Advanced Palladium-Catalyzed Synthesis for High-Purity Pharmaceutical Intermediates at Commercial Scale
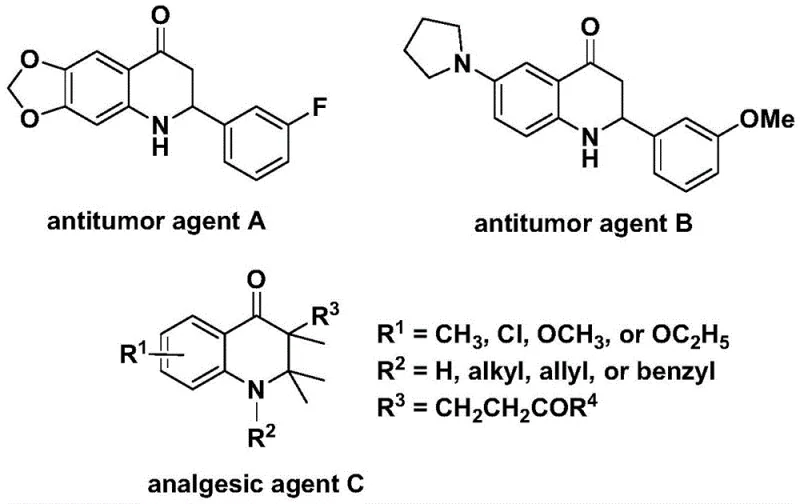
Patent CN112239456B introduces a groundbreaking palladium-catalyzed methodology for synthesizing substituted 2,3-dihydroquinolone compounds that serve as essential scaffolds in critical pharmaceutical agents. This innovative approach addresses longstanding industry challenges by enabling direct carbonylation using commercially available starting materials including N-pyridine sulfonyl-o-iodoaniline and olefins under mild reaction conditions. The process operates efficiently at temperatures between 100°C and 120°C over a controlled timeframe of 24 to 48 hours in dioxane solvent, yielding high-purity products with exceptional functional group tolerance across diverse substrates. Notably, the synthesized compounds include antitumor agents A and B which demonstrate significant activity against human cancer cells as documented in J.Med.chem. literature from 1998 and 2000 respectively, alongside analgesic agent C with potent pain-relieving properties reported in earlier studies. This patent represents a strategic advancement for pharmaceutical manufacturers seeking reliable routes to complex heterocyclic intermediates while maintaining stringent quality requirements essential for drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes to dihydroquinolone scaffolds often require multi-step sequences involving harsh reaction conditions such as strong acids or high temperatures that compromise functional group compatibility and increase impurity profiles. These methods typically suffer from low yields due to competing side reactions when processing substrates containing sensitive functional groups like halogens or alkoxy moieties. The necessity for specialized reagents and extensive purification protocols significantly elevates production costs while introducing scalability challenges during commercial manufacturing transitions. Furthermore, conventional cyclization techniques frequently generate toxic byproducts requiring complex waste treatment procedures that conflict with modern environmental regulations. The limited substrate scope of existing methodologies restricts structural diversity in final products, thereby constraining medicinal chemistry optimization efforts for drug discovery programs targeting specific biological pathways.
The Novel Approach
The patented methodology overcomes these limitations through an elegant palladium-catalyzed carbonylation strategy that operates under significantly milder conditions while maintaining exceptional efficiency across diverse substrates. By utilizing N-pyridine sulfonyl-o-iodoaniline as a directing group precursor, the process achieves remarkable functional group tolerance including methyl, chloro, fluoro, and methoxy substituents without requiring protective group strategies. The carefully optimized catalyst system comprising Pd(acac)₂ and dppp ligand enables high conversion rates at moderate temperatures between 100°C and 120°C within a practical timeframe of 24 to 48 hours. This streamlined approach eliminates multiple intermediate isolation steps while generating minimal byproducts through its atom-economical design. The inherent simplicity of the reaction setup combined with standard purification techniques ensures seamless scalability from laboratory to industrial production environments without compromising product quality or consistency.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The catalytic cycle begins with oxidative addition where palladium inserts into the carbon-nitrogen bond of N-pyridine sulfonyl-o-iodoaniline to form a key aryl palladium intermediate that serves as the foundation for subsequent transformations. Carbon monoxide released from the triethylamine additive coordinates with this intermediate to generate an acyl palladium species through migratory insertion, establishing the carbonyl functionality essential for the dihydroquinolone core structure. The olefin substrate then coordinates with the acyl palladium complex followed by alkene insertion that forms a new carbon-carbon bond while extending the molecular framework. This critical step determines the regioselectivity of the final product based on the olefin's substitution pattern. Reductive elimination subsequently occurs to release the substituted dihydroquinolone compound while regenerating the active palladium catalyst for further cycles. The precise control over each mechanistic step ensures high regioselectivity and minimizes undesired side products through careful optimization of ligand-to-metal ratios and reaction temperature parameters.
Impurity control is achieved through strategic substrate design that leverages the directing effect of the pyridine sulfonyl group to prevent undesired cyclization pathways or isomerization byproducts commonly observed in alternative synthetic routes. The reaction conditions specifically avoid strong acids or bases that could promote decomposition pathways or racemization in chiral centers when present in certain substrates. Careful selection of dioxane as the solvent medium provides optimal polarity balance that facilitates intermediate stability while preventing solvolysis side reactions that might occur in more protic environments. The standardized post-treatment protocol involving silica gel filtration followed by column chromatography effectively removes residual catalysts and minor impurities without requiring specialized equipment or hazardous solvents. This integrated approach ensures consistent production of compounds meeting pharmaceutical purity standards while maintaining excellent yield reproducibility across different substrate types as demonstrated in multiple experimental examples.
How to Synthesize Substituted 2,3-Dihydroquinolone Efficiently
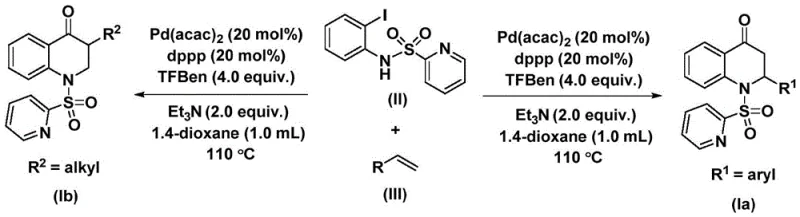
This patented methodology provides a robust framework for synthesizing structurally diverse substituted dihydroquinolone compounds through a carefully optimized palladium-catalyzed carbonylation process that demonstrates exceptional operational simplicity and scalability potential. The reaction sequence leverages commercially available starting materials including N-pyridine sulfonyl-o-iodoaniline derivatives and various olefin substrates that can be readily sourced from standard chemical suppliers without requiring specialized handling procedures. Key advantages include precise control over regioselectivity through substrate engineering capabilities that allow targeted synthesis of both aryl-substituted (Ia) and alkyl-substituted (Ib) product variants based on specific application requirements. The process maintains consistent performance across different scales while delivering high-quality intermediates suitable for direct incorporation into pharmaceutical manufacturing workflows without additional purification steps beyond standard chromatography protocols.
- Add palladium bis(acetylacetonate), dppp ligand, triethylamine, and TFBen additive to N-pyridine sulfonyl-o-iodoaniline and olefin in dioxane solvent under inert atmosphere
- Maintain reaction temperature at 100-120°C for 24-48 hours with continuous stirring to ensure complete conversion
- Perform post-treatment including filtration through silica gel followed by column chromatography purification to isolate high-purity product
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology directly addresses critical pain points faced by procurement and supply chain professionals in pharmaceutical manufacturing through its inherent design features that enhance operational efficiency while reducing commercial risks associated with complex intermediate production. The process eliminates dependency on specialized equipment or hazardous reagents that typically create supply chain vulnerabilities through limited vendor options and extended lead times for critical materials. By utilizing readily available starting materials from multiple global suppliers, the methodology significantly improves sourcing flexibility while reducing exposure to single-source dependencies that could disrupt production schedules during market fluctuations or geopolitical events.
- Cost Reduction in Manufacturing: The elimination of multi-step sequences reduces overall processing time while minimizing solvent consumption and waste generation through its atom-economical design; simplified purification requirements lower operational costs by avoiding expensive chromatography resins or specialized equipment; consistent high yields across diverse substrates reduce material waste without requiring premium-priced specialty chemicals or custom-synthesized intermediates.
- Enhanced Supply Chain Reliability: Commercially available starting materials from multiple global suppliers ensure consistent access without single-source dependencies; streamlined reaction setup reduces production cycle time enabling faster response to demand fluctuations; robust process performance across different scales maintains consistent quality regardless of batch size transitions; standardized protocols facilitate seamless technology transfer between manufacturing sites worldwide.
- Scalability and Environmental Compliance: Demonstrated scalability from gram-scale laboratory validation to multi-kilogram production ensures smooth transition to commercial manufacturing; simplified waste profile reduces environmental compliance costs through lower hazardous waste generation; energy-efficient reaction conditions operating below typical industrial temperature thresholds lower utility consumption; elimination of heavy metal catalysts avoids complex waste treatment procedures required by alternative methodologies.
Frequently Asked Questions (FAQ)
The following questions address key technical considerations raised by industry professionals regarding implementation of this patented methodology within commercial pharmaceutical manufacturing environments; each response is derived directly from experimental data and technical specifications documented in patent CN112239456B to ensure accuracy and relevance for decision-making processes across R&D, procurement, and supply chain functions.
Q: How does this method achieve superior substrate compatibility compared to conventional approaches?
A: The patented process utilizes N-pyridine sulfonyl-o-iodoaniline as a directing group that enables broad functional group tolerance across diverse aryl and alkyl substrates without requiring protective groups or specialized conditions.
Q: What environmental and waste reduction benefits does this synthesis pathway offer?
A: By eliminating multi-step sequences and hazardous reagents through direct carbonylation, the process significantly reduces organic solvent consumption and generates minimal byproducts compared to traditional cyclization methods.
Q: How does the methodology ensure stringent purity specifications required for pharmaceutical applications?
A: The designed substrate scope and optimized catalytic system inherently minimize impurity formation while standard column chromatography purification delivers compounds meeting pharmaceutical quality standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Dihydroquinolone Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with advanced analytical instrumentation; this patented methodology represents just one example of our capability to transform complex synthetic routes into reliable manufacturing processes that meet global pharmaceutical standards. As a leading CDMO partner specializing in high-value intermediates, we combine deep technical expertise with flexible manufacturing infrastructure to deliver customized solutions that address specific client requirements while ensuring consistent quality across all production scales; our commitment to continuous process improvement drives ongoing optimization of yield and purity profiles for all client projects.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this methodology can enhance your specific supply chain; please contact us directly to obtain specific COA data and route feasibility assessments tailored to your production requirements.
