Scaling Cyclopropyl Methyl Cyanide Derivatives: Advanced Synthesis for Global Pharma Supply Chains
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable pathways for high-value intermediates, and the technology disclosed in patent CN102249949A represents a significant leap forward in the synthesis of cyclopropyl methyl cyanide derivatives. This specific intellectual property outlines a comprehensive methodology that transforms gamma-butyrolactone, a readily available and cost-effective bulk chemical, into complex cyclopropane-containing nitriles through a series of optimized transformations. Unlike previous laboratory-scale methods that struggled with toxicity and poor scalability, this invention focuses heavily on industrial feasibility, addressing critical pain points such as energy consumption, reagent stability, and overall process safety. For R&D directors and procurement specialists alike, understanding this route is essential because it offers a viable path to producing high-purity pharmaceutical intermediates without relying on hazardous heavy metals or prohibitively expensive reagents. The strategic shift from traditional, small-batch academic protocols to this streamlined industrial process underscores a commitment to efficiency and environmental compliance that modern supply chains demand.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of cyclopropyl carboxylic acid derivatives, which are crucial precursors to the target nitriles, was fraught with significant operational and economic challenges that hindered large-scale adoption. Historical literature, such as J.Org.Chem 54, 6100 (1989), described methods using gamma-butyrolactone but were severely limited by a maximum synthetic scale of merely 200 grams, rendering them useless for commercial manufacturing. Furthermore, these legacy processes often required reaction times extending up to 40 hours for intermediate formation, leading to excessive energy consumption and low throughput in a production environment. Another critical bottleneck was the reliance on potassium tert-butoxide for hydrolysis steps; this reagent is not only expensive but also notoriously difficult to store and handle safely due to its sensitivity to moisture and air. Additionally, alternative routes cited in J.M.Synthesis 1983 utilized mercuric oxide, a highly toxic heavy metal that poses severe environmental disposal issues and regulatory hurdles for any modern chemical facility aiming for green certification.
The Novel Approach
The methodology presented in CN102249949A fundamentally reengineers the synthesis pathway to overcome these historical inefficiencies by introducing a sequence that is both chemically elegant and industrially robust. By optimizing the cyclization step through precise temperature control and the use of phase transfer catalysts, the reaction time is drastically reduced while maintaining high conversion rates, directly translating to improved production capacity. The replacement of unstable and costly bases with common industrial alkalis like potassium hydroxide or sodium carbonate eliminates the supply chain risks associated with specialized reagents, ensuring consistent availability and lower raw material costs. Moreover, the process incorporates a novel dehydration strategy in the final steps that avoids the harsh acidic conditions typical of older methods, thereby preserving the integrity of the sensitive cyclopropane ring. This holistic optimization ensures that the transition from gram-scale discovery to ton-scale production is seamless, providing a reliable cyclopropyl methyl cyanide derivative supplier with a distinct competitive advantage in the market.
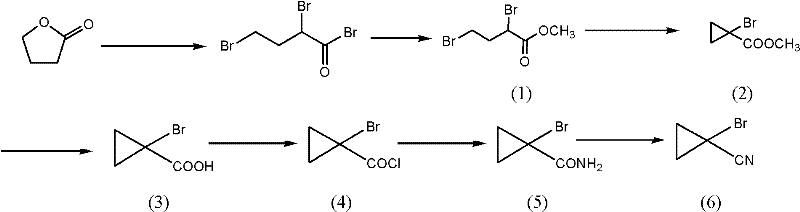
Mechanistic Insights into Cyclization and Dehydration Catalysis
The core chemical innovation lies in the meticulous control of the cyclization and dehydration mechanisms, which are the two most critical junctures determining the overall yield and impurity profile of the final product. During the cyclization of the dihalobutyrate intermediate, the use of a phase transfer catalyst facilitates the interaction between the organic substrate and the inorganic base in a biphasic system, promoting the intramolecular nucleophilic substitution that forms the strained cyclopropane ring. This mechanistic pathway is carefully tuned to operate at elevated temperatures between 70°C and 100°C, which accelerates the kinetics without triggering unwanted decomposition, a balance that is crucial for maintaining high space-time yields in a reactor. Following the formation of the carboxylic acid derivative, the conversion to the nitrile involves a two-stage activation where the acid is first transformed into an acyl chloride and then amidated. The subsequent dehydration of the amide is where the true brilliance of this patent shines; by employing liquid phosgene as the dehydrating agent, the reaction proceeds under milder conditions compared to traditional acidic dehydrators like phosphorus pentoxide or thionyl chloride alone. This specific choice of reagent prevents the acid-catalyzed side reactions that typically degrade the cyclopropane ring or cause polymerization, ensuring that the carbon-nitrogen triple bond is formed with exceptional selectivity and minimal byproduct generation.
Impurity control is inherently built into this mechanistic design, particularly through the management of the hydrolysis and amidation steps which are prone to generating carboxylic acid contaminants. In conventional amidation using aqueous ammonia, a portion of the reactive acyl chloride is inevitably hydrolyzed back to the carboxylic acid, creating a difficult-to-remove impurity that lowers the overall mass balance. The patented process mitigates this by utilizing pre-cooled acyl chlorides and controlling the addition rate into the ammoniating reagent at sub-zero temperatures, effectively suppressing the hydrolysis pathway while favoring amide formation. Furthermore, the final dehydration step using liquid phosgene is conducted in anhydrous solvents like acetonitrile, which prevents the introduction of water that could reverse the reaction or hydrate the nitrile product. This rigorous exclusion of moisture and control over reaction thermodynamics results in a crude product profile that is significantly cleaner, reducing the burden on downstream purification units like distillation columns and crystallizers. For quality assurance teams, this means the final high-purity OLED material or pharmaceutical intermediate meets stringent specifications with less reprocessing, directly impacting the cost of goods sold.
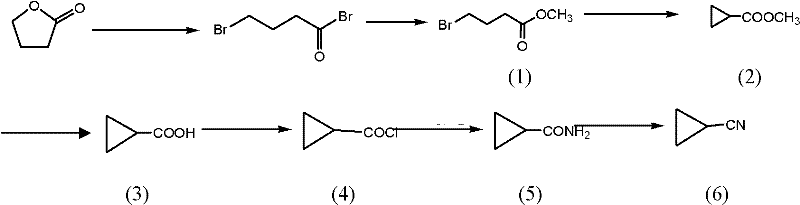
How to Synthesize Cyclopropyl Methyl Cyanide Efficiently
Executing this synthesis requires a disciplined approach to process parameters, specifically regarding temperature gradients and reagent stoichiometry, to ensure the reproducibility demonstrated in the patent examples. The procedure begins with the halogenation of gamma-butyrolactone, where strict temperature control during the exothermic addition of halogens is vital to prevent runaway reactions and ensure the correct regioselectivity of the dihalo-intermediate. Following esterification, the cyclization step demands efficient mixing to maintain the suspension of the inorganic base, ensuring that the phase transfer catalyst can effectively shuttle ions across the interface to drive the ring closure. The subsequent hydrolysis and activation steps must be monitored closely via GC or HPLC to determine the exact endpoint, preventing over-reaction that could lead to ring opening or decarboxylation. Finally, the dehydration step requires specialized handling equipment for liquid phosgene due to its toxicity, necessitating closed systems and efficient scrubbing of tail gases to meet environmental safety standards. For a detailed breakdown of the specific operational parameters, reagent ratios, and workup procedures, please refer to the standardized guide below.
- Halogenation and Esterification: React gamma-butyrolactone with phosphorus halides and halogen gas, followed by methanolysis to form methyl dihalobutyrate.
- Cyclization and Hydrolysis: Perform intramolecular cyclization using phase transfer catalysts, followed by alkaline hydrolysis to yield cyclopropanecarboxylic acid derivatives.
- Amidation and Dehydration: Convert the acid to an acyl chloride, react with ammonia to form the amide, and dehydrate using liquid phosgene to obtain the final nitrile.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers substantial strategic benefits that directly address the primary concerns of procurement managers and supply chain heads regarding cost stability and operational reliability. The shift away from exotic, unstable reagents like potassium tert-butoxide and toxic heavy metals like mercuric oxide simplifies the raw material sourcing strategy, allowing purchasers to leverage commodity chemicals that are available from multiple global vendors. This diversification of the supply base reduces the risk of single-source bottlenecks and price volatility, ensuring a more predictable cost structure for long-term contracts. Additionally, the reduction in reaction times and the elimination of energy-intensive reflux periods significantly lower the utility consumption per kilogram of product, contributing to a leaner manufacturing cost profile. By minimizing side reactions through superior mechanistic control, the process also reduces the volume of waste streams generated, which translates to lower disposal costs and a smaller environmental footprint, aligning with increasingly strict global sustainability mandates.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the substitution of high-cost specialty reagents with inexpensive, bulk-grade chemicals that do not compromise reaction efficiency. Eliminating the need for mercuric oxide removes the substantial costs associated with hazardous waste treatment and regulatory compliance, while the use of common bases like potassium hydroxide ensures that raw material expenses remain low and stable. Furthermore, the improved yields in the critical dehydration step mean that less starting material is wasted, maximizing the output from each batch and effectively lowering the unit cost of the final active ingredient. These cumulative efficiencies create a robust margin structure that allows for competitive pricing in the global market without sacrificing quality.
- Enhanced Supply Chain Reliability: Operational continuity is significantly strengthened by the use of reagents that are shelf-stable and easy to transport, removing the logistical complexities associated with handling moisture-sensitive or pyrophoric materials. The process is designed to tolerate standard industrial equipment, meaning that production does not require specialized, hard-to-source reactors, thereby reducing the lead time for setting up new production lines or scaling existing ones. This flexibility ensures that suppliers can respond rapidly to fluctuations in demand, providing a dependable source of high-purity pharmaceutical intermediates even during periods of market stress. The robustness of the chemistry also means fewer batch failures and less downtime for cleaning or maintenance, guaranteeing a steady flow of material to downstream customers.
- Scalability and Environmental Compliance: The transition from laboratory to plant scale is facilitated by the process inherent safety features, such as controlled exotherms and the avoidance of highly toxic heavy metals, which simplifies the permitting and validation process for new facilities. The ability to run reactions at moderate temperatures and pressures reduces the engineering constraints on reactor design, allowing for larger batch sizes that improve overall equipment effectiveness. Moreover, the reduced generation of hazardous byproducts aligns with green chemistry principles, making it easier for manufacturing sites to maintain their environmental certifications and avoid potential fines or shutdowns. This forward-thinking approach to process design ensures long-term viability in a regulatory landscape that is becoming progressively more stringent regarding chemical safety and emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this synthesis technology, based on the detailed disclosures within the patent documentation. Understanding these nuances is critical for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios or sourcing strategies. The answers provided reflect the specific improvements in yield, safety, and scalability that distinguish this method from prior art, offering clarity on how it resolves historical production challenges. Stakeholders are encouraged to review these points to gain a deeper appreciation of the value proposition offered by this advanced chemical process.
Q: What are the key advantages of using gamma-butyrolactone as a starting material?
A: Gamma-butyrolactone is significantly cheaper and less toxic than alternative precursors like those requiring mercuric oxide. It allows for a safer, more environmentally compliant process suitable for large-scale manufacturing.
Q: How does the new dehydration method improve product purity?
A: By utilizing liquid phosgene instead of traditional strong acidic dehydrating agents, the process minimizes side reactions such as polymerization or ring-opening, thereby significantly enhancing the final yield and purity of the nitrile derivative.
Q: Is this process suitable for commercial scale-up?
A: Yes, the patent explicitly addresses industrial scalability by optimizing reaction times, reducing solvent usage, and replacing unstable reagents like potassium tert-butoxide with robust industrial bases like potassium hydroxide.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclopropyl Methyl Cyanide Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex molecules like cyclopropyl methyl cyanide derivatives requires more than just a patent; it demands deep process engineering expertise and a commitment to quality that spans from gram-scale development to full commercial production. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this innovative synthesis are fully realized in a practical, industrial setting. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to verify that every batch meets the exacting standards required by the global pharmaceutical industry. Our infrastructure is designed to handle the specific safety requirements of reagents like liquid phosgene, providing a secure and compliant environment for the manufacture of these high-value intermediates.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can be tailored to your specific project needs and volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into how adopting this methodology can reduce your overall cost of goods while improving supply security. We are prepared to provide specific COA data and route feasibility assessments to demonstrate our capability to deliver consistent, high-quality material that supports your drug development timelines. Let us collaborate to bring this efficient, scalable technology to your supply chain, ensuring a reliable source of critical intermediates for your future success.
