Revolutionizing Aryl Boronation: High-Efficiency Thiosilane Ligands for Industrial Pharmaceutical Synthesis
Introduction to Next-Generation Catalytic Ligands
The landscape of fine chemical synthesis is constantly evolving, driven by the urgent need for more efficient, sustainable, and cost-effective methodologies. Patent CN111217847B represents a significant breakthrough in this domain, introducing a novel class of thiosilane ligands designed specifically for iridium-catalyzed aryl boronation reactions. This technology addresses critical bottlenecks in the production of arylboronic acid esters, which are indispensable building blocks for modern pharmaceuticals and agrochemicals. By replacing traditional ligand systems with these innovative thiosilane structures, manufacturers can achieve superior reaction yields, simplified purification processes, and enhanced atom economy. The strategic implementation of these ligands allows for the direct functionalization of carbon-hydrogen bonds, bypassing the need for pre-functionalized starting materials and thereby reducing waste generation. For industry leaders seeking to optimize their supply chains and reduce the environmental footprint of their manufacturing processes, this patent offers a compelling pathway forward. The stability and ease of synthesis of these ligands further underscore their potential for widespread adoption in large-scale industrial applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of arylboronic acid esters has relied heavily on catalytic systems utilizing bipyridine ligands or borane pyridine ligands in conjunction with iridium catalysts. While effective in certain contexts, these conventional methods present substantial drawbacks that hinder industrial scalability and economic efficiency. A primary concern is the relatively low yield observed, particularly when processing electron-rich substrates, which limits the overall throughput of the synthesis. Furthermore, the separation and purification of the final product are often complicated by the fact that the polarity of the bipyridine ligand is remarkably similar to that of the desired boronic acid ester product. This similarity necessitates complex and resource-intensive purification steps, such as repeated chromatography or recrystallization, which drive up operational costs and extend production timelines. Additionally, the synthesis of the borane pyridine ligand system itself is chemically complex, involving multiple steps that increase the risk of impurities and reduce the overall atom economy of the process. These cumulative inefficiencies make traditional methods less attractive for the high-volume manufacturing required by the global pharmaceutical market.
The Novel Approach
In stark contrast to these legacy systems, the thiosilane ligands described in patent CN111217847B offer a streamlined and highly efficient alternative. These ligands, characterized by structures L1, L2, and L3, are designed to coordinate effectively with iridium catalysts while maintaining distinct physical properties that facilitate downstream processing. 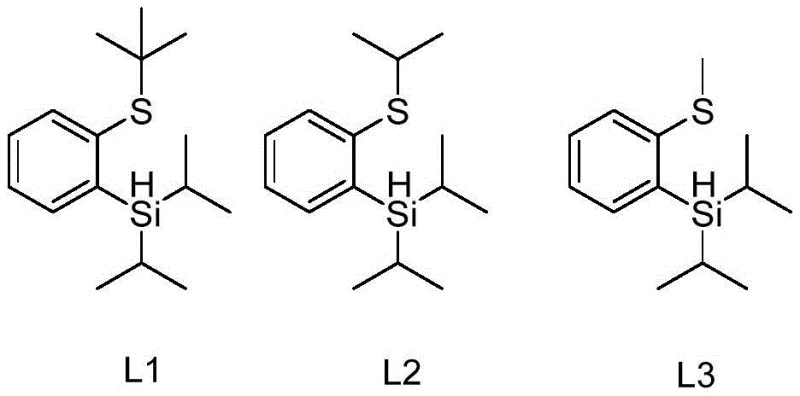 The novel approach leverages a unique structural motif where a sulfur atom and a silicon hydride group are positioned ortho to each other on a benzene ring, creating a robust chelating environment for the metal center. This configuration not only enhances the catalytic activity but also ensures that the ligand remains stable at room temperature, simplifying storage and handling logistics. The synthesis of these ligands is remarkably straightforward, typically requiring only one or two reaction steps from commercially available precursors like 2-bromothioanisole. This simplicity translates directly into lower raw material costs and reduced manufacturing complexity. Moreover, the distinct polarity of the thiosilane ligands compared to the boronic acid products allows for facile separation, often achievable through standard column chromatography or even distillation, significantly reducing the burden on purification teams and accelerating the time-to-market for critical intermediates.
The novel approach leverages a unique structural motif where a sulfur atom and a silicon hydride group are positioned ortho to each other on a benzene ring, creating a robust chelating environment for the metal center. This configuration not only enhances the catalytic activity but also ensures that the ligand remains stable at room temperature, simplifying storage and handling logistics. The synthesis of these ligands is remarkably straightforward, typically requiring only one or two reaction steps from commercially available precursors like 2-bromothioanisole. This simplicity translates directly into lower raw material costs and reduced manufacturing complexity. Moreover, the distinct polarity of the thiosilane ligands compared to the boronic acid products allows for facile separation, often achievable through standard column chromatography or even distillation, significantly reducing the burden on purification teams and accelerating the time-to-market for critical intermediates.
Mechanistic Insights into Iridium-Catalyzed C-H Borylation
The efficacy of the thiosilane ligand system stems from its sophisticated interaction with the iridium catalyst and the substrate. Mechanistically, the process begins with the coordination of the thiosilane ligand to the iridium center, forming a highly active catalytic intermediate. This intermediate is then capable of engaging with the substrate, specifically targeting the ortho-position carbon-hydrogen bond adjacent to the directing group, such as an ester moiety. The presence of the sulfur and silicon atoms in the ligand framework modulates the electronic density at the metal center, enhancing its ability to activate inert C-H bonds under mild conditions. Once the C-H bond is activated, the pinacol diboron reagent is introduced, facilitating the transfer of the boron group to the aromatic ring. This sequence results in the formation of the arylboronic acid ester with high regioselectivity and minimal byproduct formation. The high atom economy of this reaction is a direct result of this precise mechanistic pathway, which avoids the generation of stoichiometric salt waste common in traditional cross-coupling reactions.
Furthermore, the robustness of this catalytic system is evident in its tolerance towards a wide array of functional groups. 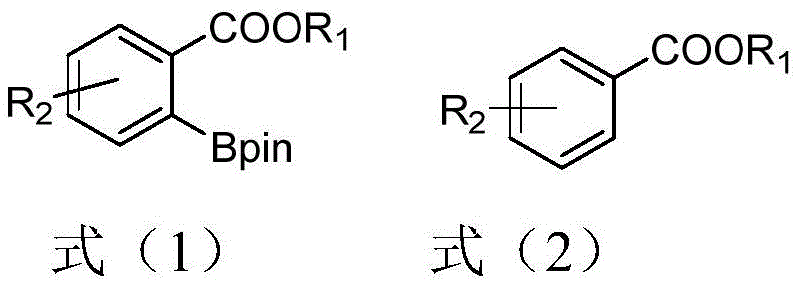 As illustrated in the general reaction scheme, the system successfully processes substrates bearing halogens, alkyl chains, methoxy groups, and even strong electron-withdrawing groups like trifluoromethyl and cyano functionalities. This broad substrate scope is particularly valuable for R&D directors who require flexible synthetic routes for diverse drug candidates. The mechanism also inherently suppresses the formation of common impurities associated with over-borylation or homocoupling, leading to a cleaner crude reaction profile. This purity advantage reduces the load on downstream purification units and ensures that the final API intermediates meet stringent quality specifications. The ability to maintain high yields even with sterically hindered or electronically deactivated substrates underscores the versatility of the thiosilane ligand architecture, making it a superior choice for complex molecule synthesis.
As illustrated in the general reaction scheme, the system successfully processes substrates bearing halogens, alkyl chains, methoxy groups, and even strong electron-withdrawing groups like trifluoromethyl and cyano functionalities. This broad substrate scope is particularly valuable for R&D directors who require flexible synthetic routes for diverse drug candidates. The mechanism also inherently suppresses the formation of common impurities associated with over-borylation or homocoupling, leading to a cleaner crude reaction profile. This purity advantage reduces the load on downstream purification units and ensures that the final API intermediates meet stringent quality specifications. The ability to maintain high yields even with sterically hindered or electronically deactivated substrates underscores the versatility of the thiosilane ligand architecture, making it a superior choice for complex molecule synthesis.
How to Synthesize Thiosilane Ligands Efficiently
The preparation of these high-performance ligands is designed for operational simplicity and scalability, making it accessible for both laboratory research and pilot plant operations. The process generally involves a bromine-lithium exchange reaction followed by a silanization step, both of which utilize standard reagents and equipment found in most fine chemical facilities. The detailed standardized synthesis steps provided below outline the precise conditions required to maximize yield and purity, ensuring consistent batch-to-batch performance. By adhering to these protocols, manufacturers can reliably produce the necessary quantities of ligand to support continuous catalytic campaigns. The use of common solvents like tetrahydrofuran and readily available silanes further enhances the economic viability of this route.
- Perform bromine-lithium exchange on 2-bromobenzene thioether compounds using n-butyllithium in tetrahydrofuran at -30 to -80°C under inert atmosphere.
- Add diisopropylchlorosilane to the lithiated intermediate and allow silanization reaction to proceed at 0-25°C for 8-12 hours.
- Quench the reaction, extract the organic phase, and purify the crude thiosilane ligand via silica gel column chromatography to obtain the final light yellow oily liquid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this thiosilane ligand technology presents a multitude of strategic benefits that extend beyond mere technical performance. The primary advantage lies in the significant reduction of manufacturing costs associated with the production of arylboronic acid intermediates. By eliminating the need for expensive and difficult-to-separate bipyridine ligands, companies can streamline their purification workflows, thereby reducing solvent consumption, energy usage, and labor hours. The simplified synthesis of the ligands themselves, often achievable in just one or two steps from low-cost commodity chemicals, further drives down the cost of goods sold (COGS). This cost efficiency is compounded by the high atom economy of the catalytic reaction, which minimizes raw material waste and maximizes the output per unit of input. Consequently, organizations can achieve substantial cost savings while maintaining or even improving the quality of their final products.
- Cost Reduction in Manufacturing: The transition to thiosilane ligands eliminates the reliance on complex ligand systems that require multi-step syntheses and rigorous purification. Since the precursors for L1, L2, and L3 are commercially available or easily synthesized from bulk chemicals like 2-bromothioanisole, the raw material expenditure is drastically lowered. Furthermore, the ease of separating the ligand from the product reduces the volume of silica gel and solvents required for chromatography, leading to direct savings in consumable costs. The high yields reported in the patent examples, often exceeding 80% for the ligand synthesis and showing robust performance in borylation, ensure that material throughput is optimized, reducing the need for reprocessing batches.
- Enhanced Supply Chain Reliability: Supply chain continuity is critical for pharmaceutical manufacturing, and the stability of these thiosilane ligands offers a distinct advantage. Unlike some sensitive catalytic additives that require cold chain storage or inert atmosphere handling, these ligands are stable at room temperature and can be stored for extended periods without degradation. This stability simplifies inventory management and reduces the risk of supply disruptions due to spoilage. Additionally, the synthetic route relies on widely available reagents such as n-butyllithium and diisopropylchlorosilane, minimizing the risk of bottlenecks associated with specialty chemical shortages. This reliability ensures that production schedules can be maintained consistently, supporting just-in-time manufacturing models.
- Scalability and Environmental Compliance: The patent explicitly highlights the suitability of this method for large-scale industrial synthesis, addressing a key concern for supply chain heads planning for commercial expansion. The reaction conditions, operating between 30°C and 120°C in solvents like 2-methyltetrahydrofuran, are amenable to standard reactor setups without requiring exotic high-pressure or cryogenic equipment. From an environmental perspective, the high atom economy and reduced waste generation align with green chemistry principles, helping companies meet increasingly stringent regulatory requirements regarding solvent discharge and hazardous waste disposal. The ability to scale from milligram to kilogram levels without loss of efficiency provides a clear pathway for rapid commercialization of new drug candidates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this thiosilane ligand technology. These insights are derived directly from the experimental data and beneficial effects outlined in patent CN111217847B, providing a factual basis for decision-making. Understanding these nuances is essential for R&D teams evaluating the feasibility of integrating this chemistry into their existing pipelines.
Q: What are the advantages of thiosilane ligands over traditional bipyridine ligands in C-H borylation?
A: Unlike bipyridine ligands which often suffer from low yields with electron-rich substrates and difficult separation due to similar polarity to products, thiosilane ligands offer higher atom economy, easier purification, and stable storage at room temperature.
Q: Can this catalytic system be scaled for industrial production?
A: Yes, the patent explicitly states the method is suitable for large-scale industrial synthesis, demonstrating successful preparation from milligram to potential kilogram scales with robust yields and simplified workup procedures.
Q: What types of substrates are compatible with this thiosilane ligand system?
A: The system exhibits broad substrate scope, effectively processing aryl compounds with various substituents including halogens, alkyl groups, methoxy groups, and strong electron-withdrawing groups like esters and trifluoromethyl groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thiosilane Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the thiosilane ligand system described in CN111217847B. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate these innovative laboratory methods into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from discovery to market is seamless and efficient. We are committed to delivering high-purity pharmaceutical intermediates that meet the most rigorous quality standards, supported by our state-of-the-art rigorous QC labs and comprehensive analytical capabilities. By leveraging our deep understanding of organometallic catalysis and process optimization, we can help you unlock the full value of this technology.
We invite you to collaborate with us to explore how these thiosilane ligands can enhance your specific synthesis projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your current manufacturing challenges, demonstrating exactly where efficiencies can be gained. We encourage you to reach out to request specific COA data for our catalog of boronic acid esters or to discuss route feasibility assessments for your proprietary molecules. Together, we can drive innovation and efficiency in the global supply of critical fine chemical intermediates.
