Optimizing Abiraterone Acetate Production via Novel Chlorination Technology for Global Supply Chains
The pharmaceutical industry continuously seeks robust synthetic pathways for oncology drugs, particularly for prostate cancer treatments like Abiraterone Acetate. Patent CN112125942A introduces a transformative synthetic method that addresses long-standing inefficiencies in producing this critical API intermediate. By shifting from traditional halogenation methods to a novel, highly selective chlorination protocol, this technology promises to redefine the manufacturing landscape for abiraterone precursors. The core innovation lies in the precise control of reaction parameters during the chlorination of a specific steroidal hydrazone, utilizing reagents such as dichlorohydantoin or N-chlorosuccinimide. This approach not only enhances the chemical purity of the intermediate but also streamlines the downstream processing, offering a compelling value proposition for global supply chains seeking reliability and cost-efficiency in high-volume API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of abiraterone acetate has relied heavily on three primary routes: the triflate method, the iodination method, and the Grignard method, each presenting significant bottlenecks for industrial scale-up. The triflate route, while offering controllable processes and high purity, is economically prohibitive due to the exorbitant cost of triflic anhydride and the starting material, rendering it unsuitable for cost-sensitive mass production. Alternatively, the widely used iodination method, which employs dehydroepiandrosterone, hydrazine hydrate, and iodine, suffers from inherently low yields in the key halogenation step. As illustrated in the traditional pathway below, the iodination process is plagued by long reaction times and arduous purification challenges, often leading to inconsistent batch quality and increased waste generation.
Furthermore, attempts to substitute iodine with more active brominating agents like N-bromosuccinimide have historically failed to provide a viable solution. While bromination increases reaction activity, it concurrently escalates the probability of uncontrollable side reactions, making it nearly impossible to achieve the stringent purity profiles required for pharmaceutical intermediates. The Grignard approach adds another layer of complexity, involving difficult-to-control elimination reactions and low overall yields. These conventional limitations create a volatile supply environment, where raw material price fluctuations and process inefficiencies directly impact the availability and cost of the final life-saving medication.
The Novel Approach
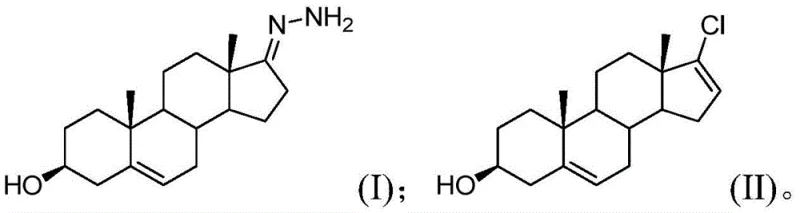
In stark contrast to these legacy methods, the technology disclosed in CN112125942A pioneers a chlorination strategy that effectively balances reactivity with selectivity. The process initiates with a compound of Formula (I), which undergoes a chlorination reaction in the presence of a specific chlorinating reagent and a base. The breakthrough lies in the meticulous optimization of the mass ratio between the substrate and the base, maintained strictly between 1:1.5 and 1:3. This stoichiometric precision allows the use of highly active chlorinating agents like dichlorohydantoin without compromising product integrity. The result is a dramatic improvement in both yield and purity of the abiraterone acetate intermediate (Formula II), as depicted in the reaction scheme below, establishing a new benchmark for efficiency in steroidal synthesis.
This novel chlorination route eliminates the need for expensive triflates and avoids the purification nightmares associated with iodination. By operating at mild temperatures ranging from -30°C to 30°C and utilizing common protic solvents like dichloromethane, the process is inherently safer and more energy-efficient. The subsequent steps involve a Suzuki coupling with diethyl 3-pyridine borane followed by a straightforward esterification, culminating in the final API. This streamlined sequence reduces the total number of unit operations and minimizes the accumulation of impurities, thereby ensuring a consistent supply of high-quality abiraterone acetate that meets rigorous international pharmacopeial standards.
Mechanistic Insights into Selective Chlorination and Impurity Control
The mechanistic success of this synthesis hinges on the delicate interplay between the chlorinating agent and the basic environment. In traditional halogenation, the high reactivity of the halogen source often leads to over-halogenation or degradation of the sensitive steroid backbone. However, by employing dichlorohydantoin or N-chlorosuccinimide in conjunction with a sterically hindered base like tetramethylguanidine, the reaction kinetics are finely tuned. The base acts not merely as an acid scavenger but as a regulator of the electrophilic chlorine species, ensuring that the substitution occurs exclusively at the desired position on the five-membered ring. This selectivity is further reinforced by the specific mass ratio of the substrate to the base, which creates an optimal microenvironment that suppresses competing side reactions.
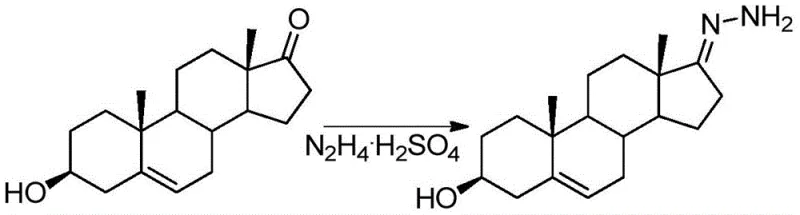
Moreover, the preparation of the starting material, Formula (I), is itself a critical determinant of final quality. As shown in the condensation reaction below, dehydroepiandrosterone reacts with hydrazine sulfate under mild thermal conditions to form the hydrazone precursor. This step is executed with high conversion rates, providing a clean feedstock for the subsequent chlorination. The integration of this efficient precursor synthesis with the novel chlorination step creates a cohesive process flow where impurity carryover is minimized. The rigorous control over reaction temperature, specifically maintaining the chlorination between -10°C and 15°C, further stabilizes the transition state, preventing thermal degradation and ensuring that the stereochemical integrity of the molecule is preserved throughout the transformation.
How to Synthesize Abiraterone Acetate Efficiently
The synthesis of abiraterone acetate via this patented route involves a logical sequence of condensation, chlorination, coupling, and esterification, designed for maximum operational simplicity and yield. The process begins with the formation of the hydrazone intermediate, followed by the pivotal chlorination step where reaction conditions must be strictly monitored to maintain the 1:1.5 to 1:3 substrate-to-base ratio. Following the isolation of the chlorinated intermediate, a palladium-catalyzed Suzuki coupling introduces the pyridine moiety, and the final step involves acetylation to yield the active pharmaceutical ingredient. For detailed operational parameters, solvent choices, and workup procedures, please refer to the standardized synthesis guide provided below.
- Condense dehydroepiandrosterone with hydrazine sulfate to form the hydrazone intermediate (Formula I).
- Perform selective chlorination on Formula I using dichlorohydantoin and tetramethylguanidine at -5°C to 3°C to obtain the chlorinated intermediate (Formula II).
- Execute a Suzuki coupling reaction between the chlorinated intermediate and diethyl 3-pyridine borane using a palladium catalyst to synthesize Formula III.
- Conduct final esterification of Formula III with acetic anhydride catalyzed by DMAP to yield Abiraterone Acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this chlorination-based synthesis route offers substantial strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the raw material portfolio. By replacing expensive and supply-constrained reagents like triflic anhydride or elemental iodine with commodity chemicals such as dichlorohydantoin and tetramethylguanidine, manufacturers can significantly insulate their production costs from market volatility. This shift to readily available, low-cost reagents translates directly into a more stable pricing structure for the final intermediate, allowing for better long-term budget forecasting and reduced exposure to raw material shortages that frequently plague the fine chemical sector.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, driven primarily by the elimination of high-cost inputs and the reduction of waste disposal expenses. Traditional iodination routes generate significant amounts of iodine-containing waste, which requires specialized and costly treatment protocols to meet environmental regulations. In contrast, the chlorination method produces cleaner waste streams that are easier and cheaper to manage. Furthermore, the higher yields achieved in the key intermediate step mean that less starting material is required per kilogram of final product, effectively lowering the cost of goods sold (COGS) without the need for complex capital investment in new equipment.
- Enhanced Supply Chain Reliability: Supply continuity is paramount for oncology drugs, and this synthetic route enhances reliability by reducing dependency on single-source or geographically concentrated raw materials. The reagents used, such as dichlorohydantoin and common organic solvents like dichloromethane and isopropanol, are produced globally by multiple suppliers, mitigating the risk of supply disruptions. Additionally, the robustness of the reaction conditions, which tolerate a reasonable range of temperatures and stoichiometry, ensures that production batches are less likely to fail due to minor process deviations, thereby guaranteeing a consistent and dependable flow of material to downstream formulation partners.
- Scalability and Environmental Compliance: From a scale-up perspective, the process is exceptionally well-suited for large-volume manufacturing. The reaction exotherms are manageable, and the workup procedures rely on standard liquid-liquid extractions and crystallizations that are easily transferred from pilot plants to multi-ton reactors. This scalability is complemented by a favorable environmental profile; the avoidance of heavy metal catalysts in the early stages and the use of recyclable solvents align with modern green chemistry principles. This compliance not only reduces regulatory hurdles but also enhances the corporate sustainability profile of the manufacturing entity, a factor increasingly weighted in vendor selection criteria by major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this abiraterone acetate synthesis technology. These answers are derived directly from the experimental data and claims presented in patent CN112125942A, providing a factual basis for evaluating the process viability. Understanding these details is crucial for technical teams assessing the fit of this route within their existing manufacturing infrastructure and for quality assurance teams verifying compliance with purity specifications.
Q: What are the key advantages of the chlorination method over the traditional iodination route?
A: The chlorination method described in patent CN112125942A offers significantly higher reaction selectivity and yield compared to iodination. While iodination often suffers from low yields and difficult purification due to side reactions, the controlled chlorination using dichlorohydantoin maintains high activity while minimizing impurities, resulting in intermediate purities exceeding 98%.
Q: How does the mass ratio of base to substrate impact the reaction outcome?
A: Controlling the mass ratio of the substrate (Formula I) to the base (such as tetramethylguanidine) between 1:1.5 and 1:3 is critical. This specific stoichiometric balance ensures that the high reaction activity of the chlorinating agent is maintained without triggering excessive side reactions, directly correlating to improved yield and purity of the abiraterone acetate intermediate.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the process is designed for scalability. It utilizes relatively inexpensive raw materials like dichlorohydantoin instead of costly triflates or unstable iodine reagents. Furthermore, the workup involves standard extraction and crystallization steps using common solvents like dichloromethane and isopropanol, making it highly adaptable for multi-kilogram to ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Abiraterone Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for life-saving oncology medications. Our team of expert chemists has thoroughly analyzed the technological advancements presented in CN112125942A and is fully prepared to leverage this novel chlorination methodology for commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of abiraterone acetate intermediate we deliver adheres to the highest global quality standards.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthetic route can be integrated into your supply chain. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this chlorination-based process for your operations. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to delivering high-purity pharmaceutical intermediates with unmatched reliability and speed.
