Advanced Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for High-Value Pharmaceutical Intermediates
Advanced Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for High-Value Pharmaceutical Intermediates
The strategic incorporation of trifluoromethyl groups into heterocyclic scaffolds represents a cornerstone of modern medicinal chemistry, profoundly influencing the pharmacokinetic profiles of drug candidates. As detailed in patent CN110467579B, a novel preparation method for 5-trifluoromethyl substituted 1,2,4-triazole compounds has been developed, addressing critical bottlenecks in the synthesis of these high-value intermediates. The 1,2,4-triazole motif is ubiquitous in blockbuster pharmaceuticals, serving as a key structural element in antifungal agents like fluconazole and anticancer drugs such as letrozole, as well as in functional materials for OLED applications. 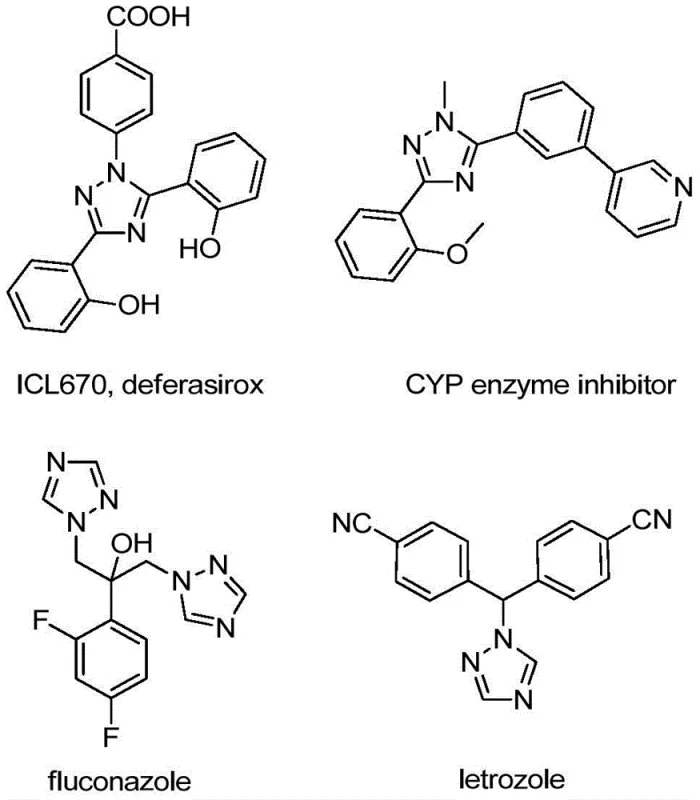 . This technological breakthrough offers a streamlined pathway to access these privileged structures, leveraging inexpensive starting materials and avoiding the complexities associated with traditional trifluoromethylation strategies. For R&D directors and procurement managers alike, this innovation signals a shift towards more sustainable and cost-efficient manufacturing protocols for complex nitrogen-containing heterocycles.
. This technological breakthrough offers a streamlined pathway to access these privileged structures, leveraging inexpensive starting materials and avoiding the complexities associated with traditional trifluoromethylation strategies. For R&D directors and procurement managers alike, this innovation signals a shift towards more sustainable and cost-efficient manufacturing protocols for complex nitrogen-containing heterocycles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted nitrogen heterocycles has relied heavily on two primary strategies, both of which present significant logistical and economic challenges for large-scale operations. The first approach involves the direct trifluoromethylation of pre-synthesized heterocyclic cores, a process that frequently necessitates the use of specialized, expensive, and often hazardous trifluoromethylating reagents. These reagents can be difficult to source in bulk quantities and often require stringent storage conditions, complicating supply chain management. The second mainstream method utilizes trifluoromethyl-bearing synthons, such as trifluorodiazoethane, which pose severe safety risks due to their explosive nature and instability. Furthermore, many existing protocols demand rigorous anhydrous and anaerobic conditions, requiring specialized reactor setups and inert gas manifolds that drive up capital expenditure and operational overhead. The reliance on transition metal catalysts in some variations also introduces the burden of heavy metal removal, a critical quality control step that adds time and cost to the purification process.
The Novel Approach
In stark contrast to these cumbersome legacy methods, the methodology disclosed in patent CN110467579B introduces a remarkably simple and efficient route utilizing non-metallic iodine promotion. This novel approach employs readily available hydrazones and trifluoroethylimidoyl chloride as the foundational building blocks, reacting them in the presence of sodium acetate and elemental iodine. The elimination of sensitive reagents and the avoidance of strict moisture-free or oxygen-free environments drastically lower the barrier to entry for production. By shifting the paradigm from complex cross-coupling or dangerous diazo chemistry to a robust condensation and oxidative cyclization sequence, this method enhances operational safety and reproducibility. The ability to synthesize diverse 1,2,4-triazole derivatives with trifluoromethyl groups at different positions through simple substrate design further underscores the versatility of this platform, making it an ideal candidate for the rapid generation of compound libraries for drug discovery programs.
Mechanistic Insights into Iodine-Promoted Cyclization
The mechanistic pathway of this transformation is a sophisticated interplay of base-promoted condensation and oxidative aromatization, offering deep insights for process chemists aiming to optimize reaction parameters. The reaction is hypothesized to initiate with a base-promoted intermolecular carbon-nitrogen bond formation between the hydrazone and the trifluoroethylimidoyl chloride, yielding a trifluoroacetamidine intermediate. This initial adduct then undergoes a critical isomerization step, setting the stage for the subsequent ring closure. The introduction of elemental iodine acts as a mild oxidant, facilitating an oxidative iodination that generates a reactive iodine-containing intermediate. This species subsequently undergoes an intramolecular electrophilic substitution reaction, effectively closing the five-membered triazole ring. The final step involves aromatization, driven by the elimination of hydrogen iodide, to furnish the stable 5-trifluoromethyl substituted 1,2,4-triazole product. 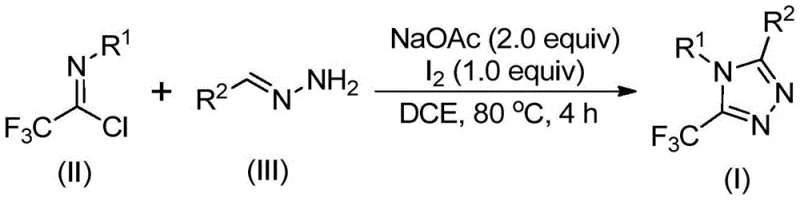 .
.
From an impurity control perspective, the use of sodium acetate as a mild base is particularly advantageous, as it minimizes the formation of side products often associated with stronger bases that might degrade sensitive functional groups on the aromatic rings. The tolerance for a wide range of substituents, including electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups like halogens and nitro groups, suggests a robust catalytic cycle that is not easily poisoned by substrate variations. This mechanistic resilience ensures a cleaner crude reaction profile, reducing the load on downstream purification units. For quality assurance teams, understanding that the reaction proceeds through a defined amidine intermediate allows for better monitoring of reaction progress, potentially enabling real-time adjustments to temperature or stoichiometry to maximize yield and minimize the formation of oligomeric byproducts.
How to Synthesize 5-Trifluoromethyl-1,2,4-Triazole Efficiently
The execution of this synthesis protocol is designed for maximum operational simplicity, allowing for seamless integration into existing pilot plant infrastructure without the need for exotic equipment. The process begins by charging a reactor with sodium acetate, trifluoroethylimidoyl chloride, and the chosen hydrazone substrate in a suitable organic solvent such as dichloroethane. The mixture is heated to a moderate temperature range of 80°C to 100°C, maintaining these conditions for a period of 2 to 4 hours to ensure complete conversion to the intermediate species. Following this initial phase, elemental iodine is introduced directly into the reaction vessel, and the heating is continued for an additional 1 to 2 hours to drive the oxidative cyclization to completion. Detailed standardized synthesis steps see the guide below.
- Combine sodium acetate, trifluoroethylimidoyl chloride, and hydrazone in an organic solvent such as dichloroethane.
- Heat the reaction mixture to 80°C for 2 to 4 hours to facilitate initial condensation and cyclization.
- Add elemental iodine to the system and continue heating for an additional 1 to 2 hours to complete the oxidative aromatization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates into tangible strategic advantages that extend far beyond the laboratory bench. The primary value driver is the substantial reduction in raw material costs, achieved by replacing expensive, specialized trifluoromethylating agents with commodity chemicals like hydrazones and imidoyl chlorides that are widely available in the global market. This shift mitigates the risk of supply disruptions caused by the limited production capacity of niche reagents, ensuring a more resilient and continuous supply of critical intermediates. Furthermore, the elimination of heavy metal catalysts removes the necessity for complex and costly metal scavenging steps, which are often required to meet the stringent residual metal limits imposed by regulatory bodies for pharmaceutical ingredients. This simplification of the downstream processing workflow not only accelerates the overall production timeline but also reduces the consumption of silica gel and solvents associated with extensive purification protocols.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, primarily driven by the replacement of high-cost reagents with inexpensive, bulk-available starting materials. By utilizing elemental iodine and sodium acetate instead of precious metal catalysts or unstable diazo compounds, the direct material cost per kilogram of product is significantly lowered. Additionally, the mild reaction conditions reduce energy consumption, as there is no need for cryogenic cooling or high-pressure reactors, leading to lower utility costs. The simplified workup procedure, which avoids complex extraction sequences required for metal removal, further contributes to overall cost efficiency by reducing labor hours and waste disposal fees associated with hazardous metal residues.
- Enhanced Supply Chain Reliability: Supply chain stability is markedly improved due to the reliance on commoditized feedstocks that possess multiple qualified suppliers globally. Unlike specialized fluorinating agents that may have long lead times or single-source dependencies, hydrazones and imidoyl chlorides can be sourced from a broad network of chemical manufacturers, reducing the risk of bottlenecking production schedules. The robustness of the reaction against moisture and oxygen means that transportation and storage requirements for raw materials are less stringent, lowering logistics costs and minimizing the potential for spoilage during transit. This reliability ensures that production timelines can be met consistently, supporting just-in-time manufacturing models essential for agile pharmaceutical development.
- Scalability and Environmental Compliance: The environmental footprint of this process is considerably smaller than traditional methods, aligning with increasingly strict global regulations on industrial emissions and waste. The absence of toxic heavy metals simplifies wastewater treatment and solid waste disposal, reducing the environmental compliance burden and associated fees. The method's proven scalability from gram-scale laboratory experiments to potential multi-ton production runs demonstrates its readiness for commercial deployment without the need for extensive re-engineering. This ease of scale-up allows manufacturers to respond rapidly to market demand surges, providing a competitive edge in the fast-paced landscape of generic drug production and custom synthesis.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this 5-trifluoromethyl-1,2,4-triazole synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the underlying patent documentation, providing a clear picture of the method's capabilities and limitations. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers below reflect the consensus on reaction performance, safety profiles, and scalability potential observed during the development of this process.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, the process utilizes elemental iodine and sodium acetate, avoiding the need for costly and toxic heavy metal catalysts often found in traditional cross-coupling reactions.
Q: What are the advantages regarding reaction conditions?
A: The method operates under mild conditions without the strict requirement for anhydrous or oxygen-free environments, significantly simplifying operational complexity and equipment requirements.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the use of cheap, commercially available starting materials and simple post-treatment procedures like filtration and column chromatography makes it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the pivotal role that high-quality heterocyclic intermediates play in the development of next-generation therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in handling fluorine chemistry and nitrogen heterocycles allows us to navigate the complexities of this specific synthesis with precision, guaranteeing batch-to-batch consistency that is critical for regulatory filings.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits this method offers for your specific supply chain. We encourage you to contact us to obtain specific COA data for our reference standards and to receive comprehensive route feasibility assessments that will help you make informed decisions about your intermediate sourcing strategy. Let us collaborate to accelerate your drug development timeline with reliable, cost-effective, and high-purity chemical solutions.
