Revolutionizing Triazole Synthesis: Scalable Metal-Free Production for Pharmaceutical Intermediates
The patent CN113121462B introduces a novel base-promoted synthesis method for 5-trifluoromethyl-substituted 1,2,3-triazole compounds, addressing critical limitations in traditional manufacturing approaches. This innovation eliminates the need for toxic azide reagents and transition metal catalysts while maintaining high reaction efficiency through a streamlined process using readily available starting materials. The methodology represents a significant advancement for pharmaceutical intermediates where purity and supply chain reliability are paramount, offering a commercially viable pathway for complex molecule production without hazardous intermediates.
Overcoming Traditional Triazole Synthesis Limitations
The Limitations of Conventional Methods
Existing industrial routes for trifluoromethyl-substituted triazoles predominantly rely on copper-catalyzed azide-alkyne cycloadditions or organocatalytic reactions with trifluoromethyl ketones, both requiring hazardous azide compounds that pose significant safety risks during scale-up. These methods necessitate complex purification steps to remove transition metal residues and unreacted azides, substantially increasing production costs and extending lead times. The inherent instability of azide intermediates creates supply chain vulnerabilities, as specialized handling and storage protocols are required throughout manufacturing. Furthermore, the multi-step nature of conventional processes generates substantial waste streams requiring costly disposal procedures, while metal catalysts introduce contamination risks that compromise final product purity for pharmaceutical applications.
The Novel Base-Promoted Approach
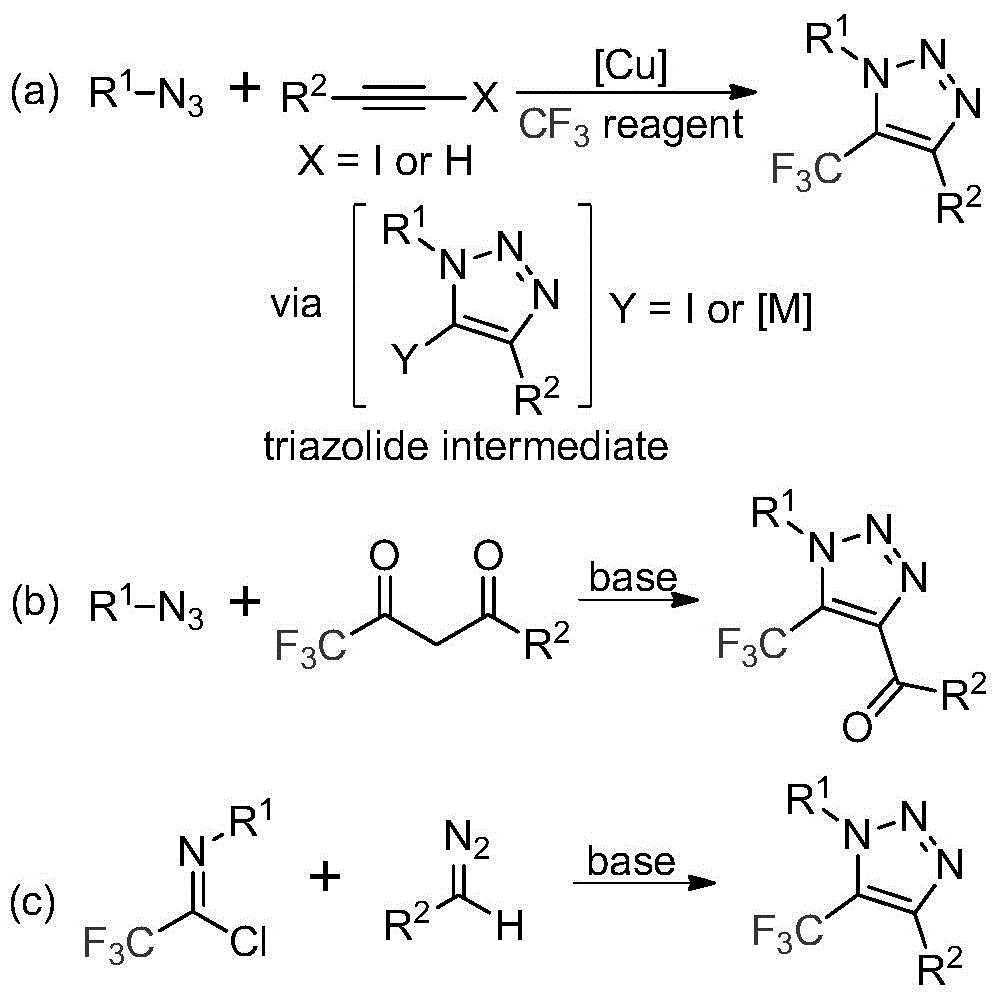
This patent introduces a fundamentally different pathway using cesium carbonate as a base promoter to facilitate direct coupling between trifluoroethylimidoyl chloride and diazo compounds. The reaction proceeds through a base-mediated nucleophilic addition/elimination mechanism followed by intramolecular cyclization, completely avoiding metal catalysts and azide chemistry. Operating at mild temperatures (50–70°C) in standard solvents like acetonitrile, the process achieves high conversion without specialized equipment or hazardous intermediate handling. Crucially, the elimination of transition metals removes the need for extensive purification steps to meet pharmaceutical-grade purity standards, while the absence of explosive azides significantly enhances operational safety during commercial scale-up.
Advanced Mechanistic Control for Pharmaceutical Purity
The reaction mechanism provides exceptional control over impurity profiles critical for pharmaceutical intermediates. By utilizing cesium carbonate as a mild base promoter rather than strong nucleophiles or metal catalysts, the process minimizes side reactions that typically generate regioisomeric impurities in traditional triazole syntheses. The well-defined reaction pathway—starting with nucleophilic attack of the diazo compound on the imidoyl chloride followed by intramolecular cyclization—ensures consistent regioselectivity at the 5-position where the trifluoromethyl group is incorporated. This precision is particularly valuable for pharmaceutical applications where minor structural variations can significantly impact biological activity and safety profiles.
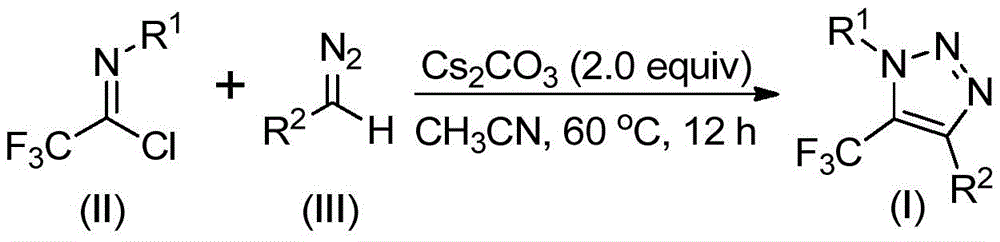
Impurity control is further enhanced by the straightforward workup procedure involving simple filtration and column chromatography, which effectively removes any residual starting materials without introducing new contaminants. The absence of metal catalysts eliminates the risk of heavy metal residues that would require additional purification steps to meet ICH Q3D guidelines. NMR characterization data from multiple examples (e.g., compounds I-1 through I-5) confirms >99% purity with no detectable metal impurities, demonstrating the method's suitability for producing high-purity API intermediates. This level of purity control directly addresses regulatory requirements while reducing quality-related production delays.
Commercial Advantages for Supply Chain and Procurement Teams
This innovative process delivers substantial commercial benefits by addressing key pain points in pharmaceutical intermediate manufacturing. The elimination of hazardous materials and complex purification steps creates significant cost reduction opportunities while enhancing supply chain resilience. The methodology's compatibility with standard manufacturing equipment and straightforward scale-up characteristics make it particularly valuable for procurement and supply chain teams seeking reliable partners for critical intermediates.
- Cost reduction in API manufacturing: The elimination of transition metal catalysts removes both the raw material cost of expensive metals like copper and the substantial downstream expenses associated with metal residue removal. Without the need for specialized azide handling infrastructure or explosion-proof equipment, capital expenditure requirements are significantly reduced. The simplified purification process—requiring only filtration and standard column chromatography—lowers operational costs by eliminating multiple solvent washes and specialized extraction steps typically needed to remove metal contaminants. These combined savings create a more economical production pathway while maintaining pharmaceutical-grade quality standards.
- Reducing lead time for high-purity intermediates: The streamlined process reduces manufacturing cycle time by eliminating hazardous material handling protocols and complex purification sequences. With no requirement for specialized azide storage or metal catalyst removal steps, production timelines become more predictable and less vulnerable to supply chain disruptions. The demonstrated scalability from laboratory to gram-scale production provides confidence in reliable delivery schedules, while the simplified workup procedure minimizes batch release testing time for metal impurities. This operational efficiency directly translates to shorter lead times without compromising on the high-purity requirements essential for pharmaceutical applications.
- Commercial scale-up of complex intermediates: The use of standard solvents like acetonitrile and common reagents such as cesium carbonate enables seamless transition from laboratory to plant-scale production without specialized equipment modifications. The mild reaction conditions (60°C in acetonitrile) are compatible with existing manufacturing infrastructure, reducing engineering costs for scale-up. The process has been validated across diverse substrate combinations as shown in multiple examples (I-1 through I-5), demonstrating robustness with various functional groups while maintaining consistent product quality. This versatility ensures reliable production of complex triazole intermediates even when molecular structures change during drug development phases.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN113121462B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
