Scalable Synthesis of Cyclopropyl Methyl Cyanide Derivatives for Global Pharmaceutical Supply Chains
The pharmaceutical and agrochemical industries constantly demand high-performance building blocks that balance structural complexity with economic viability. Patent CN102249949A introduces a transformative methodology for the preparation of cyclopropyl methyl cyanide derivatives, addressing critical bottlenecks in existing synthetic routes. This technology leverages gamma-butyrolactone as a foundational feedstock, enabling a robust, multi-step cascade that includes halogenation, cyclization, acylation, amidation, and dehydration. For R&D directors and supply chain leaders, this represents a pivotal shift from laboratory-scale curiosities to commercially viable processes capable of supporting global demand for active pharmaceutical ingredients (APIs). The innovation lies not just in the chemical transformations but in the holistic optimization of energy consumption, safety profiles, and overall yield, making it an ideal candidate for reliable pharmaceutical intermediate supplier partnerships seeking to secure long-term material availability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art in the synthesis of cyclopropyl carboxylic acid derivatives, such as the methods described in J. Org. Chem. 54, 6100 (1989) and J.M. Synthesis 1983, 915, suffered from severe scalability and safety constraints that rendered them unsuitable for modern industrial application. The earlier protocols often relied on expensive and hazardous reagents, such as mercuric oxide, which posed significant environmental and disposal challenges while driving up production costs. Furthermore, reaction conditions were frequently inefficient, with some intermediate steps requiring reaction times as long as 40 hours, leading to excessive energy consumption and low throughput. The use of unstable bases like potassium tert-butoxide for hydrolysis steps introduced storage hazards and operational complexities, limiting the maximum synthetic scale to merely 200 grams in many reported cases. These legacy methods simply could not meet the rigorous demands of cost reduction in pharmaceutical intermediate manufacturing required by today's high-volume markets.
The Novel Approach
The methodology disclosed in CN102249949A fundamentally reengineers the synthetic pathway to overcome these historical inefficiencies by utilizing gamma-butyrolactone, a cheap, low-toxicity, and readily available starting material. This novel approach streamlines the formation of the cyclopropane ring through optimized halogenation and cyclization steps that drastically reduce reaction times and eliminate the need for toxic heavy metal catalysts. By replacing hazardous reagents with safer alternatives like potassium carbonate and optimizing phase transfer catalysis, the process achieves high conversion rates under milder conditions. The strategic substitution of traditional acidic dehydrating agents with liquid phosgene in the final step prevents the degradation of the sensitive cyclopropane ring, ensuring high product integrity. This comprehensive process redesign facilitates the commercial scale-up of complex pharmaceutical intermediates, offering a safe, efficient, and consumption-reducing pathway that aligns perfectly with green chemistry principles.
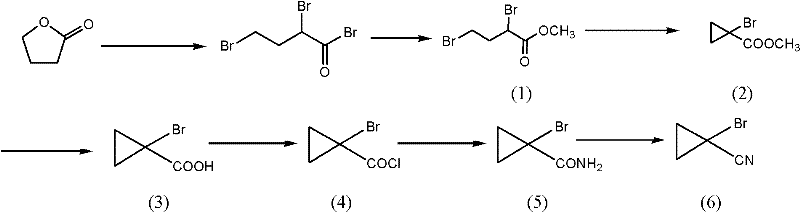
Mechanistic Insights into Halogenation and Cyclization Cascade
The core of this synthetic strategy involves a sophisticated sequence beginning with the halogenation of gamma-butyrolactone to form methyl dihalobutyrate derivatives, followed by an intramolecular nucleophilic substitution to close the cyclopropane ring. In the initial step, the lactone ring is opened and functionalized using phosphorus halides and elemental halogens under controlled temperatures, typically between 40°C and 80°C, to ensure selective formation of the dihalo-ester. The subsequent cyclization is driven by a base-mediated intramolecular displacement, where phase transfer catalysts like tetrabutylammonium hydrogensulfate play a critical role in shuttling reactive species between phases, thereby accelerating the ring closure. This mechanistic precision allows for the formation of the strained three-membered ring with high stereochemical control and minimal byproduct formation, which is essential for maintaining the purity specifications required for downstream API synthesis.
Furthermore, the conversion of the resulting cyclopropyl carboxylic acid to the final nitrile involves a carefully managed dehydration of the primary amide intermediate. Traditional acidic dehydrating agents often promote ring-opening side reactions due to the high strain energy of the cyclopropane system, leading to yields typically ranging from 60% to 70%. In contrast, the use of liquid phosgene as a dehydrating agent operates through a distinct mechanism that activates the amide without generating strongly acidic conditions that would compromise the ring structure. This specific choice of reagent directs the reaction pathway exclusively towards the desired nitrile product, effectively suppressing competing decomposition pathways. The result is a substantial improvement in step yield, often exceeding 90%, which significantly enhances the overall mass balance and economic feasibility of the entire manufacturing campaign.
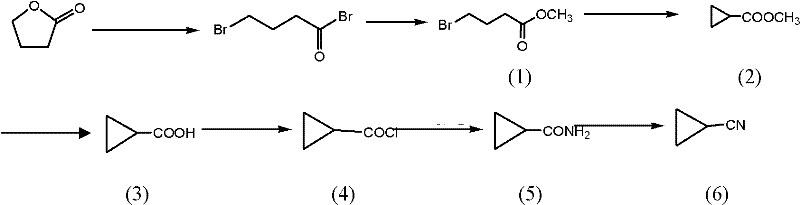
How to Synthesize Cyclopropyl Methyl Cyanide Efficiently
The synthesis of these valuable nitrile derivatives requires precise control over reaction parameters to maximize yield and safety while minimizing waste generation. The process begins with the halogenation of gamma-butyrolactone, followed by cyclization, hydrolysis, acylation, amidation, and finally dehydration, with each step optimized for industrial throughput. Detailed standard operating procedures regarding specific stoichiometry, temperature ramps, and workup protocols are critical for reproducibility and quality assurance. For a comprehensive guide on executing this synthesis with optimal results, please refer to the standardized protocol outlined below which details the exact operational parameters for each transformation stage.
- Halogenation and Esterification: React gamma-butyrolactone with phosphorus halides and halogens to form methyl dihalobutyrate intermediates.
- Cyclization and Hydrolysis: Perform intramolecular cyclization using phase transfer catalysts followed by alkaline hydrolysis to yield cyclopropyl carboxylic acids.
- Amidation and Dehydration: Convert the acid to an amide via acyl chloride, then dehydrate using liquid phosgene to obtain the final nitrile product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process offers tangible strategic benefits that extend beyond simple chemical efficiency. By shifting the raw material base to gamma-butyrolactone, manufacturers can decouple their supply chains from volatile markets associated with exotic or toxic precursors, ensuring a more stable and predictable sourcing landscape. The elimination of heavy metal catalysts like mercury not only reduces the environmental footprint but also simplifies waste treatment protocols, leading to significant cost savings in effluent management and regulatory compliance. Additionally, the reduction in reaction times and the ability to operate at higher concentrations directly translate to increased reactor turnover rates, allowing facilities to produce more material with existing infrastructure. These factors collectively contribute to a more resilient supply chain capable of meeting tight delivery schedules without compromising on quality or safety standards.
- Cost Reduction in Manufacturing: The replacement of expensive and hazardous reagents with commodity chemicals like gamma-butyrolactone and potassium carbonate drives down the direct material cost per kilogram of product. Furthermore, the improved yields in critical steps, particularly the dehydration phase, mean that less raw material is wasted, effectively lowering the cost of goods sold. The simplified workup procedures, which avoid complex purification steps needed to remove heavy metal residues, further reduce operational expenditures related to labor and consumables. This holistic approach to cost optimization ensures that the final intermediate is priced competitively in the global market.
- Enhanced Supply Chain Reliability: Utilizing widely available starting materials mitigates the risk of supply disruptions that often plague specialized chemical supply chains. The robustness of the process, characterized by its tolerance to varied reaction conditions and its scalability from pilot to full production, ensures consistent output quality. This reliability is crucial for pharmaceutical customers who require uninterrupted supply to maintain their own production schedules for finished drugs. By securing a manufacturing route that is less dependent on niche reagents, suppliers can offer longer-term contracts with greater confidence in their ability to deliver.
- Scalability and Environmental Compliance: The process is explicitly designed for large-scale industrial production, having been validated to overcome the limitations of previous gram-scale methods. The absence of toxic heavy metals and the use of contained reagents like liquid phosgene with appropriate scrubbing systems align with stringent international environmental regulations. This compliance reduces the administrative burden on supply chain teams regarding hazardous material handling and disposal permits. Consequently, the manufacturing facility can operate with greater flexibility and lower regulatory risk, facilitating smoother audits and faster time-to-market for new products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of cyclopropyl methyl cyanide derivatives based on the patented technology. Understanding these details helps stakeholders evaluate the feasibility of integrating this intermediate into their specific development pipelines. The answers provided are derived directly from the technical specifications and experimental data disclosed in the patent documentation to ensure accuracy and relevance.
Q: What are the primary advantages of using gamma-butyrolactone as a starting material?
A: Gamma-butyrolactone is significantly cheaper and less toxic than traditional precursors like those requiring mercuric oxide. It allows for large-scale industrial production with reduced environmental impact and lower raw material costs.
Q: How does the new dehydration method improve product purity?
A: By utilizing liquid phosgene instead of strong acidic dehydrating agents, the process avoids acid-catalyzed side reactions that typically degrade the strained cyclopropane ring, resulting in yields exceeding 90% and higher purity.
Q: Is this process suitable for commercial scale-up?
A: Yes, the patent explicitly addresses scalability issues found in previous literature, optimizing reaction times and temperatures to support production scales ranging from hundreds of kilograms to multi-ton annual capacities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclopropyl Methyl Cyanide Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of drug development programs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the bench to the plant. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of cyclopropyl methyl cyanide derivative meets the exacting standards required by global regulatory bodies. Our commitment to technical excellence means we can adapt the patented process to your specific needs, optimizing for cost, speed, or sustainability as your project evolves.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can benefit your specific application. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this gamma-butyrolactone-based pathway. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your volume requirements. Let us collaborate to build a supply chain that is not only efficient and cost-effective but also robust enough to support your long-term growth in the competitive pharmaceutical landscape.
