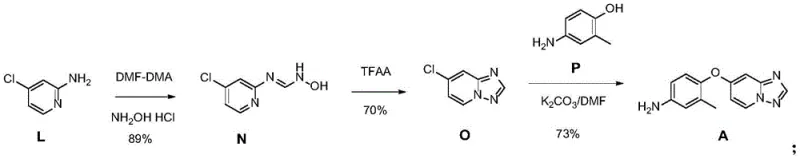
The methodology described in patent CN112898298A introduces a concise three-step sequence that fundamentally simplifies the construction of the triazolopyridine core. Instead of building the molecule from disparate fragments with extensive protection and deprotection cycles, this novel route utilizes a convergent strategy starting from readily available 2-amino-4-chloropyridine. The process initiates with a formamidination reaction followed by cyclization to establish the heterocyclic ring system early in the synthesis. This strategic reordering of bond formation allows for the introduction of the aniline fragment in the final step via a straightforward nucleophilic aromatic substitution.
By eliminating the need for palladium catalysts and high-pressure hydrogenation equipment, the new process significantly lowers the barrier to entry for manufacturing facilities. The reaction conditions are notably milder, with the final coupling step proceeding efficiently in a DMF/potassium carbonate system at 130-140°C, but crucially, without the multi-day duration of previous methods. The ability to isolate intermediates through simple crystallization rather than chromatographic separation marks a pivotal improvement in process chemistry. This shift not only enhances the environmental profile of the synthesis by reducing solvent waste but also dramatically improves the scalability, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Triazolopyridine Cyclization and Substitution
The core of this synthetic innovation lies in the efficient construction of the [1,2,4]triazolo[1,5-a]pyridine ring system. The mechanism begins with the reaction of 2-amino-4-chloropyridine with N,N-dimethylformamide dimethyl acetal (DMF-DMA), which acts as a formylating agent to generate an amidine intermediate in situ. Subsequent treatment with hydroxylamine hydrochloride facilitates the formation of the N-hydroxyformamide derivative (Compound N). This transformation is highly selective, leveraging the nucleophilicity of the exocyclic amine to install the necessary nitrogen and carbon atoms required for ring closure. The subsequent cyclization step, mediated by trifluoroacetic anhydride (TFAA), proceeds through an activation of the hydroxyl group, promoting an intramolecular attack that closes the triazole ring to yield 7-chloro-[1,2,4]triazolo[1,5-a]pyridine (Compound O).
From an impurity control perspective, this route offers distinct advantages over transition-metal catalyzed alternatives. The absence of palladium removes the risk of residual metal impurities, which are strictly regulated in final drug substances. Furthermore, the high regioselectivity of the nucleophilic aromatic substitution in the final step ensures that the ether linkage forms exclusively at the 7-position of the triazolopyridine ring. The electron-withdrawing nature of the triazole ring activates the C-Cl bond towards nucleophilic attack by the phenol, while the reaction conditions (K2CO3 in DMF) are optimized to minimize side reactions such as hydrolysis or over-alkylation. This precise control over reaction pathways results in a crude product profile that is amenable to simple recrystallization, consistently achieving purity levels exceeding 99.5%.
How to Synthesize 4-([1,2,4]triazolo[1,5-a]pyridin-7-yloxy)-3-methylaniline Efficiently
The execution of this synthesis requires careful attention to stoichiometry and temperature control to maximize the impressive yields reported in the patent data. The process is designed to be operationally simple, utilizing common organic solvents and reagents that are globally sourced. The initial formamidination and cyclization steps set the stage for a high-purity intermediate, which is critical for the success of the final coupling reaction. Operators should note that while the reaction temperatures are elevated, they are well within the standard operating range of industrial glass-lined or stainless steel reactors, ensuring that the process is compatible with existing infrastructure without requiring specialized high-pressure equipment.
- React 2-amino-4-chloropyridine with DMF-DMA followed by hydroxylamine hydrochloride to form N'-(4-chloropyridin-2-yl)-N-hydroxyformamide.
- Cyclize the intermediate using trifluoroacetic anhydride (TFAA) to generate 7-chloro-[1,2,4]triazolo[1,5-a]pyridine.
- Perform nucleophilic substitution with 4-amino-2-methylphenol in DMF/K2CO3 to yield the final Tucatinib intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route translates into tangible strategic benefits beyond mere technical elegance. The primary driver of value is the drastic simplification of the manufacturing workflow. By reducing the number of unit operations and eliminating the need for chromatographic purification, the facility throughput is significantly increased. This efficiency gain directly correlates to a reduction in the cost of goods sold (COGS), allowing for more competitive pricing models in the tendering process. The reliance on commodity chemicals like DMF-DMA and TFAA, rather than bespoke catalysts, further insulates the supply chain from volatility in the precious metals market.
- Cost Reduction in Manufacturing: The elimination of palladium catalysts and protecting group reagents removes some of the most expensive line items from the bill of materials. Additionally, the removal of column chromatography reduces solvent consumption and waste disposal costs substantially. The high yields achieved at each step (up to 89% in the first step) mean that less raw material is wasted, maximizing the atom economy of the process and driving down the effective cost per kilogram of the intermediate.
- Enhanced Supply Chain Reliability: The use of robust, non-sensitive reagents ensures that the process is less prone to batch failures caused by catalyst poisoning or moisture sensitivity. The shortened reaction times, particularly in the final coupling step which reduces from days to hours compared to some prior art, allow for faster turnaround times. This agility enables manufacturers to respond more quickly to fluctuations in demand, reducing the need for excessive safety stock and improving cash flow dynamics for the entire supply network.
- Scalability and Environmental Compliance: The process is inherently greener due to the reduction in solvent usage and the avoidance of heavy metals. This aligns with increasingly stringent environmental regulations and corporate sustainability goals. The simplicity of the work-up procedures, primarily involving filtration and crystallization, makes the technology transfer to large-scale production units seamless. This scalability ensures that the supply of this critical intermediate can be ramped up to meet commercial demands without the engineering bottlenecks typically associated with complex multi-step syntheses.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the operational feasibility and quality implications for potential partners.
Q: What are the key advantages of the new synthesis route compared to prior art?
A: The new route eliminates the need for expensive palladium catalysts, protecting group manipulations (Boc), and column chromatography purification, resulting in a shorter process time and significantly lower production costs.
Q: What is the overall yield and purity profile of this method?
A: The process achieves high yields at each step (up to 89% for step 1, 70% for step 2, and 73% for step 3) with final product purity exceeding 99.5% after simple recrystallization.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the method uses readily available raw materials, mild reaction conditions, and avoids hazardous high-pressure hydrogenation steps found in previous methods, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-([1,2,4]triazolo[1,5-a]pyridin-7-yloxy)-3-methylaniline Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of oncology drugs like Tucatinib depends on a secure and high-quality supply of key intermediates. Our R&D team has thoroughly analyzed the pathway described in CN112898298A and validated its potential for industrial application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of material that meets rigorous quality standards. Our state-of-the-art facilities are equipped with stringent purity specifications and rigorous QC labs capable of detecting impurities at trace levels, guaranteeing that every batch of 4-([1,2,4]triazolo[1,5-a]pyridin-7-yloxy)-3-methylaniline supports your regulatory filings and clinical needs.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to leverage this advanced synthetic technology. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing capabilities can enhance your supply chain resilience and drive down your overall production costs.
