Revolutionizing Phenylbenzimidazole Production with Advanced Copper Complex Catalysis for Commercial Scale-Up
Revolutionizing Phenylbenzimidazole Production with Advanced Copper Complex Catalysis for Commercial Scale-Up
The pharmaceutical and agrochemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways for synthesizing critical heterocyclic scaffolds. A significant breakthrough in this domain is detailed in patent CN113200921A, which discloses a novel method for the catalytic synthesis of phenylbenzimidazole compounds using specialized copper complexes. This technology represents a paradigm shift from traditional high-energy processes to mild, room-temperature catalytic systems. By leveraging a unique meta-carborane ligand framework, the invention enables the direct coupling of benzimidazole with readily available halogenated hydrocarbons. For R&D directors and procurement managers alike, this development signals a potential for drastically reduced operational costs and simplified supply chains. The ability to produce high-purity phenylbenzimidazole intermediates under such benign conditions addresses long-standing challenges regarding waste generation and energy intensity in fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzimidazole derivatives has relied heavily on the condensation of o-phenylenediamine with organic carboxylic acids or their derivatives. This classical approach typically necessitates the use of strong acidic catalysts such as polyphosphoric acid, concentrated hydrochloric acid, or mixed acid systems to drive the cyclization and dehydration reactions. These conditions are inherently harsh, often requiring high-temperature reflux for extended periods to achieve moderate yields. Furthermore, the post-reaction processing is notoriously complex, involving neutralization of large volumes of corrosive acids and difficult purification steps to remove inorganic salts and tarry by-products. From a supply chain perspective, the reliance on aggressive reagents increases safety risks, equipment corrosion, and environmental disposal costs, making the conventional route less attractive for modern, green chemistry-compliant manufacturing facilities seeking reliable pharmaceutical intermediates suppliers.
The Novel Approach
In stark contrast to the legacy methods, the technology described in CN113200921A utilizes a sophisticated copper complex containing a meta-carborane ligand to catalyze the reaction between benzimidazole and halogenated hydrocarbons. This innovative route operates effectively at room temperature, eliminating the need for external heating and the associated energy expenditures. The use of cheap and easily obtainable halogenated hydrocarbons as substrates, coupled with simple inorganic bases like potassium carbonate, streamlines the raw material sourcing process. The reaction proceeds via a one-pot synthesis strategy, which significantly minimizes the number of unit operations required. This not only enhances the overall catalytic efficiency but also ensures that the final products are easy to separate with minimal waste residue generation. Such a robust and universal methodology offers substantial advantages for the commercial scale-up of complex heterocycles, providing a cleaner and more economically viable alternative for producing high-purity phenylbenzimidazole.
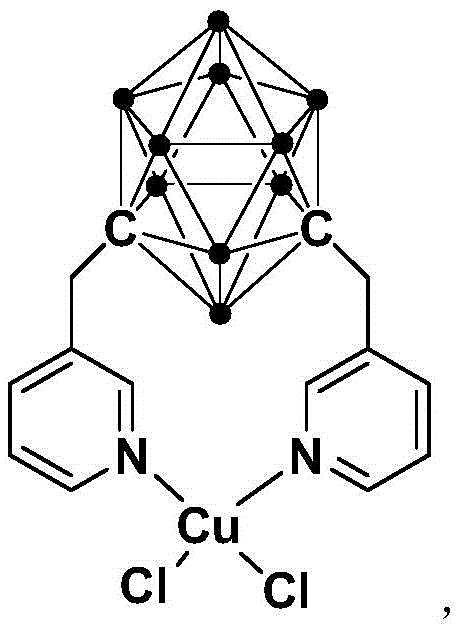
Mechanistic Insights into Copper-Catalyzed C-H Arylation
The core of this technological advancement lies in the unique structural properties of the copper complex catalyst featuring the meta-carborane ligand. The meta-carborane cage acts as a robust, three-dimensional scaffold that imparts exceptional stability to the metal center. Unlike traditional planar ligands, the steric bulk and electronic characteristics of the carborane moiety likely facilitate the oxidative addition of the aryl halide and the subsequent activation of the C2-H bond on the benzimidazole ring. This specific coordination environment prevents the aggregation of copper species, which is a common deactivation pathway in homogeneous catalysis. Consequently, the catalyst maintains high activity over prolonged reaction times, allowing for low catalyst loading while achieving excellent conversion rates. For technical teams, understanding this ligand effect is crucial, as it explains the system's tolerance to various functional groups on the aryl halide substrate, ranging from electron-donating methyl groups to electron-withdrawing nitro and fluoro substituents.
Furthermore, the mechanism inherently favors the formation of the desired 2-phenylbenzimidazole product with high selectivity, thereby suppressing the formation of regioisomers or poly-arylated impurities. The mild reaction conditions prevent the degradation of sensitive functional groups that might otherwise decompose under the strong acidic and thermal stress of conventional methods. This high level of chemoselectivity translates directly into a cleaner crude reaction profile, simplifying downstream purification efforts. The catalyst's insensitivity to air and moisture, as noted in the patent data, further suggests that the active catalytic species is kinetically stable, reducing the need for rigorous exclusion of atmospheric oxygen during the reaction setup. This robustness is a key factor in ensuring consistent batch-to-batch quality, a critical metric for any reliable pharmaceutical intermediates supplier aiming to meet stringent regulatory standards.
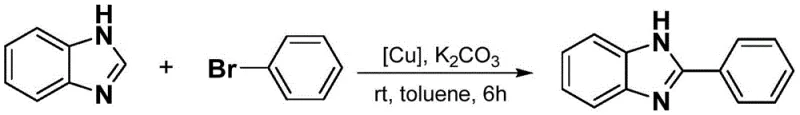
How to Synthesize Phenylbenzimidazole Efficiently
The practical implementation of this synthesis route is designed for ease of operation, making it highly suitable for both laboratory-scale optimization and industrial production. The process begins with the preparation of the reaction mixture, where the meta-carborane ligand copper complex is combined with benzimidazole, the chosen halogenated hydrocarbon, and a base in an organic solvent such as toluene. The simplicity of mixing these commercially available reagents at ambient temperature removes the complexity associated with cryogenic conditions or high-pressure reactors. Detailed standard operating procedures for this synthesis, including specific molar ratios and workup protocols, are outlined in the comprehensive guide below to ensure reproducibility and optimal yield.
- Prepare the reaction mixture by dissolving the meta-carborane ligand copper complex catalyst, benzimidazole substrate, halogenated hydrocarbon, and an inorganic base such as potassium carbonate in an organic solvent like toluene.
- Maintain the reaction system at room temperature and stir continuously for a duration of 6 to 8 hours to ensure complete conversion of the starting materials into the target phenylbenzimidazole compound.
- Upon completion, concentrate the reaction liquid and perform purification via silica gel column chromatography using a petroleum ether and ethyl acetate mixture to isolate the high-purity final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed methodology offers transformative benefits that extend beyond mere technical feasibility. The shift from expensive, hazardous acid catalysts to a reusable, stable copper complex fundamentally alters the cost structure of phenylbenzimidazole manufacturing. By eliminating the need for high-temperature reflux and corrosive acid handling, facilities can realize significant reductions in utility costs and maintenance expenses related to reactor lining and safety equipment. Moreover, the use of commodity chemicals like bromobenzene derivatives as starting materials ensures a stable and predictable supply chain, mitigating the risks associated with sourcing specialized precursors. This process intensification allows for faster turnaround times, effectively reducing lead time for high-purity intermediates and enabling manufacturers to respond more agilely to market demands.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the elimination of energy-intensive heating steps and the removal of costly acid neutralization and waste treatment processes. Since the reaction proceeds at room temperature, the energy footprint is drastically lowered compared to traditional thermal cyclizations. Additionally, the high catalytic efficiency means that less catalyst is required per kilogram of product, and the ease of product separation reduces solvent consumption during purification. These factors collectively contribute to a leaner manufacturing process with substantially lower variable costs, enhancing the overall competitiveness of the final API or agrochemical active ingredient in the global market.
- Enhanced Supply Chain Reliability: The robustness of the meta-carborane copper complex catalyst plays a pivotal role in securing supply continuity. Its stability against air and moisture simplifies logistics, as the catalyst does not require specialized cold-chain shipping or inert atmosphere storage. This resilience ensures that production schedules are not disrupted by catalyst degradation during transport or storage. Furthermore, the broad substrate scope allows manufacturers to switch between different aryl halide feedstocks based on availability and price fluctuations without needing to re-optimize the entire process, providing a flexible and resilient supply chain capable of adapting to raw material market dynamics.
- Scalability and Environmental Compliance: Scaling this process from grams to tons is facilitated by the one-pot nature of the reaction and the absence of hazardous gaseous by-products. The generation of minimal waste residue aligns perfectly with increasingly strict environmental regulations regarding industrial effluent and solid waste disposal. The simplified workup procedure, often requiring only filtration and chromatography, reduces the volume of aqueous waste streams typically generated by acid-base extractions. This green chemistry profile not only lowers compliance costs but also enhances the corporate sustainability image, a growing priority for multinational corporations evaluating their tier-one chemical partners.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the viability of this synthesis route for their specific applications, we have compiled answers to common inquiries based on the patent specifications. These questions address critical aspects such as catalyst stability, reaction scope, and operational parameters. Understanding these details is essential for assessing the fit of this technology within existing manufacturing frameworks and for planning successful technology transfer projects.
Q: What are the primary advantages of using the meta-carborane copper complex over traditional acid catalysts?
A: Unlike traditional methods requiring strong acids like polyphosphoric acid and high-temperature reflux, this copper complex catalyzes the reaction at room temperature. This eliminates the need for harsh acidic conditions, reduces energy consumption, and simplifies post-treatment procedures, leading to a greener and more cost-effective process.
Q: Is this catalytic system sensitive to air or moisture during storage and handling?
A: The patent data indicates that the synthesized copper complex possesses excellent physical and chemical stability, including thermal stability. It is described as being insensitive to air and water, which significantly facilitates its handling, storage, and transportation for industrial applications without requiring stringent inert atmosphere conditions.
Q: What is the substrate scope for the halogenated hydrocarbons in this synthesis?
A: The method demonstrates broad universality, successfully accommodating various substituted aryl halides including bromobenzene, methyl-bromobenzenes, fluoro-bromobenzenes, nitro-bromobenzenes, and methoxy-bromobenzenes. This versatility allows for the efficient production of a diverse library of phenylbenzimidazole derivatives essential for drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenylbenzimidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced catalytic technologies like the one described in CN113200921A for driving innovation in the pharmaceutical sector. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries are seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of phenylbenzimidazole intermediate delivered meets the highest quality standards required for drug substance manufacturing. We are committed to leveraging our technical expertise to optimize these copper-catalyzed routes for maximum efficiency and yield.
We invite you to collaborate with us to explore how this cutting-edge synthesis method can enhance your product portfolio and reduce your overall cost of goods. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us today to request specific COA data for our available intermediates and to discuss detailed route feasibility assessments for your upcoming projects. Let us be your partner in navigating the complexities of modern chemical synthesis and securing a competitive advantage in the global marketplace.
