Revolutionizing Pyridine Functionalization: A Strategic Breakthrough for High-Purity Pharmaceutical Intermediates
The landscape of heterocyclic chemistry is constantly evolving, driven by the relentless demand for more efficient pathways to construct complex molecular architectures found in modern therapeutics. Patent CN111533684B, published in late 2021, introduces a transformative methodology for the alkenylation of pyridine compounds, a structural motif ubiquitous in pharmaceuticals, agrochemicals, and functional materials. This innovation addresses long-standing challenges in organic synthesis by employing a sophisticated quaternary ammonium activation strategy. Unlike conventional approaches that often struggle with the inherent electron deficiency of the pyridine ring, this technique leverages the enhanced acidity of the C-H bond in quaternized pyridinium salts to facilitate smooth C-H functionalization. For R&D directors and process chemists seeking reliable pharmaceutical intermediate supplier solutions, this patent represents a significant leap forward, offering a route that bypasses the need for cumbersome pre-functionalization steps while delivering high yields and excellent stereoselectivity.
The significance of this technology extends beyond mere academic interest; it provides a tangible solution for cost reduction in API manufacturing by streamlining synthetic sequences. By utilizing a dual catalytic system involving palladium and copper, coupled with a specific ligand environment, the method achieves direct alkenylation with internal alkynes. This capability is crucial for the construction of stilbene-like pyridine derivatives, which serve as key scaffolds in drug discovery. The patent details a robust protocol that tolerates a wide range of substituents, ensuring that diverse chemical spaces can be explored efficiently. As we delve deeper into the technical specifics, it becomes evident that this approach not only solves immediate synthetic hurdles but also lays the groundwork for more sustainable and economically viable production processes in the fine chemical sector.
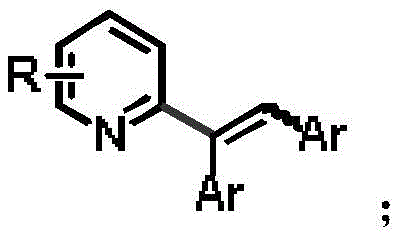
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of pyridine rings, particularly at the C2 position, has been fraught with difficulties due to the nitrogen atom's ability to coordinate with metal catalysts, often leading to catalyst poisoning and deactivated species. Traditional methods such as nucleophilic substitution or Friedel-Crafts alkylation frequently suffer from limited substrate scope, requiring harsh reaction conditions that are incompatible with sensitive functional groups. Furthermore, existing strategies often necessitate the installation of specific activating groups prior to the coupling reaction, followed by an additional step to remove these groups post-reaction. This multi-step sequence not only increases the overall processing time and resource consumption but also generates substantial chemical waste, posing challenges for environmental compliance and commercial scale-up of complex polymer additives or pharmaceutical precursors. The low activity of pyridine compounds towards aromatic electrophilic substitution further exacerbates these issues, often resulting in poor yields and difficult purification processes that hinder industrial adoption.
The Novel Approach
In stark contrast, the method disclosed in CN111533684B utilizes a quaternary ammonium activation strategy that fundamentally alters the reactivity profile of the pyridine substrate. By converting the pyridine into a quaternary pyridinium salt, typically using methyl iodide, the electron density of the ring is modulated, significantly increasing the acidity of the adjacent C-H bonds. This activation allows for direct C-H functionalization under much milder conditions compared to traditional routes. A distinct advantage of this approach is the spontaneous de-quaternization that occurs during the reaction process; the increased steric hindrance introduced by the alkenyl group facilitates the loss of the methyl group without the need for a separate demethylation step. This telescoping of activation, coupling, and de-protection into a single operational sequence drastically simplifies the workflow, reduces solvent usage, and minimizes the formation of by-products, thereby enhancing the overall atom economy and process efficiency for high-purity OLED material or drug intermediate synthesis.
Mechanistic Insights into Pd/Cu-Catalyzed C-H Alkenylation
The core of this innovation lies in its sophisticated catalytic cycle, which orchestrates the interaction between the quaternized pyridine and the internal alkyne. The reaction employs a palladium catalyst, such as Pd(OAc)2, in conjunction with a monovalent copper salt like CuBr and a bulky phosphine ligand, specifically tricyclohexylphosphine tetrafluoroborate. The mechanism initiates with the coordination of the palladium species to the activated C-H bond of the quaternary pyridinium salt, facilitated by the base system comprising potassium pivalate and triethylamine. This C-H activation step generates a key organopalladium intermediate. Subsequently, the internal alkyne inserts into the palladium-carbon bond, a step that is critically influenced by the steric and electronic properties of the ligand. The presence of the copper salt is believed to assist in the transmetallation or stabilization of reactive intermediates, ensuring the smooth progression of the catalytic cycle. The final reductive elimination releases the alkenylated product and regenerates the active palladium catalyst, ready for another turnover.
Impurity control is inherently built into this mechanistic design. The use of a specific ligand system and the dual base combination helps to suppress side reactions such as homocoupling of the alkyne or over-alkenylation. Moreover, the spontaneous de-quaternization acts as a driving force that pushes the equilibrium towards the desired neutral pyridine product, effectively preventing the accumulation of charged quaternary salt by-products. The reaction conditions, typically heating at 100-150°C for 15-32 hours, are optimized to balance reaction rate with selectivity, favoring the formation of the Z-isomer over the E-isomer with ratios often exceeding 8:1. This high stereoselectivity is vital for pharmaceutical applications where the biological activity can be strictly dependent on the geometric configuration of the double bond. Understanding these mechanistic nuances allows process chemists to fine-tune reaction parameters for optimal outcomes in large-scale production environments.
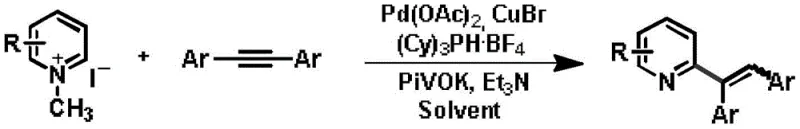
How to Synthesize Alkenylated Pyridine Efficiently
Implementing this synthesis route requires careful attention to reagent quality and reaction parameters to ensure reproducibility and high yield. The process begins with the preparation of the quaternary pyridinium salt, which can be readily synthesized by refluxing the corresponding pyridine derivative with methyl iodide in acetonitrile. This precursor is then subjected to the alkenylation conditions in a mixture of fluorobenzene, DMAc, and water, utilizing the specific catalyst and base loading described in the patent. The detailed standardized synthesis steps below outline the precise stoichiometry and workup procedures required to achieve the reported yields of up to 97%.
- Prepare the reaction mixture by combining pyridine quaternary ammonium salt, internal alkyne, palladium catalyst (e.g., Pd(OAc)2), phosphine ligand, monovalent copper salt, and a dual base system in a suitable solvent.
- Heat the mixture to a temperature range of 100-150°C and maintain stirring for 15 to 32 hours to facilitate the C-H activation and olefin insertion.
- Perform post-treatment involving filtration, solvent removal, extraction, and silica gel column chromatography to isolate the high-purity Z/E isomeric products.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers compelling economic and logistical benefits. The primary advantage stems from the significant simplification of the synthetic route. By eliminating the need for separate activation and de-protection steps, the overall number of unit operations is reduced, leading to lower labor costs, reduced energy consumption, and decreased solvent waste disposal fees. The raw materials required, such as pyridine derivatives, methyl iodide, and common internal alkynes, are commodity chemicals that are readily available from multiple global suppliers, ensuring a stable and resilient supply chain. This availability mitigates the risk of raw material shortages that often plague more exotic synthetic pathways, providing a secure foundation for long-term production planning.
- Cost Reduction in Manufacturing: The streamlined nature of this process directly translates to substantial cost savings. The telescoping of multiple reaction steps into a single pot operation reduces the requirement for intermediate isolation and purification, which are typically the most expensive phases of chemical manufacturing. Furthermore, the high selectivity of the reaction minimizes the formation of difficult-to-remove impurities, reducing the load on downstream purification units like chromatography columns. The use of earth-abundant copper salts alongside palladium helps optimize catalyst costs, and the ability to recover and recycle solvents like fluorobenzene further enhances the economic viability of the process for large volume production.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials ensures that production schedules are not held hostage by niche reagent lead times. The robustness of the reaction conditions, which tolerate a broad range of functional groups and substituents, means that the same platform technology can be applied to synthesize a diverse library of analogues without needing to requalify entirely new processes. This flexibility allows manufacturers to respond rapidly to changing market demands or clinical trial requirements, shortening the time from bench scale to commercial supply. The stability of the quaternary salt intermediates also allows for potential batch storage, adding another layer of security to the supply chain.
- Scalability and Environmental Compliance: From an environmental perspective, this method aligns well with green chemistry principles. The reduction in step count inherently lowers the E-factor (mass of waste per mass of product), making it easier to meet stringent environmental regulations. The reaction operates at moderate temperatures without the need for cryogenic conditions or extremely high pressures, simplifying the engineering requirements for scale-up. The spontaneous de-quaternization eliminates the generation of stoichiometric amounts of demethylation waste, further reducing the environmental footprint. These factors combined make the process highly attractive for contract development and manufacturing organizations (CDMOs) looking to offer sustainable and scalable solutions to their clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this pyridine alkenylation technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on yield expectations, substrate scope, and operational safety. Understanding these details is essential for evaluating the feasibility of integrating this method into existing manufacturing pipelines.
Q: What is the primary advantage of the quaternary ammonium activation strategy in pyridine synthesis?
A: The quaternary ammonium activation strategy significantly enhances the acidity of the C-H bond at the 2-position of the pyridine ring, facilitating easier C-H functionalization without the need for harsh pre-installation of activating groups. Furthermore, the activated group spontaneously removes itself via de-quaternization due to steric hindrance, simplifying the overall synthetic route.
Q: How does this method improve selectivity compared to traditional electrophilic substitution?
A: Traditional electrophilic substitution on pyridine is often challenging due to the electron-deficient nature of the ring. This patented method utilizes a transition metal catalytic cycle (Pd/Cu) that allows for precise regioselective alkenylation, predominantly yielding the Z-isomer with ratios as high as 9:1, thereby reducing the burden of separating complex isomeric mixtures.
Q: Is this synthesis method scalable for industrial production of API intermediates?
A: Yes, the method is highly scalable. It utilizes commercially available starting materials like pyridine and methyl iodide, operates under relatively mild heating conditions (100-150°C), and employs robust catalyst systems. The elimination of multiple protection/deprotection steps makes it economically viable for large-scale manufacturing of complex heterocyclic intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkenylated Pyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced synthetic technologies to maintain a competitive edge in the global pharmaceutical and fine chemical markets. Our team of expert process chemists has thoroughly analyzed the methodology presented in CN111533684B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with state-of-the-art facilities and rigorous QC labs capable of meeting stringent purity specifications required for GMP-grade intermediates. Our commitment to technical excellence ensures that we can translate this innovative laboratory-scale protocol into a robust, safe, and cost-effective industrial process tailored to your specific project needs.
We invite you to collaborate with us to leverage this cutting-edge alkenylation strategy for your next drug development program. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis that quantifies the potential efficiencies of switching to this route. We encourage you to reach out today to request specific COA data for similar pyridine derivatives and discuss route feasibility assessments. Let us help you accelerate your timeline to market with high-quality, reliably sourced chemical building blocks.
