Advanced Pyridine Alkenylation Technology for Commercial Scale-up of Complex Heterocycles
The pharmaceutical and agrochemical industries continuously demand efficient synthetic routes for functionalized heterocycles, particularly pyridine derivatives, which serve as critical scaffolds in numerous active pharmaceutical ingredients. Patent CN111533684A introduces a groundbreaking method for the alkenylation of pyridine compounds that addresses long-standing challenges in C-H functionalization. This technology utilizes a novel quaternary ammonium activation strategy to overcome the inherent low reactivity of pyridine rings towards electrophilic substitution. By leveraging a palladium-catalyzed system combined with a monovalent copper salt and a dual base system, this process enables the direct coupling of pyridine quaternary ammonium salts with internal alkynes. The significance of this innovation lies in its ability to bypass the harsh conditions and multi-step protection-deprotection sequences typically associated with traditional pyridine functionalization, thereby offering a streamlined pathway for the production of high-purity pharmaceutical intermediates.
Furthermore, the broad substrate scope demonstrated in the patent data suggests that this methodology is not limited to simple pyridine structures but extends to complex substituted variants, including those with alkyl, phenyl, and heteroaryl groups. This versatility is crucial for R&D directors seeking to diversify their chemical libraries without compromising on yield or purity. The reaction operates under relatively mild thermal conditions, typically between 100°C and 150°C, which enhances safety profiles and reduces energy consumption compared to high-temperature pyrolysis methods. As a reliable pharmaceutical intermediates supplier, understanding such technological advancements is key to maintaining a competitive edge in the global supply chain, ensuring that clients receive materials produced via the most efficient and sustainable methods available.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of pyridine rings has been plagued by significant synthetic hurdles, primarily due to the electron-deficient nature of the pyridine nitrogen which deactivates the ring towards electrophilic attack. Conventional strategies often rely on N-oxidation to activate the ring, followed by C-H functionalization and a subsequent reduction step to restore the pyridine core. This multi-step sequence not only increases the overall production time but also generates substantial chemical waste, negatively impacting the environmental footprint of the manufacturing process. Additionally, the requirement for pre-functionalization means that specific activating groups must be installed and later removed, adding unnecessary complexity and cost to the synthesis. These traditional methods often suffer from limited substrate scope, where only specific substitution patterns are tolerated, restricting the chemical diversity accessible to medicinal chemists. The harsh reaction conditions frequently required for these older protocols can also lead to decomposition of sensitive functional groups, resulting in lower overall yields and difficult purification challenges.
The Novel Approach
In stark contrast, the method disclosed in patent CN111533684A employs a quaternary ammonium activation strategy that fundamentally simplifies the synthetic workflow. By converting the pyridine into a quaternary ammonium salt, the C-H acidity at the ortho-position is significantly enhanced, facilitating direct C-H activation without the need for prior N-oxidation. This approach allows the reaction to proceed through a catalytic cycle involving C-H activation, alkyne insertion, and reductive elimination, followed by a spontaneous dequaternization step that regenerates the neutral pyridine product. 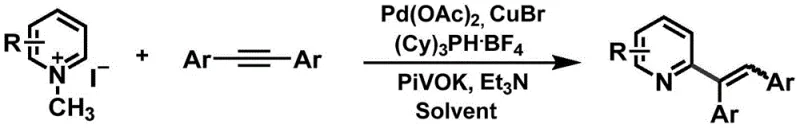 This elimination of extra activation and deprotection steps represents a major leap forward in process efficiency, directly translating to cost reduction in fine chemical manufacturing. The use of internal alkynes as coupling partners further expands the structural diversity of the resulting alkenylated pyridines, enabling the synthesis of complex motifs that are difficult to access via nucleophilic substitution or traditional cross-coupling reactions. The robustness of this new approach is evidenced by its ability to tolerate a wide range of functional groups, making it an ideal candidate for the late-stage functionalization of complex drug candidates.
This elimination of extra activation and deprotection steps represents a major leap forward in process efficiency, directly translating to cost reduction in fine chemical manufacturing. The use of internal alkynes as coupling partners further expands the structural diversity of the resulting alkenylated pyridines, enabling the synthesis of complex motifs that are difficult to access via nucleophilic substitution or traditional cross-coupling reactions. The robustness of this new approach is evidenced by its ability to tolerate a wide range of functional groups, making it an ideal candidate for the late-stage functionalization of complex drug candidates.
Mechanistic Insights into Pd-Catalyzed C-H Alkenylation
The core of this technological breakthrough lies in the sophisticated interplay between the palladium catalyst, the phosphine ligand, and the copper co-catalyst. The mechanism initiates with the coordination of the palladium species to the pyridine quaternary ammonium salt, facilitated by the increased acidity of the ortho-C-H bond. The presence of a monovalent copper salt, such as CuBr, plays a critical role in stabilizing the intermediate species and promoting the transmetallation step. The phosphine ligand, particularly bulky variants like tricyclohexylphosphine tetrafluoroborate, creates a specific steric environment around the metal center that dictates the stereochemical outcome of the reaction. 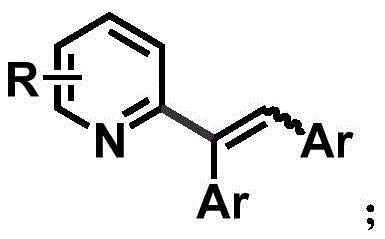 This steric control is essential for achieving the high Z-selectivity observed in the experimental data, where Z:E ratios can reach as high as 9:1. The reaction proceeds through a concerted metalation-deprotonation pathway, followed by the insertion of the internal alkyne into the palladium-carbon bond. The final reductive elimination step releases the alkenylated product and regenerates the active catalyst, completing the cycle. Understanding these mechanistic details is vital for R&D teams aiming to optimize reaction conditions for specific substrates, ensuring maximum efficiency and minimal impurity formation during scale-up.
This steric control is essential for achieving the high Z-selectivity observed in the experimental data, where Z:E ratios can reach as high as 9:1. The reaction proceeds through a concerted metalation-deprotonation pathway, followed by the insertion of the internal alkyne into the palladium-carbon bond. The final reductive elimination step releases the alkenylated product and regenerates the active catalyst, completing the cycle. Understanding these mechanistic details is vital for R&D teams aiming to optimize reaction conditions for specific substrates, ensuring maximum efficiency and minimal impurity formation during scale-up.
Impurity control is another critical aspect where this mechanism offers distinct advantages. The high selectivity of the catalytic system minimizes the formation of regioisomers and over-alkenylated by-products, which are common issues in less selective C-H functionalization methods. The spontaneous dequaternization step, driven by steric hindrance and nucleophilic assistance, ensures that the final product is the neutral pyridine derivative without requiring additional chemical reagents for demethylation. This inherent cleanliness of the reaction profile simplifies downstream processing, reducing the load on purification units and improving the overall yield of the target compound. For procurement managers, this translates to a more predictable supply of high-purity materials, reducing the risk of batch failures and ensuring consistent quality for downstream drug synthesis. The ability to fine-tune the Z/E ratio through ligand and base selection further empowers chemists to tailor the physical properties of the intermediates to meet specific formulation requirements.
How to Synthesize Alkenylated Pyridines Efficiently
Implementing this synthesis route in a laboratory or production setting requires careful attention to reagent quality and reaction parameters to replicate the high yields reported in the patent. The process begins with the preparation of the pyridine quaternary ammonium salt, which can be readily synthesized from pyridine and methyl iodide or purchased commercially, ensuring a reliable starting point for the reaction. The subsequent coupling reaction involves mixing the salt with the internal alkyne, palladium catalyst, ligand, copper salt, and a dual base system comprising both inorganic and organic bases.
- Prepare the reaction mixture by combining pyridine quaternary ammonium salt, internal alkyne, palladium catalyst, phosphine ligand, monovalent copper salt, and a dual base system in a suitable solvent.
- Heat the reaction mixture to a temperature range of 100-150°C and maintain stirring for 15 to 32 hours to ensure complete conversion and high Z-selectivity.
- Perform post-processing by filtering the mixture, washing with organic solvents, removing solvents under reduced pressure, and purifying the crude product via silica gel column chromatography.
This standardized approach ensures reproducibility and safety, making it accessible for both small-scale discovery chemistry and larger pilot plant operations. The use of common solvents like fluorobenzene and N,N-dimethylacetamide further enhances the practicality of the method, as these materials are widely available and easy to handle. By following these optimized protocols, manufacturers can achieve consistent results with total yields ranging from 56% to 97%, depending on the specific substrate combination. This level of performance underscores the viability of the technology for commercial applications, providing a robust foundation for the production of valuable pyridine-based intermediates.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this alkenylation technology offers substantial benefits for procurement and supply chain management, primarily driven by the simplification of the synthetic route. The elimination of pre-functionalization and deprotection steps significantly reduces the number of unit operations required, leading to a drastic simplification of the manufacturing process. This reduction in complexity directly correlates with lower operational costs, as fewer reactors, less labor, and reduced utility consumption are needed to produce the same amount of final product. Furthermore, the use of readily available starting materials, such as pyridine, methyl iodide, and common internal alkynes, mitigates supply chain risks associated with exotic or hard-to-source reagents. This availability ensures a stable supply of raw materials, reducing the likelihood of production delays caused by material shortages. The mild reaction conditions also contribute to enhanced safety and lower energy costs, making the process more sustainable and economically attractive for large-scale production.
- Cost Reduction in Manufacturing: The streamlined synthetic pathway eliminates the need for expensive activating groups and the reagents required for their removal, resulting in substantial cost savings. By reducing the number of synthetic steps, the overall material throughput is improved, and the consumption of solvents and reagents is minimized. This efficiency gain allows for a more competitive pricing structure without compromising on the quality of the final intermediate. Additionally, the high selectivity of the reaction reduces the burden on purification processes, further lowering the cost of goods sold. The ability to achieve high yields with common catalysts and ligands ensures that the process remains economically viable even when scaled to multi-ton quantities, providing a clear financial advantage over traditional methods.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals for the starting materials ensures a robust and resilient supply chain. Pyridine and methyl iodide are produced in large volumes globally, reducing the dependency on single-source suppliers for specialized precursors. This diversification of the supply base enhances the reliability of delivery schedules, ensuring that production timelines are met consistently. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, further stabilizing the supply chain. For supply chain heads, this translates to reduced lead time for high-purity intermediates and a lower risk of disruption due to raw material scarcity. The scalability of the process ensures that supply can be ramped up quickly to meet sudden increases in demand.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard equipment and conditions that are easily transferable from the laboratory to the pilot plant and full-scale production. The reduction in chemical waste generated by the elimination of extra steps aligns with increasingly stringent environmental regulations, reducing the cost and complexity of waste disposal. The use of less hazardous reagents and milder conditions improves the overall safety profile of the manufacturing site, reducing the risk of accidents and associated liabilities. This environmental and safety compliance is crucial for maintaining operational licenses and meeting the sustainability goals of modern pharmaceutical companies. The efficient use of resources and minimization of waste contribute to a greener manufacturing footprint, enhancing the corporate social responsibility profile of the production facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this pyridine alkenylation technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing accurate and reliable information for decision-makers. Understanding these details is essential for evaluating the feasibility of integrating this method into existing production workflows or new product development pipelines. The clarity provided here aims to eliminate uncertainties regarding reaction performance, scalability, and material availability.
Q: What are the primary advantages of the quaternary ammonium activation strategy over traditional N-oxidation methods?
A: The quaternary ammonium activation strategy eliminates the need for pre-installation and subsequent removal of activating groups, which are required in traditional N-oxidation methods. This simplifies the synthetic route, reduces waste generation, and improves overall atom economy.
Q: How does this method control the Z/E selectivity of the alkenylated products?
A: The method achieves high Z-selectivity, with Z:E ratios ranging from 3.4:1 to 9:1, by leveraging steric hindrance effects during the catalytic cycle. The specific combination of palladium catalyst, phosphine ligand, and copper salt facilitates a stereoselective insertion and elimination process.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the route is highly suitable for scale-up due to the use of readily available starting materials like pyridine and methyl iodide, mild reaction conditions (120°C), and a robust catalyst system that delivers high yields up to 97% without requiring cryogenic temperatures or ultra-high pressure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkenylated Pyridines Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the one described in patent CN111533684A for the production of high-value chemical intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure required to translate such innovative laboratory processes into robust commercial manufacturing operations. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of alkenylated pyridines meets the highest industry standards. Our capability to handle complex catalytic systems and sensitive intermediates positions us as an ideal partner for your most challenging synthesis projects.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through the adoption of this efficient alkenylation technology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production requirements. We encourage you to contact us to request specific COA data and route feasibility assessments for your target molecules. By leveraging our expertise in process development and scale-up, you can accelerate your time to market and secure a competitive advantage in the global marketplace. Let us help you turn this innovative chemistry into a commercial reality, ensuring a steady supply of high-quality intermediates for your pharmaceutical and agrochemical applications.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
