Advanced Thiosilane Ligands for High-Efficiency Aryl Boronation in Pharmaceutical Manufacturing
The landscape of organic synthesis for pharmaceutical intermediates is undergoing a significant transformation driven by the need for more efficient and scalable catalytic systems. Patent CN111217847B introduces a groundbreaking class of thiosilane ligands, specifically designated as L1, L2, and L3, which offer a robust alternative to traditional catalytic ligands used in aryl boronation reactions. These novel ligands are engineered to overcome the longstanding limitations of bipyridine and borane-pyridine systems, providing a pathway to high-yield synthesis of arylboronic acid compounds that are critical building blocks in drug discovery. The structural versatility of these ligands allows for precise tuning of steric and electronic properties, ensuring optimal performance across a diverse range of substrates. By integrating sulfur and silicon functionalities into a unified ligand framework, this technology delivers exceptional stability and ease of handling, characteristics that are paramount for maintaining consistency in large-scale manufacturing environments.
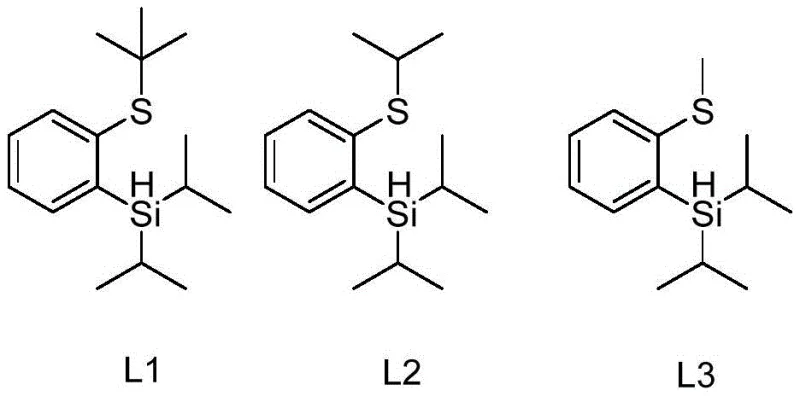
Furthermore, the industrial applicability of these thiosilane ligands is underscored by their straightforward preparation methods and high atom economy. Unlike previous generations of ligands that required complex multi-step syntheses, these compounds can be generated efficiently through a streamlined process involving bromine-lithium exchange and subsequent silanization. This simplicity not only reduces the overall cost of goods but also minimizes the generation of hazardous waste, aligning with modern green chemistry principles. For procurement managers and supply chain directors, this represents a tangible opportunity to secure a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials with reduced lead times. The ability to synthesize these ligands on a massive scale without compromising quality ensures a continuous supply chain for downstream API production, mitigating risks associated with raw material shortages or batch-to-batch variability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of arylboronic acid esters has relied heavily on catalytic systems utilizing bipyridine ligands in conjunction with iridium catalysts. While effective in certain contexts, these conventional methods suffer from significant drawbacks that hinder their efficiency in commercial settings. A primary challenge is the low yield observed particularly with electron-rich substrates, which limits the scope of molecules that can be effectively functionalized. Moreover, the polarity of bipyridine ligands is often remarkably similar to that of the desired boronated products, creating severe difficulties in separation and purification. This similarity necessitates extensive chromatographic processes or recrystallization steps, driving up operational costs and extending production cycles. Additionally, alternative systems based on borane-pyridine ligands are plagued by complex synthesis routes that are not conducive to large-scale industrial production, creating bottlenecks in the supply of critical intermediates.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes thiosilane ligands that fundamentally alter the reaction dynamics to favor high yield and easy isolation. The introduction of the thiosilane moiety creates a distinct chemical environment that enhances the solubility differences between the catalyst system and the final product, thereby simplifying purification protocols. This method allows for the efficient conversion of aryl compounds into their corresponding boronic acid esters with remarkable atom economy, ensuring that a maximum proportion of reactant atoms are incorporated into the final product. The versatility of this system is evident in its ability to accommodate various substituents on the aromatic ring, including halogens, alkyl groups, and ester functions, without significant loss in efficiency. By shifting away from the constraints of bipyridine chemistry, manufacturers can achieve cost reduction in pharmaceutical intermediate manufacturing through streamlined workflows and reduced solvent consumption.
Mechanistic Insights into Thiosilane-Iridium Catalyzed C-H Boronation
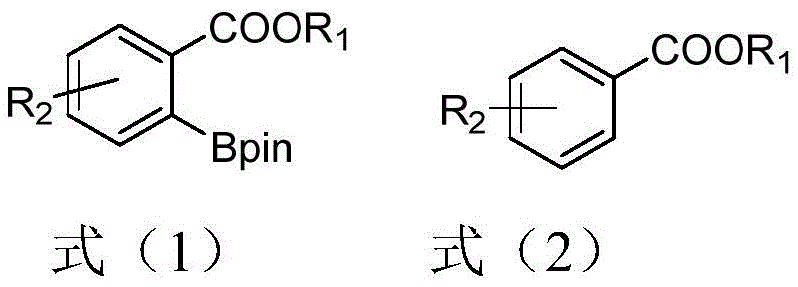
The efficacy of the thiosilane ligand system stems from its unique coordination behavior with the iridium catalyst, which facilitates a highly selective activation of carbon-hydrogen bonds. Mechanistically, the thiosilane ligand first coordinates with the iridium center, likely the (1,5-cyclooctadiene)(methoxy)iridium(I) dimer, to form a reactive intermediate species. This intermediate then engages with the guiding group ester functionality present on the substrate, effectively positioning the metal center in close proximity to the ortho-position carbon-hydrogen bond. This proximity effect is crucial for overcoming the high activation energy typically associated with C-H bond cleavage, allowing the reaction to proceed under relatively mild conditions ranging from 30°C to 120°C. Once the C-H bond is activated, the pinacol ester diboron reagent is introduced to complete the boronation process, resulting in the formation of the stable arylboronic acid ester. This directed ortho-boronation strategy ensures high regioselectivity, minimizing the formation of unwanted isomers and simplifying the impurity profile of the crude reaction mixture.
From an impurity control perspective, the specific structural features of the thiosilane ligand play a pivotal role in suppressing side reactions. The steric bulk provided by the isopropyl and tert-butyl groups on the silicon and sulfur atoms, respectively, creates a protective shield around the active catalytic site. This steric hindrance prevents non-specific interactions with other parts of the substrate molecule, thereby reducing the likelihood of over-boronation or decomposition of sensitive functional groups. Furthermore, the stability of the ligand-catalyst complex ensures that the active species remains intact throughout the reaction duration, which can extend up to 48 hours depending on the substrate reactivity. This sustained activity translates to consistent product quality and predictable reaction outcomes, which are essential metrics for R&D directors evaluating the feasibility of scaling a new synthetic route. The ability to maintain high purity specifications throughout the synthesis underscores the robustness of this catalytic platform for producing high-purity OLED materials or pharmaceutical intermediates.
How to Synthesize Thiosilane Ligands Efficiently
The preparation of these advanced ligands is designed for operational simplicity, requiring standard laboratory equipment and commonly available reagents. The synthesis begins with the generation of a lithiated intermediate via halogen-metal exchange, followed by the introduction of the silyl group to finalize the ligand structure. This two-step sequence is highly reproducible and can be adapted for both small-scale optimization and large-scale production runs. The detailed standardized synthesis steps provided below outline the precise conditions necessary to achieve optimal yields and purity, serving as a comprehensive guide for process chemists looking to implement this technology. By adhering to these protocols, manufacturers can ensure the consistent production of high-quality ligands that drive the efficiency of the subsequent boronation reactions.
- Perform a bromine-lithium exchange reaction on a 2-bromobenzene thioether compound using n-butyl lithium in tetrahydrofuran at temperatures between -30°C and -80°C under a protective atmosphere.
- Add diisopropylchlorosilane to the resulting lithium-exchanged thioether compound to initiate the silanization reaction.
- Purify the final reaction mixture via column chromatography to isolate the thiosilane ligand as a light yellow oily liquid with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this thiosilane ligand technology offers profound strategic benefits that extend beyond mere technical performance. The simplified synthesis of the ligands themselves means that the upstream supply chain is less vulnerable to disruptions, as the raw materials such as 2-bromobenzene thioethers and diisopropylchlorosilane are commodity chemicals with stable market availability. This reliability ensures that production schedules can be maintained without the risk of delays caused by specialized reagent shortages. Moreover, the elimination of complex purification steps associated with older ligand systems translates directly into substantial cost savings by reducing solvent usage, energy consumption, and labor hours. These efficiencies contribute to a leaner manufacturing process that is both economically advantageous and environmentally sustainable, aligning with corporate sustainability goals.
- Cost Reduction in Manufacturing: The implementation of this catalytic system drives significant cost optimization by removing the need for expensive and difficult-to-separate bipyridine ligands. Since the thiosilane ligands are easily distinguishable from the product during purification, the reliance on resource-intensive chromatographic separations is drastically minimized. This reduction in downstream processing requirements lowers the overall cost of goods sold, allowing for more competitive pricing in the global market. Additionally, the high atom economy of the reaction ensures that raw materials are utilized efficiently, further enhancing the economic viability of the process for commercial scale-up of complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The stability of the thiosilane ligands at room temperature is a critical factor for supply chain resilience. Unlike moisture-sensitive or thermally unstable catalysts that require cold chain logistics and specialized storage facilities, these ligands can be stored under standard conditions without degradation. This ease of storage simplifies inventory management and reduces the risk of material spoilage during transit or warehousing. Consequently, suppliers can maintain higher stock levels with confidence, ensuring that they can meet sudden spikes in demand from clients without compromising on quality or delivery timelines.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, having been validated for potential industrial mass synthesis. The use of standard solvents like tetrahydrofuran and 2-methyltetrahydrofuran, combined with the absence of heavy metal contaminants in the final ligand product, simplifies waste treatment and regulatory compliance. The high yields and selectivity reduce the volume of chemical waste generated per unit of product, supporting greener manufacturing practices. This alignment with environmental regulations reduces the burden of waste disposal costs and facilitates smoother regulatory approvals for new drug applications relying on these intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this thiosilane ligand technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on performance metrics and operational parameters. Understanding these details is essential for stakeholders evaluating the integration of this method into existing production workflows or new product development pipelines.
Q: What are the stability advantages of the new thiosilane ligands compared to bipyridine ligands?
A: Unlike bipyridine ligands which often suffer from polarity issues close to the product making separation difficult, the novel thiosilane ligands described in CN111217847B are stable at room temperature and convenient to store. Their distinct structural properties facilitate easier purification processes, significantly reducing downstream processing time and costs in industrial settings.
Q: Can this catalytic system handle electron-rich or sterically hindered substrates?
A: Yes, the thiosilane ligand system demonstrates superior performance specifically for substrates that traditionally exhibit low reactivity. The unique coordination mechanism allows for effective activation of ortho-position carbon-hydrogen bonds even in the presence of strong electron-withdrawing groups, ensuring high yields where conventional bipyridine systems often fail.
Q: Is the synthesis of these ligands scalable for commercial production?
A: Absolutely. The preparation method involves only 1 to 2 reaction steps using readily available raw materials like 2-bromobenzene thioether compounds and diisopropylchlorosilane. The process boasts high atom economy and avoids complex synthetic routes, making it highly suitable for large-scale industrial synthesis ranging from kilogram to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thiosilane Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the thiosilane ligand technology described in CN111217847B for the next generation of pharmaceutical synthesis. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of ligand or intermediate we produce adheres to the highest international standards. We are committed to leveraging our technical expertise to optimize these catalytic processes for your specific needs, delivering solutions that enhance both efficiency and profitability.
We invite you to engage with our technical procurement team to discuss how this innovative ligand system can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into the potential economic benefits of switching to this superior catalytic platform. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability as a trusted partner in your supply chain. Together, we can accelerate the development of life-saving medicines by ensuring a reliable and cost-effective supply of critical chemical intermediates.
