Advanced Intramolecular Cyclization for High-Purity Quinoline Derivatives and Commercial Scale-Up
Introduction to Patent CN116410136A
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex heterocyclic scaffolds, particularly quinoline derivatives which serve as critical cores for numerous bioactive molecules. Patent CN116410136A, published in July 2023, introduces a groundbreaking methodology for the preparation of quinoline derivatives that fundamentally shifts the paradigm from traditional intermolecular condensation to a highly efficient intramolecular dehydrogenation cyclization. This technology leverages phenyl azide ketone derivatives as key substrates, utilizing strong organic acids to drive the ring-closing process under remarkably mild conditions. For R&D directors and procurement specialists, this represents a significant opportunity to streamline supply chains for high-value intermediates used in the synthesis of drugs like Topotecan and RORgt modulators. The process eliminates the need for pre-installed carbon-carbon double bonds at the 1,2-positions of the raw materials, thereby vastly expanding the scope of accessible substrates and enhancing the practical utility of the synthetic route for commercial manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinoline skeleton has relied heavily on classical named reactions such as the Skraup, Doebner-Miller, or Combes syntheses, which predominantly utilize aniline or its derivatives as starting materials. These conventional strategies often suffer from significant drawbacks that hinder their applicability in modern green chemistry and large-scale production environments. Many of these traditional routes require the use of expensive and toxic metal oxidants, harsh acidic conditions, or high temperatures that can lead to poor selectivity and the formation of complex impurity profiles. Furthermore, existing literature indicates that most synthetic strategies focus on derivatization at the 1, 2, 3, or 4 positions of the quinoline ring, while direct functionalization on the benzene ring, particularly at the 6-position, remains challenging and often requires multi-step protection and deprotection sequences. The reliance on intermolecular reactions also limits the structural diversity that can be achieved in a single operational step, forcing manufacturers to endure longer lead times and higher material costs.
The Novel Approach
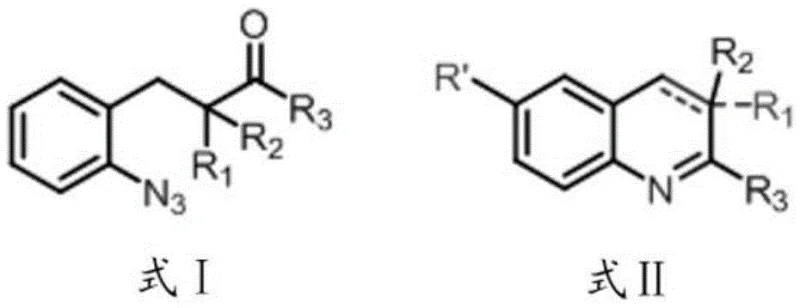
In stark contrast to these legacy methods, the technology disclosed in patent CN116410136A employs a novel intramolecular dehydrogenation ring-closing reaction driven by strong protonic acids. By utilizing phenyl azide ketone derivatives (Formula I) as the substrate, the process enables the direct formation of the quinoline ring structure (Formula II) without the prerequisite of a 1,2-carbon-carbon double bond in the starting material. This mechanistic shift allows for the simultaneous construction of the quinoline core and derivatization on the benzene ring, effectively solving the long-standing challenge of 6-position functionalization. The use of organic acids such as trifluoromethanesulfonic acid (TfOH) or methanesulfonic acid (MsOH) not only simplifies the reaction setup but also ensures high yields, with reported efficiencies reaching up to 90 percent under optimized conditions. This approach significantly reduces the equipment requirements and operational complexity, making it an ideal candidate for reliable quinoline derivative supplier networks aiming for cost-effective production.
Mechanistic Insights into Organic Acid Catalyzed Cyclization
The core of this technological advancement lies in the unique role of strong protonic acids in facilitating the dehydrogenation and cyclization steps. Unlike transition metal catalysts that often leave behind trace metal impurities requiring costly removal processes, the organic acids employed here act as clean promoters. The mechanism likely involves the protonation of the azide or carbonyl functionality, increasing the electrophilicity of the system and promoting an intramolecular nucleophilic attack by the nitrogen species. This is followed by a dehydrogenation step that aromatizes the newly formed pyridine ring, completing the quinoline scaffold. The inventors have meticulously optimized the molar ratios of the organic acid to the substrate, finding that a ratio of 3:1 for TfOH or 5:1 to 8:1 for MsOH provides the optimal balance between reaction rate and product purity. This precise control over stoichiometry is crucial for minimizing side reactions and ensuring that the final product meets the stringent purity specifications required for pharmaceutical intermediates.
Furthermore, the impurity control mechanism inherent in this process is superior to oxidative methods. Because the reaction proceeds under inert gas atmosphere and relatively low temperatures ranging from -40°C to 25°C, thermal degradation and uncontrolled oxidation are minimized. The absence of heavy metal oxidants means that the impurity profile is dominated by organic by-products that are generally easier to separate via standard chromatographic techniques or crystallization. For quality control teams, this translates to a more predictable and manageable purification process. The ability to tune the reaction temperature, with an optimal range identified between -20°C and 25°C, allows manufacturers to fine-tune the kinetics to favor the desired regioisomer, thereby enhancing the overall quality of the high-purity quinoline derivatives produced. This level of mechanistic understanding provides a robust foundation for scaling the process from laboratory grams to industrial tons.
How to Synthesize Quinoline Derivative T-1 Efficiently
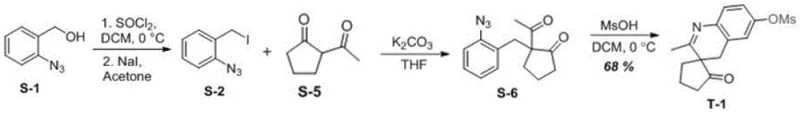
To implement this synthesis in a practical setting, one must adhere to the specific procedural details outlined in the patent examples, particularly regarding the preparation of the azide precursor and the subsequent cyclization. The process begins with the conversion of 2-azidobenzyl alcohol to the corresponding iodide, followed by alkylation with a cyclic ketone to generate the key phenyl azide ketone intermediate. The critical cyclization step requires careful addition of the organic acid to a solution of the precursor in dichloromethane at 0°C to manage the exotherm and ensure selectivity. Detailed standardized synthesis steps for producing compound T-1, including specific reagent quantities and workup procedures, are provided in the structured guide below to assist technical teams in replicating these results.
- Preparation of Azide Intermediate: Convert 2-azidobenzyl alcohol to 2-azidobenzyl iodide using thionyl chloride and sodium iodide in acetone.
- Alkylation Reaction: React the azide iodide with a cyclic ketone derivative in the presence of potassium carbonate and THF to form the phenyl azide ketone precursor.
- Acid-Promoted Cyclization: Treat the precursor with methanesulfonic acid (6 equivalents) in dichloromethane at 0°C to induce intramolecular dehydrogenation and ring closure.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers substantial advantages that directly address the pain points of procurement managers and supply chain heads. The elimination of expensive transition metal catalysts and ligands drastically simplifies the raw material sourcing strategy, reducing dependency on volatile metal markets and lowering the overall bill of materials. Additionally, the simplified workup procedure, which avoids complex metal scavenging steps, leads to significant time savings in the production cycle. This efficiency gain allows for faster turnover of manufacturing batches, thereby enhancing the responsiveness of the supply chain to market demands. The use of common organic solvents like dichloromethane and readily available organic acids further contributes to cost reduction in pharmaceutical intermediate manufacturing, making the process economically viable for large-scale operations.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the complete removal of precious metal catalysts and stoichiometric metal oxidants, which are traditionally expensive and difficult to dispose of. By replacing these with commodity organic acids, the direct material costs are significantly lowered. Furthermore, the high yield of up to 90 percent minimizes raw material waste, ensuring that a greater proportion of the input mass is converted into saleable product. The simplified purification process also reduces the consumption of silica gel and solvents during chromatography, leading to additional operational savings that accumulate over large production volumes.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, such as azidobenzyl derivatives and cyclic ketones, are commercially available and stable, reducing the risk of supply disruptions associated with specialized reagents. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent choices, ensures consistent production output even with minor variations in raw material quality. This reliability is critical for maintaining continuous supply lines for downstream API manufacturers who depend on timely delivery of key intermediates. The ability to produce diverse derivatives from a common platform further strengthens supply security by allowing for flexible production scheduling based on demand.
- Scalability and Environmental Compliance: The reaction operates at near-ambient temperatures (-20°C to 25°C), which means it can be scaled up using standard glass-lined or stainless steel reactors without the need for specialized cryogenic or high-pressure equipment. This lowers the capital expenditure required for capacity expansion. From an environmental standpoint, the absence of heavy metal waste streams simplifies effluent treatment and aligns with increasingly strict global environmental regulations. The generation of less hazardous waste reduces disposal costs and enhances the sustainability profile of the manufacturing site, a key factor for modern corporate social responsibility goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this quinoline synthesis technology. These answers are derived directly from the experimental data and technical disclosures within patent CN116410136A, providing clarity on reaction scope, optimization parameters, and downstream utility. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific product portfolios.
Q: What are the advantages of this quinoline synthesis over traditional Skraup methods?
A: Unlike traditional methods requiring harsh oxidants and high temperatures, this patent utilizes mild organic acids (MsOH/TfOH) at low temperatures (-20°C to 25°C), eliminating heavy metal waste and improving safety profiles.
Q: Can this method achieve derivatization on the benzene ring of the quinoline scaffold?
A: Yes, a key innovation of patent CN116410136A is the ability to perform derivatization on the benzene ring (specifically the 6-position) during the one-step skeleton construction, which is difficult with conventional intermolecular approaches.
Q: What yields can be expected from this organic acid catalyzed process?
A: The patent reports optimized yields reaching up to 90% when using trifluoromethanesulfonic acid with a specific molar ratio of 3:1, significantly outperforming many traditional multi-step sequences.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoline Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methodologies described in patent CN116410136A for the production of high-value quinoline intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of materials. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of quinoline derivative delivered meets the highest industry standards. We are committed to leveraging our technical expertise to optimize these novel routes for maximum efficiency and cost-effectiveness.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this metal-free process. We encourage you to contact us today to obtain specific COA data for our available quinoline intermediates and to receive comprehensive route feasibility assessments tailored to your project requirements. Together, we can accelerate the development of next-generation pharmaceuticals through innovative chemistry and reliable manufacturing partnerships.
