Advanced Palladium-Catalyzed Synthesis of 1,2,4-Triazol-3-one Compounds for Commercial Scale-up
Advanced Palladium-Catalyzed Synthesis of 1,2,4-Triazol-3-one Compounds for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and cost-effective methodologies for constructing nitrogen-containing heterocycles, which serve as the backbone for countless bioactive molecules. A significant breakthrough in this domain is detailed in patent CN112538054B, which discloses a highly efficient preparation method for 1,2,4-triazol-3-one compounds. This specific class of five-membered nitrogen-containing heterocycles is ubiquitous in medicinal chemistry, exhibiting a profound spectrum of biological activities including antifungal, anti-inflammatory, antitumor, antiviral, and anticonvulsant properties. As illustrated in the structural diversity of bioactive molecules below, the ability to access these cores efficiently is paramount for drug discovery and development pipelines.
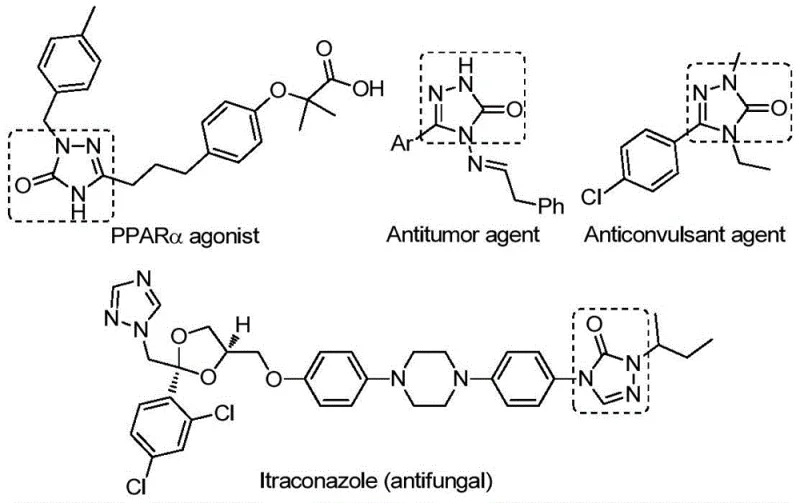
The innovation presented in this patent addresses critical bottlenecks in the existing synthetic landscape. By leveraging a transition metal palladium-catalyzed carbonylation tandem cyclization reaction, the method transforms cheap and easily accessible chlorohydrazones and sodium azide into valuable 1,2,4-triazol-3-one derivatives. For R&D directors and procurement managers alike, this represents a shift towards more sustainable and economically viable manufacturing processes. The protocol not only simplifies the operational complexity but also expands the substrate compatibility, allowing for the synthesis of compounds substituted with various functional groups, thereby widening the practical applicability of this methodology in the creation of reliable pharmaceutical intermediate supplier portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2,4-triazol-3-one compounds has been fraught with significant technical and economic challenges that hinder large-scale adoption. Conventional literature reports describe several traditional pathways, such as the cyclization of benzoyl hydrazide with urea under the influence of potassium hydroxide, or the tandem cyclization of hydrazides with isocyanates. Other methods involve the condensation of thioamides with hydrazines at elevated temperatures or the reaction of acyl isocyanates with monosubstituted hydrazines. These legacy processes are generally characterized by severe limitations, including harsh reaction conditions that demand specialized equipment and rigorous safety protocols. Furthermore, they often necessitate the pre-activation of reaction substrates, adding extra steps and increasing the overall cost of goods sold. Perhaps most critically for process chemists, these traditional routes frequently suffer from low yields and a narrow substrate scope, making them unsuitable for the diverse library synthesis required in modern drug discovery.
The Novel Approach
In stark contrast to these cumbersome legacy methods, the novel approach outlined in patent CN112538054B offers a streamlined and highly efficient alternative. This method employs a transition metal palladium-catalyzed carbonylation tandem cyclization strategy that fundamentally changes the input-output dynamic of the synthesis. By utilizing chlorohydrazones and sodium azide as starting materials, the process bypasses the need for unstable or hazardous isocyanates and high-pressure carbon monoxide gas. The reaction is conducted in an organic solvent at moderate temperatures, demonstrating exceptional tolerance for a wide range of functional groups. This versatility allows chemists to design and synthesize 1,2,4-triazol-3-one compounds with different substituents at various positions without compromising reaction efficiency. The operational simplicity, combined with the use of commercially available and inexpensive reagents, positions this technology as a superior choice for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Pd-Catalyzed Carbonylation Tandem Cyclization
To fully appreciate the technical sophistication of this synthesis, one must delve into the catalytic cycle that drives the transformation. The reaction initiates with the oxidative addition of the palladium catalyst into the carbon-chlorine bond of the chlorohydrazone substrate, generating a reactive divalent palladium intermediate. Simultaneously, the solid carbon monoxide substitute, TFBen (1,3,5-tricarboxylic acid phenol ester), undergoes thermal decomposition to release carbon monoxide in situ. This generated CO then inserts into the carbon-palladium bond, forming a crucial acyl-palladium intermediate. This step is pivotal as it avoids the logistical nightmares associated with handling gaseous CO in a plant setting. Subsequently, the acyl-palladium species reacts with sodium azide to generate an acyl azide compound, which spontaneously undergoes a Curtius rearrangement to yield an isocyanate intermediate. Finally, an intramolecular nucleophilic addition occurs, closing the ring to afford the final 1,2,4-triazol-3-one product. This elegant cascade ensures high atom economy and minimizes waste generation.

From an impurity control perspective, this mechanism offers distinct advantages over traditional high-temperature condensations. The mild reaction conditions (100°C to 120°C) and the specificity of the palladium catalyst significantly reduce the formation of by-products often seen in non-catalytic thermal reactions. The use of TFBen as a controlled CO source prevents the local concentration spikes of carbon monoxide that can lead to side reactions or safety incidents. Furthermore, the compatibility with various aprotic solvents like 1,4-dioxane allows for optimization of solubility and reaction kinetics, ensuring that the starting materials are fully dissolved and converted. This level of control is essential for producing high-purity 1,2,4-triazol-3-one compounds that meet the stringent quality specifications required for active pharmaceutical ingredients (APIs).
How to Synthesize 1,2,4-Triazol-3-one Efficiently
The practical implementation of this synthesis is designed for ease of execution in both laboratory and pilot plant settings. The protocol involves charging a reaction vessel with the palladium catalyst system, the ligand, the CO source, and the substrates in a suitable solvent. The mixture is then heated to facilitate the tandem cyclization. Following the reaction, a straightforward workup procedure involving filtration and purification yields the target molecule. This simplicity reduces the training burden on operators and minimizes the risk of human error during scale-up. For detailed procedural specifics regarding stoichiometry, temperature profiles, and purification techniques, please refer to the standardized synthesis guide below.
- Charge a reaction vessel with Pd2(dba)3 catalyst, Xantphos ligand, TFBen, chlorohydrazone substrate, and sodium azide in an aprotic organic solvent such as 1,4-dioxane.
- Heat the reaction mixture to a temperature range of 100°C to 120°C and maintain stirring for a duration of 16 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, perform silica gel treatment, and purify the crude product via column chromatography to isolate the high-purity 1,2,4-triazol-3-one compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic benefits that extend beyond mere chemical yield. The shift from complex, multi-step traditional syntheses to this direct catalytic route fundamentally alters the cost structure and supply reliability of these critical intermediates. By eliminating the need for pre-activated, expensive, or hazardous reagents, the process inherently lowers the raw material costs. Moreover, the operational simplicity reduces the consumption of utilities and the complexity of waste treatment, contributing to a greener and more sustainable manufacturing footprint. These factors collectively enhance the competitiveness of the final product in the global market.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the utilization of commodity chemicals such as sodium azide and chlorohydrazones, which are significantly cheaper than the specialized reagents required for conventional methods. Additionally, the replacement of gaseous carbon monoxide with the solid surrogate TFBen eliminates the need for high-pressure autoclaves and specialized gas handling infrastructure, resulting in substantial capital expenditure savings. The high reaction efficiency and broad substrate scope mean that fewer batches are rejected due to poor conversion, further optimizing the overall cost of production.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the ready availability of all key reagents on the open market. Unlike proprietary catalysts or unstable intermediates that may face sourcing bottlenecks, the components of this reaction system are standard industrial chemicals. This accessibility ensures consistent production schedules and reduces the risk of delays caused by raw material shortages. Furthermore, the robustness of the reaction conditions allows for flexible manufacturing planning, enabling suppliers to respond rapidly to fluctuating market demands without compromising product quality.
- Scalability and Environmental Compliance: The transition from bench scale to commercial production is facilitated by the homogeneous nature of the reaction and the absence of extreme physical conditions. The simplified post-treatment process, which typically involves filtration and standard chromatographic purification, is easily adaptable to large-scale continuous or batch processing. From an environmental standpoint, the method aligns with green chemistry principles by minimizing waste and avoiding toxic reagents, thereby simplifying regulatory compliance and reducing the environmental impact associated with chemical manufacturing.
Frequently Asked Questions (FAQ)
Understanding the nuances of this advanced synthesis is crucial for stakeholders evaluating its integration into their supply chains. The following questions address common inquiries regarding the technical feasibility, safety profile, and commercial potential of this method. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a factual basis for decision-making.
Q: What are the primary advantages of this Pd-catalyzed method over traditional synthesis routes?
A: Unlike traditional methods that require harsh conditions, pre-activated substrates, or toxic isocyanates, this novel approach utilizes cheap and readily available chlorohydrazones and sodium azide. It operates under milder conditions with a broad substrate scope and significantly higher reaction efficiency.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly demonstrates scalability. The use of stable solid reagents like TFBen instead of gaseous carbon monoxide simplifies equipment requirements, and the straightforward workup procedure involving filtration and chromatography facilitates easy scale-up from gram to multi-kilogram quantities.
Q: What is the role of TFBen in this reaction mechanism?
A: TFBen (1,3,5-tricarboxylic acid phenol ester) serves as a safe and effective carbon monoxide substitute. Under heating conditions, it releases CO in situ, which inserts into the carbon-palladium bond to form the necessary acyl-palladium intermediate, driving the cyclization forward without the safety hazards of handling CO gas.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,4-Triazol-3-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the technologies described in patent CN112538054B for the pharmaceutical industry. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of high-quality intermediates. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 1,2,4-triazol-3-one compound delivered meets the highest industry standards. We are committed to leveraging our technical expertise to bring innovative synthetic routes like this to market efficiently.
We invite you to collaborate with us to explore how this advanced palladium-catalyzed synthesis can optimize your specific project requirements. Our team is prepared to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how this method can improve your bottom line. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a competitive advantage in the global marketplace.
