Revolutionizing Aryl Boronate Production with Stable Thiosilane Ligands for Industrial Scale-Up
Revolutionizing Aryl Boronate Production with Stable Thiosilane Ligands for Industrial Scale-Up
The landscape of fine chemical synthesis is constantly evolving, driven by the relentless demand for more efficient, cost-effective, and scalable methodologies for producing critical building blocks. In the realm of pharmaceutical and agrochemical development, aryl boronic acid esters stand out as indispensable intermediates, serving as key precursors for Suzuki-Miyaura cross-coupling reactions which construct the carbon-carbon bonds found in countless active pharmaceutical ingredients. A groundbreaking advancement in this field is detailed in patent CN111217847A, which introduces a novel class of thiosilane ligands designed to overcome the persistent limitations of traditional catalytic systems. This patent discloses a robust method for preparing these ligands and demonstrates their exceptional utility in iridium-catalyzed aryl boronization reactions, offering a pathway to high-yield, high-purity products that are essential for modern drug discovery pipelines.
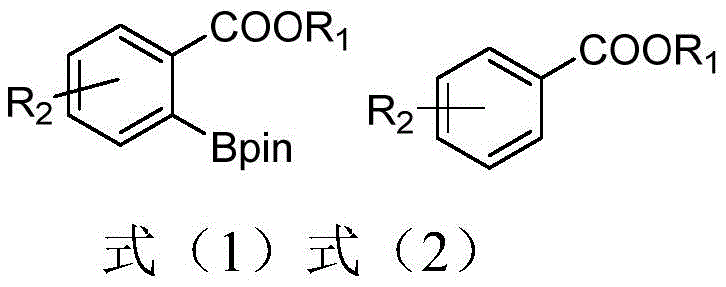
The significance of this technology lies not merely in the chemical transformation itself, but in the strategic advantages it offers to reliable pharmaceutical intermediate suppliers seeking to optimize their manufacturing processes. By leveraging the unique coordination chemistry of sulfur and silicon within the ligand framework, the disclosed method achieves superior atom economy and operational simplicity. For procurement managers and supply chain directors, this represents a tangible opportunity for cost reduction in API manufacturing, as the process eliminates the need for exotic, difficult-to-source ligands while maintaining rigorous quality standards. The ability to synthesize these ligands efficiently and apply them across a broad substrate scope positions this technology as a cornerstone for the commercial scale-up of complex pharmaceutical intermediates, ensuring a steady flow of high-quality materials for global markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aryl boronic acid esters via carbon-hydrogen bond borylation has relied heavily on catalytic systems employing bipyridine ligands or borane pyridine complexes in conjunction with iridium catalysts. While these methods have served the industry for some time, they are plagued by significant inefficiencies that hinder large-scale industrial adoption. Specifically, bipyridine ligands often exhibit disappointingly low yields, particularly when reacting with electron-rich substrates which are common in medicinal chemistry scaffolds. Furthermore, the polarity of bipyridine ligands is frequently too similar to that of the desired boronate products, creating a nightmare scenario for purification teams who must employ resource-intensive chromatographic techniques to separate the catalyst residue from the final product. On the other hand, borane pyridine systems, while effective in some contexts, suffer from synthetic complexity; their preparation is multi-step and cumbersome, making them unsuitable for the rapid, cost-sensitive environment of commercial chemical production.
The Novel Approach
In stark contrast to these legacy systems, the thiosilane ligands described in the patent represent a paradigm shift in ligand design, offering a solution that is both chemically elegant and industrially pragmatic. These ligands, characterized by structures L1, L2, and L3, can be synthesized efficiently in just one or two reaction steps from readily available starting materials, drastically simplifying the supply chain for the catalyst itself. 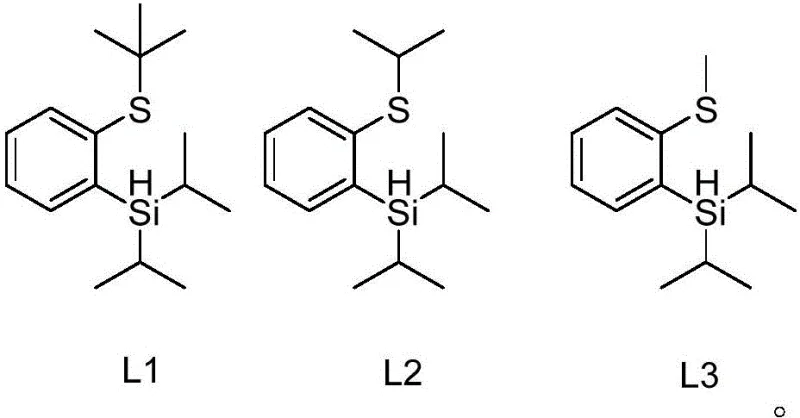
The structural integrity of these thiosilane ligands allows them to exist stably at room temperature, facilitating easy storage and handling without the need for specialized inert atmosphere conditions typically required for sensitive organometallic reagents. When applied to the borylation reaction, these ligands demonstrate remarkable versatility, delivering high yields even for substrates that are traditionally difficult to functionalize, such as those bearing strong electron-withdrawing groups. The distinct chemical nature of the thiosilane framework also ensures that the ligand possesses a polarity profile significantly different from the product, thereby streamlining the purification process and enabling the isolation of high-purity OLED material or pharmaceutical precursors with minimal effort. This combination of synthetic accessibility, stability, and performance makes the novel approach a superior choice for modern chemical manufacturing.
Mechanistic Insights into Thiosilane-Iridium Catalyzed C-H Borylation
The exceptional performance of the thiosilane ligands stems from their unique ability to modulate the electronic and steric environment of the iridium center during the catalytic cycle. Mechanistically, the process begins with the coordination of the thiosilane ligand to the iridium catalyst, forming a highly active intermediate species that is primed for substrate activation. Unlike simple monodentate ligands, the thiosilane framework likely engages in a cooperative interaction where the sulfur atom and potentially the silicon center work in tandem to stabilize the metal complex. This stabilized intermediate then coordinates with the directing group, specifically the ester moiety present on the aryl substrate, which serves to orient the molecule precisely for ortho-position carbon-hydrogen bond activation. This directed metallation is the critical selectivity-determining step, ensuring that the boron atom is installed exclusively at the desired position on the aromatic ring, thereby minimizing the formation of regio-isomeric impurities that complicate downstream processing.
Following the activation of the C-H bond, the introduction of the pinacol ester diboron reagent allows for the transmetallation step, where the boron group is transferred to the iridium center and subsequently to the aryl ring, completing the borylation cycle. The high atom economy of this reaction is a direct result of this efficient mechanism, as nearly all atoms in the reagents are incorporated into the final product or benign byproducts. From an impurity control perspective, the robustness of the thiosilane-iridium complex prevents the premature decomposition of the catalyst, which is a common source of metallic contamination in finished products. The specific steric bulk provided by the isopropyl and tert-butyl groups on the silicon and sulfur atoms, respectively, further shields the active site from non-productive side reactions. This precise control over the reaction environment ensures that the resulting aryl boronic acid esters meet the stringent purity specifications required for regulatory submission, reducing the burden on quality control laboratories and accelerating time-to-market for new drug candidates.
How to Synthesize Thiosilane Ligands Efficiently
The preparation of these high-performance ligands is designed to be operationally simple, utilizing standard laboratory equipment and commercially available reagents to ensure reproducibility and scalability. The synthesis generally proceeds through a bromine-lithium exchange reaction followed by a silanization step, both of which are well-understood transformations in organic synthesis. The process begins with the treatment of a 2-bromobenzene thioether precursor with n-butyl lithium at low temperatures to generate a reactive lithiated intermediate, which is then trapped with diisopropylchlorosilane to install the crucial silicon-hydrogen motif. Detailed standardized synthetic protocols, including specific molar ratios, temperature profiles, and workup procedures, are essential for achieving the optimal yield and purity described in the patent documentation. For a comprehensive breakdown of the exact experimental conditions and safety precautions required for each step, please refer to the structured guide below.
- Perform bromine-lithium exchange on 2-bromobenzene thioether compounds with n-butyl lithium at -30 to -80°C, followed by silanization with diisopropylchlorosilane to obtain the ligand.
- Mix the thiosilane ligand with (1,5-cyclooctadiene)(methoxy)iridium(I) dimer and pinacol diboron in 2-methyltetrahydrofuran under protective atmosphere.
- Add the aryl substrate and react at 30-120°C for 10-48 hours, followed by filtration and column chromatography purification to isolate the arylboronic acid ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this thiosilane ligand technology translates directly into tangible operational benefits that impact the bottom line. The primary advantage lies in the drastic simplification of the raw material supply chain; since the ligands are derived from common chemicals like 2-bromothioanisole and simple silanes, there is no reliance on single-source suppliers for exotic proprietary ligands. This diversification of supply sources significantly mitigates the risk of production stoppages due to raw material shortages, ensuring a continuous flow of intermediates for manufacturing lines. Furthermore, the elimination of complex ligand synthesis steps reduces the overall consumption of solvents and energy, aligning with modern green chemistry initiatives and reducing the environmental footprint of the production facility. The stability of the ligands at ambient conditions also removes the need for expensive cold-chain logistics during transportation, further driving down logistical costs and simplifying inventory management.
- Cost Reduction in Manufacturing: The economic impact of switching to this catalytic system is profound, primarily driven by the elimination of expensive and difficult-to-remove bipyridine ligands. Because the thiosilane ligands facilitate easier separation via standard silica gel chromatography due to their distinct polarity, the volume of solvent and silica required for purification is significantly reduced compared to traditional methods. Additionally, the higher yields achieved, especially on challenging electron-rich substrates, mean that less starting material is wasted, directly improving the mass balance and reducing the cost per kilogram of the final active ingredient. The ability to use lower catalyst loadings while maintaining high turnover numbers further contributes to substantial cost savings, making the process economically viable for large-scale commercial production without compromising on quality.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robust nature of the thiosilane ligands, which do not degrade rapidly upon exposure to air or moisture compared to many sensitive organometallic catalysts. This stability allows for longer shelf lives and reduces the frequency of waste disposal associated with expired reagents. Moreover, the synthesis of the ligands themselves is short and high-yielding, meaning that backup stock can be generated quickly in-house if external supply lines are disrupted. This self-sufficiency capability is a critical strategic asset for any organization aiming to secure its supply of high-purity pharmaceutical intermediates against global market volatility, ensuring that production schedules remain uninterrupted regardless of external pressures.
- Scalability and Environmental Compliance: Scaling this process from gram-level laboratory experiments to multi-ton industrial production is facilitated by the use of standard reaction conditions and non-hazardous solvents like 2-methyltetrahydrofuran. The reaction operates effectively at moderate temperatures and does not require extreme pressures, reducing the engineering complexity and capital expenditure required for reactor design. From an environmental compliance standpoint, the high atom economy and reduced solvent usage for purification result in a significantly lower E-factor (mass of waste per mass of product), simplifying waste treatment protocols and helping facilities meet increasingly strict environmental regulations. This scalability ensures that the technology is not just a laboratory curiosity but a viable solution for the commercial scale-up of complex polymer additives and fine chemicals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this thiosilane ligand technology in industrial settings. These answers are derived directly from the experimental data and technical specifications outlined in the patent, providing a reliable basis for decision-making. Understanding these nuances is crucial for R&D teams evaluating the feasibility of adopting this new catalytic system for their specific pipeline projects. For more detailed technical support or custom route assessments, our team is available to provide deeper insights tailored to your specific chemical challenges.
Q: What are the primary advantages of thiosilane ligands over traditional bipyridine ligands in borylation reactions?
A: Thiosilane ligands offer significantly higher yields, particularly for electron-rich substrates where bipyridine ligands often fail. Furthermore, their distinct polarity facilitates easier separation from products compared to bipyridine, reducing downstream purification costs and complexity.
Q: How does the stability of these ligands impact supply chain reliability?
A: Unlike complex borane pyridine systems that require intricate synthesis, thiosilane ligands are stable at room temperature and can be synthesized efficiently in 1-2 steps. This stability ensures consistent quality during storage and transport, minimizing supply chain disruptions for large-scale manufacturing.
Q: Is this catalytic system suitable for substrates with strong electron-withdrawing groups?
A: Yes, the unique structure of the thiosilane ligand enhances reactivity towards substrates with low reactivity and strong electron-withdrawing substituents, achieving high conversion rates where conventional catalysts might struggle, thus expanding the scope of accessible chemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thiosilane Ligand Supplier
As the demand for efficient and sustainable chemical synthesis methods grows, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM becomes a strategic imperative for staying competitive. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and risk-free. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of thiosilane ligand or aryl boronate intermediate we produce meets the highest international standards. We understand the critical nature of supply continuity in the pharmaceutical sector and have built our operations to prioritize reliability, quality, and speed.
We invite you to engage with our technical procurement team to discuss how this innovative technology can be integrated into your current manufacturing processes. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits specific to your product portfolio. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will drive efficiency and profitability in your organization. Let us be your partner in navigating the complexities of modern chemical synthesis and securing your supply chain for the future.
