Advanced Palladium-Catalyzed Carbonylation for Scalable 1,2,4-Triazol-3-one Production
Advanced Palladium-Catalyzed Carbonylation for Scalable 1,2,4-Triazol-3-one Production
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for constructing nitrogen-containing heterocycles, which serve as privileged scaffolds in drug discovery. Patent CN112538054B introduces a groundbreaking preparation method for 1,2,4-triazole-3-ketone compounds, addressing long-standing challenges in synthetic efficiency and substrate versatility. This technology leverages a transition metal palladium-catalyzed carbonylation tandem cyclization reaction, utilizing readily available chlorinated hydrazones and sodium azide as starting materials. The significance of this innovation lies in its ability to bypass harsh traditional conditions while delivering high-purity intermediates essential for developing antifungal, antitumor, and anticonvulsant agents. As a reliable pharmaceutical intermediate supplier, understanding such patented advancements allows us to offer superior process solutions that align with modern green chemistry principles and cost-effective manufacturing goals.
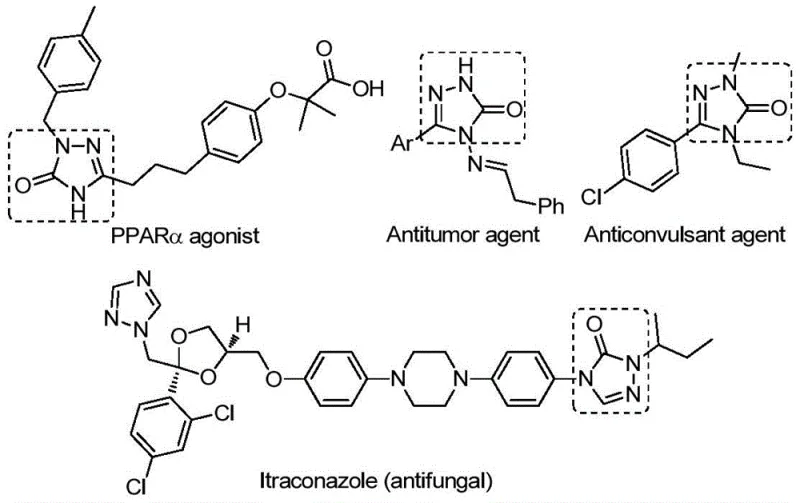
The structural diversity achievable through this method is exemplified by the wide range of biologically active molecules incorporating the 1,2,4-triazol-3-one core. As illustrated in the provided data, these scaffolds are integral to potent PPARα agonists, antitumor agents, and even established antifungal drugs like Itraconazole. The ability to efficiently synthesize these cores with varying substituents at the R1 and R2 positions opens new avenues for medicinal chemists to explore structure-activity relationships without being bottlenecked by complex synthesis routes. This patent specifically highlights the utility of the method in generating compounds that might otherwise be inaccessible or prohibitively expensive to produce using legacy techniques, thereby accelerating the timeline from benchtop discovery to clinical candidate selection.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2,4-triazol-3-one compounds has been plagued by significant operational hurdles that hinder large-scale production. Traditional pathways often rely on the cyclization of benzoyl hydrazides with urea under strong basic conditions, or the tandem reaction of hydrazides with isocyanates, which require stringent moisture control and hazardous reagents. Other methods involve the condensation of thioamides with hydrazines at elevated temperatures, leading to poor atom economy and difficult purification profiles due to the formation of sulfur-containing byproducts. Furthermore, many existing protocols necessitate the pre-activation of substrates, adding extra synthetic steps that cumulatively reduce overall yield and increase waste generation. These limitations result in narrow substrate scopes where sensitive functional groups are incompatible, forcing chemists to employ protecting group strategies that further complicate the manufacturing process and inflate costs.
The Novel Approach
In stark contrast, the methodology disclosed in CN112538054B offers a streamlined, one-pot solution that dramatically simplifies the synthetic landscape. By employing a palladium-catalyzed system with a solid carbon monoxide substitute (TFBen), the reaction proceeds under relatively mild thermal conditions, typically around 100°C, avoiding the extreme temperatures and pressures associated with gaseous CO usage. The use of chlorinated hydrazones as electrophiles coupled with sodium azide as a nitrogen source enables a direct construction of the triazole ring via a cascade sequence. This approach not only eliminates the need for pre-activated acyl isocyanates but also tolerates a broad spectrum of functional groups, including halogens and ethers, which are often sensitive in traditional protocols. The operational simplicity of mixing reagents in a common solvent like 1,4-dioxane makes this process highly amenable to scale-up, providing a distinct competitive advantage for cost reduction in pharmaceutical intermediate manufacturing.

Mechanistic Insights into Pd-Catalyzed Carbonylation Cyclization
The elegance of this transformation lies in its intricate catalytic cycle, which orchestrates multiple bond-forming events in a single operation. The reaction initiates with the oxidative addition of the low-valent palladium catalyst into the carbon-chlorine bond of the chlorohydrazone substrate, generating a reactive organopalladium intermediate. Subsequently, the carbon monoxide surrogate, TFBen, thermally decomposes to release CO in situ, which then inserts into the carbon-palladium bond to form an acyl-palladium species. This acyl intermediate then reacts with sodium azide to generate an acyl azide, which undergoes a Curtius rearrangement to yield a highly reactive isocyanate intermediate. Finally, an intramolecular nucleophilic attack by the pendant hydrazine nitrogen onto the isocyanate carbon closes the ring, furnishing the desired 1,2,4-triazol-3-one scaffold. This mechanistic pathway ensures high regioselectivity and minimizes the formation of oligomeric side products that often plague intermolecular cyclizations.
From an impurity control perspective, the choice of ligands and solvents plays a pivotal role in maintaining product purity. The patent specifies the use of Xantphos as a bidentate ligand, which stabilizes the palladium center and facilitates the reductive elimination step, thereby preventing catalyst decomposition and the formation of palladium black. Additionally, the selection of aprotic solvents like 1,4-dioxane is critical, as protic solvents can interfere with the azide reactivity or promote hydrolysis of the intermediate isocyanate. The method's robustness is further evidenced by its compatibility with diverse substrates, as shown in the specific examples where R1 and R2 groups vary from simple phenyl rings to bulky naphthyl and tert-butyl groups. This broad tolerance suggests that the steric and electronic properties of the substrates do not significantly impede the catalytic turnover, ensuring consistent quality across different batches of high-purity pharmaceutical intermediates.

How to Synthesize 1,2,4-Triazol-3-one Efficiently
To implement this cutting-edge synthesis in a laboratory or pilot plant setting, precise adherence to the optimized reaction parameters is essential for maximizing yield and minimizing impurities. The process begins with the careful weighing of the palladium catalyst system, specifically tridibenzylideneacetone dipalladium and the Xantphos ligand, alongside the solid CO source TFBen. These components are combined with the chlorohydrazone substrate and an excess of sodium azide in dry 1,4-dioxane to create a homogeneous reaction mixture. The detailed standardized synthesis steps see the guide below, which outlines the specific molar ratios and thermal profiles required to drive the reaction to completion while maintaining safety standards regarding azide handling.
- Charge a reaction vessel with Pd2(dba)3 catalyst, Xantphos ligand, TFBen carbon monoxide substitute, chlorohydrazone substrate, and sodium azide in 1,4-dioxane solvent.
- Heat the reaction mixture to 100°C and maintain stirring for 16 to 30 hours to ensure complete conversion of the starting materials.
- Upon completion, filter the mixture, adsorb the crude product onto silica gel, and purify via column chromatography to isolate the target 1,2,4-triazol-3-one.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates into tangible strategic benefits that extend beyond mere chemical yield. The primary advantage stems from the utilization of commodity chemicals as starting materials; chlorinated hydrazones and sodium azide are commercially available in bulk quantities at relatively low costs compared to specialized acyl isocyanates or activated hydrazides. This shift in raw material sourcing significantly reduces the bill of materials and mitigates supply chain risks associated with niche reagents that may have limited vendors or long lead times. Furthermore, the elimination of harsh reaction conditions and the use of a solid CO surrogate remove the need for specialized high-pressure reactors and gas handling infrastructure, allowing for production in standard glass-lined or stainless steel vessels commonly found in multipurpose chemical plants.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the simplification of the synthetic route, which reduces the number of unit operations and solvent exchanges required. By avoiding the use of expensive activating agents and minimizing the need for cryogenic conditions or high-pressure gas equipment, the capital expenditure and operational costs are drastically lowered. The high catalytic efficiency means that lower loadings of precious metal catalysts can be employed without sacrificing conversion, directly reducing the cost associated with palladium consumption and subsequent metal scavenging steps. Additionally, the high yields reported, reaching up to 96% in optimized examples, ensure that raw material waste is minimized, contributing to a more sustainable and cost-effective manufacturing profile.
- Enhanced Supply Chain Reliability: Dependence on complex, multi-step syntheses often introduces fragility into the supply chain, where a failure in any single step can halt the entire production schedule. This one-pot tandem reaction consolidates multiple transformations into a single vessel, thereby reducing the potential points of failure and shortening the overall production cycle time. The robustness of the reaction conditions allows for greater flexibility in scheduling and batch sizing, enabling manufacturers to respond more agilely to fluctuating market demands. Moreover, the stability of the reagents involved ensures that inventory can be held safely for extended periods without significant degradation, providing a buffer against supply disruptions and ensuring continuous availability of critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling chemical processes from gram to ton scale often reveals hidden challenges related to heat transfer and mixing, but this methodology is designed with scalability in mind. The use of 1,4-dioxane as a solvent, while requiring careful recovery, is a well-established industrial solvent with known recycling protocols, facilitating compliance with environmental regulations. The absence of gaseous carbon monoxide eliminates the risk of toxic gas leaks, enhancing workplace safety and reducing the regulatory burden associated with handling hazardous gases. The high atom economy of the tandem cyclization ensures that the majority of the reactant mass is incorporated into the final product, resulting in less chemical waste for treatment and disposal, which aligns with the increasing global emphasis on green chemistry and sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this palladium-catalyzed synthesis. These insights are derived directly from the experimental data and optimization studies presented in the patent documentation, providing clarity on reaction scope, safety, and scalability. Understanding these nuances is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the critical safety considerations when using sodium azide in this synthesis?
A: Sodium azide is toxic and potentially explosive; therefore, strict safety protocols including proper ventilation, personal protective equipment, and avoidance of heavy metal contact are mandatory during handling and waste disposal.
Q: Can this palladium-catalyzed method tolerate diverse functional groups on the substrate?
A: Yes, the method demonstrates excellent substrate compatibility, successfully tolerating various substituents such as halogens, alkyl groups, alkoxy groups, and heteroaryl moieties without significant loss in yield.
Q: Is the TFBen carbon monoxide substitute safer than using gaseous CO?
A: Yes, using solid CO surrogates like TFBen significantly enhances operational safety by eliminating the need for high-pressure gas cylinders and specialized gas handling infrastructure typically required for gaseous carbon monoxide.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,4-Triazol-3-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development and securing supply chains. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. By leveraging technologies such as the palladium-catalyzed carbonylation described in CN112538054B, we can offer customized synthesis solutions that optimize both cost and quality for our global partners.
We invite you to collaborate with us to explore how this innovative chemistry can enhance your project's success. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary molecules. Together, we can build a resilient and efficient supply chain that supports the next generation of life-saving therapies.
