Advanced Palladium-Catalyzed Carbonylation for Scalable 2,3-Dihydroquinolone Pharmaceutical Intermediates
Advanced Palladium-Catalyzed Carbonylation for Scalable 2,3-Dihydroquinolone Pharmaceutical Intermediates
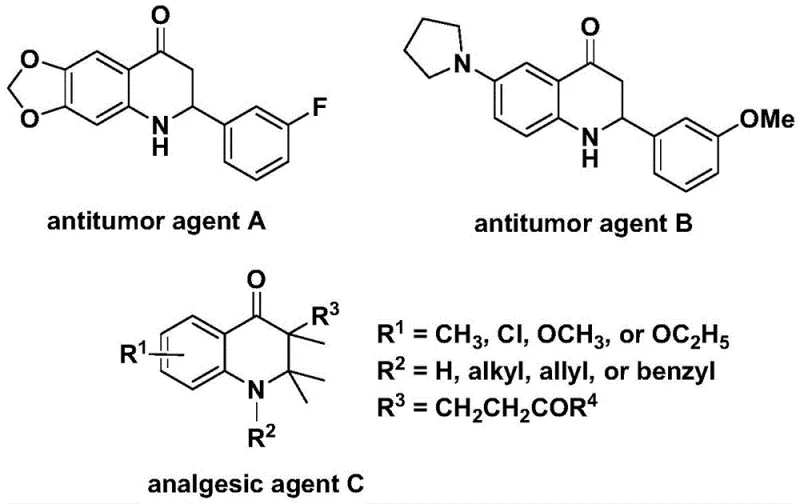
The development of efficient synthetic routes for nitrogen-containing heterocycles remains a cornerstone of modern medicinal chemistry, particularly for scaffolds exhibiting potent biological activities. Patent CN112239456B introduces a robust and versatile preparation method for substituted 2,3-dihydroquinolone compounds, a class of molecules renowned for their presence in bioactive agents ranging from antitumor drugs to analgesics. As illustrated in the structural diversity of known bioactive molecules, the 2,3-dihydroquinolone core is a privileged structure in drug discovery, underpinning the efficacy of agents such as antitumor agent A, antitumor agent B, and analgesic agent C. This patent discloses a transition metal palladium-catalyzed carbonylation reaction that utilizes N-pyridine sulfonyl-o-iodoaniline and olefins as starting materials, offering a streamlined pathway to these valuable intermediates.
The significance of this technology lies in its ability to construct the carbonyl-containing six-membered heterocyclic ring directly and efficiently. Unlike traditional methods that may rely on hazardous gaseous carbon monoxide or multi-step sequences with poor atom economy, this novel approach employs a solid carbon monoxide substitute, thereby enhancing process safety and operational simplicity. For R&D directors and process chemists, the method offers a high degree of designability, allowing for the synthesis of both 2-aryl and 3-alkyl substituted derivatives through simple substrate modification. This flexibility is crucial for generating diverse libraries during lead optimization phases in pharmaceutical development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,3-dihydroquinolone skeletons has faced significant hurdles regarding safety, scalability, and functional group tolerance. Conventional carbonylation reactions often necessitate the use of high-pressure carbon monoxide gas, which poses severe safety risks in both laboratory and industrial settings due to its toxicity and flammability. Furthermore, many existing protocols suffer from limited substrate scope, failing to accommodate sensitive functional groups or sterically hindered olefins, which restricts their utility in synthesizing complex drug candidates. The reliance on harsh reaction conditions can also lead to decomposition of sensitive intermediates, resulting in lower yields and difficult purification processes that increase overall production costs and waste generation.
The Novel Approach
The methodology described in patent CN112239456B overcomes these challenges by utilizing a palladium-catalyzed system with a solid CO surrogate, specifically 1,3,5-trimesic acid phenol ester (TFBen). This innovation eliminates the need for high-pressure gas equipment, making the reaction accessible in standard glassware and scalable reactors alike. The reaction proceeds under relatively mild thermal conditions, typically between 100 °C and 120 °C, and demonstrates exceptional compatibility with a wide array of functional groups including halogens, alkyls, and silyl groups. By enabling the direct coupling of N-pyridine sulfonyl-o-iodoaniline with various olefins, this approach provides a convergent synthesis strategy that significantly reduces step count and improves overall process efficiency for manufacturing high-purity pharmaceutical intermediates.
Mechanistic Insights into Palladium-Catalyzed Carbonylative Cyclization
The reaction mechanism involves a sophisticated catalytic cycle driven by the palladium center, which orchestrates the formation of multiple bonds in a single operation. Initially, the palladium catalyst undergoes oxidative addition into the carbon-iodine bond of the N-pyridine sulfonyl-o-iodoaniline substrate, generating a reactive aryl-palladium intermediate. This step is critical as it activates the aromatic ring for subsequent transformations. Following this activation, carbon monoxide, which is released in situ from the decomposition of the TFBen additive, inserts into the aryl-palladium bond to form an acyl-palladium species. This insertion step effectively builds the carbonyl functionality directly into the growing molecular framework.
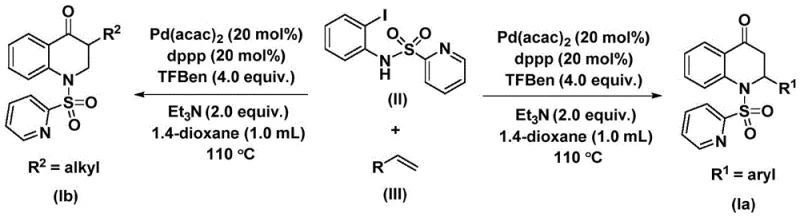
Subsequently, the olefin substrate coordinates to the palladium center and undergoes migratory insertion into the acyl-palladium bond, creating a new carbon-carbon bond and extending the carbon chain. The final step involves an intramolecular reductive elimination or cyclization event that closes the six-membered ring, releasing the substituted 2,3-dihydroquinolone product and regenerating the active palladium catalyst. This mechanistic pathway ensures high atom economy and minimizes the formation of byproducts. The use of ligands such as 1,3-bis(diphenylphosphino)propane (dppp) stabilizes the palladium species throughout the cycle, preventing catalyst deactivation and ensuring consistent performance across different substrate classes, which is vital for maintaining batch-to-batch consistency in commercial production.
How to Synthesize Substituted 2,3-Dihydroquinolone Efficiently
The practical execution of this synthesis is designed for ease of operation, requiring standard laboratory equipment and commercially available reagents. The process begins by combining the palladium catalyst, ligand, base, CO source, and substrates in an aprotic organic solvent such as 1,4-dioxane. The mixture is then heated to facilitate the reaction, after which standard workup procedures involving filtration and chromatography yield the pure product. This straightforward protocol minimizes the need for specialized handling techniques, making it highly attractive for rapid process development and scale-up activities.
- Charge a reaction vessel with bis(acetylacetone)palladium, dppp ligand, triethylamine, TFBen (CO source), N-pyridine sulfonyl-o-iodoaniline, and the desired olefin substrate in an organic solvent like 1,4-dioxane.
- Heat the reaction mixture to a temperature range of 100-120 °C and maintain stirring for a duration of 24 to 48 hours to ensure complete conversion.
- Upon completion, filter the mixture, adsorb the crude product onto silica gel, and purify via column chromatography to isolate the target substituted 2,3-dihydroquinolone.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthetic route offers substantial strategic benefits, primarily driven by the simplification of raw material sourcing and hazard management. The shift from gaseous carbon monoxide to a solid ester substitute drastically reduces the regulatory burden and infrastructure costs associated with handling toxic gases. This change not only enhances workplace safety but also streamlines the logistics of raw material storage and transport, as solid reagents are generally more stable and easier to manage than compressed gases. Consequently, this leads to a more resilient supply chain capable of withstanding disruptions related to hazardous material transport regulations.
- Cost Reduction in Manufacturing: The elimination of high-pressure reactor requirements and the associated safety systems translates to significant capital expenditure savings for manufacturing facilities. Additionally, the high reaction efficiency and broad substrate tolerance reduce the need for expensive protecting group strategies or low-yielding alternative routes. By minimizing waste generation and simplifying purification steps through high conversion rates, the overall cost of goods sold (COGS) for these pharmaceutical intermediates is effectively optimized without compromising on quality standards.
- Enhanced Supply Chain Reliability: The starting materials, including the palladium catalyst, ligands, and olefins, are widely available from global chemical suppliers, ensuring a stable and continuous supply stream. The robustness of the reaction conditions means that production schedules are less likely to be impacted by minor variations in raw material quality or environmental factors. This reliability is critical for maintaining just-in-time inventory levels and meeting the stringent delivery timelines demanded by downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The protocol is inherently scalable, having been demonstrated to work effectively from milligram to gram scales with consistent results. The use of a solid CO source aligns with green chemistry principles by reducing the risk of accidental releases and lowering the overall environmental footprint of the manufacturing process. This compliance with increasingly strict environmental regulations facilitates smoother permitting processes for new production lines and supports corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this palladium-catalyzed carbonylation technology. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, aiming to clarify operational parameters and expected outcomes for potential licensees or manufacturing partners.
Q: What is the primary advantage of using TFBen in this synthesis?
A: TFBen (1,3,5-trimethoxybenzene phenol ester) serves as a solid, safe, and easy-to-handle substitute for toxic carbon monoxide gas, significantly improving operational safety and simplifying reactor requirements for industrial scale-up.
Q: What types of substrates are compatible with this palladium catalytic system?
A: The method demonstrates excellent substrate compatibility, successfully accommodating various olefins including aryl-substituted alkenes (with electron-donating or withdrawing groups), linear and branched alkyl alkenes, and even silyl-substituted alkenes.
Q: How does the reaction temperature affect the yield?
A: Optimal yields are achieved at temperatures between 100 °C and 120 °C. Lower temperatures may result in incomplete conversion due to insufficient activation energy for the oxidative addition and insertion steps, while the specified range ensures high efficiency within 48 hours.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Dihydroquinolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly transitioned into full-scale manufacturing. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee product integrity.
We invite you to collaborate with us to leverage this innovative carbonylation technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and timeline. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive efficiency and value in your supply chain.
