Advanced Chiral Bisoxazoline Ligands with Imidazolium Salts for Scalable Asymmetric Synthesis
Advanced Chiral Bisoxazoline Ligands with Imidazolium Salts for Scalable Asymmetric Synthesis
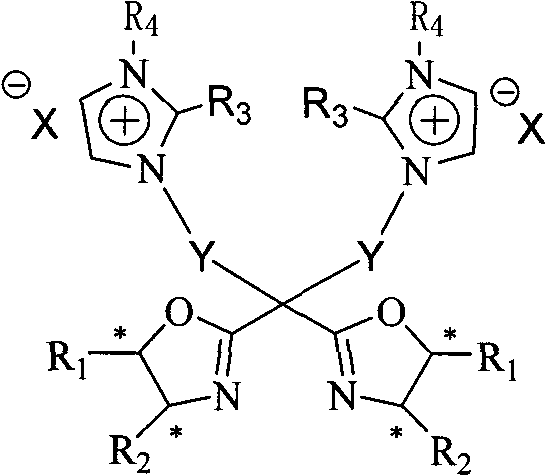
The landscape of asymmetric catalysis is undergoing a significant transformation driven by the urgent industrial need for sustainable and recoverable chiral systems. Patent CN102040594A discloses a groundbreaking class of C2-axis symmetric chiral bisoxazoline ligand compounds containing imidazolium salt ion-pair groups. This innovation addresses the critical bottleneck in homogeneous catalysis where expensive chiral ligands are often lost during product isolation. By integrating ionic liquid characteristics into the ligand structure, this technology enables a unique quasi-homogeneous catalytic environment that combines the high activity of homogeneous systems with the easy separability of heterogeneous ones. For R&D directors and process chemists, this represents a pivotal shift towards greener, more cost-effective manufacturing of high-value pharmaceutical intermediates and fine chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional chiral bisoxazoline metal complexes have long been revered for their exceptional catalytic performance in various asymmetric transformations such as cyclopropanation and aldol condensations. However, their industrial application has been severely hindered by the inherent difficulty in separating the catalyst from the reaction mixture post-synthesis. Conventional homogeneous catalysts require complex purification steps like column chromatography or extensive washing, which not only increases operational costs but also leads to significant ligand loss. Alternatively, immobilizing these catalysts on solid supports like silica gel or polymers often results in a dramatic decrease in catalytic activity and stereoselectivity due to the alteration of the chiral microenvironment around the metal center. This trade-off between recyclability and efficiency has remained a persistent challenge for procurement managers seeking to optimize supply chain costs.
The Novel Approach
The novel approach detailed in the patent utilizes a strategic molecular design where the chiral bisoxazoline backbone is functionalized with imidazolium salt ion pairs. This modification imparts ionic liquid-like properties to the ligand, allowing it to remain soluble in specific polar media during the reaction to ensure maximum substrate-catalyst contact. Crucially, upon completion of the reaction, the catalyst can be easily separated from organic products through simple extraction methods or phase separation, effectively preventing catalyst leaching. This dual nature allows the system to retain the high reaction rates and selectivity typical of homogeneous catalysis while offering the recovery benefits of heterogeneous systems. The result is a robust catalytic cycle that can be repeated numerous times without compromising performance, directly addressing the economic pain points of large-scale chemical manufacturing.
Mechanistic Insights into Imidazolium-Functionalized Bisoxazoline Catalysis
The core mechanism of this catalytic system relies on the precise spatial arrangement provided by the C2-axis symmetry of the bisoxazoline ligand. When coordinated with metal centers such as copper or zinc, the ligand creates a rigid chiral pocket that effectively discriminates between enantiotopic faces of the substrate. The presence of the imidazolium cation and its counter-anion (such as PF6, OTs, or halides) plays a multifaceted role beyond mere solubility. These ion pairs can interact with the transition state of the reaction, potentially stabilizing charged intermediates through electrostatic interactions, thereby accelerating the reaction rate. Furthermore, the ionic nature of the ligand ensures that the metal complex remains intact within the ionic phase during workup, minimizing metal contamination in the final API intermediate product, which is a critical quality attribute for regulatory compliance.
Impurity control is inherently enhanced by this design. In traditional systems, ligand decomposition or metal leaching can generate difficult-to-remove impurities that require costly downstream processing. The stability of the imidazolium-bisoxazoline framework under reaction conditions ensures that the ligand skeleton remains intact over multiple cycles. The patent data indicates that the catalyst maintains its activity and enantioselectivity even after fifteen recovery cycles, suggesting a highly durable coordination environment. This stability reduces the formation of side products associated with catalyst degradation, simplifying the purification process and improving the overall yield of the desired chiral building blocks. For supply chain heads, this reliability translates to consistent batch-to-batch quality and reduced waste disposal costs.
How to Synthesize Chiral Bisoxazoline Ligands Efficiently
The synthesis of these advanced ligands follows a logical and scalable multi-step pathway that begins with readily available chiral amino alcohols. The process involves the initial formation of the bisoxazoline ring system, followed by regioselective lithiation at the bridgehead position. This key intermediate is then alkylated with protected hydroxy-alkyl halides to introduce the linker arm. Subsequent deprotection reveals the diol functionality, which is activated via tosylation or halogenation. The final quaternization step involves reacting the activated intermediate with substituted imidazoles to install the ionic head groups. This modular approach allows for the tuning of steric and electronic properties by varying the substituents on the oxazoline rings or the imidazolium nitrogen atoms, providing a versatile platform for optimizing catalytic performance for specific substrates.
- Perform lithiation of the bisoxazoline precursor using butyllithium at low temperature followed by alkylation with silyl-protected alkyl halides.
- Hydrolyze the silyl protecting groups using tetrabutylammonium fluoride to reveal the dihydroxy intermediate.
- Activate the hydroxyl groups via tosylation or halogenation, then react with dimethylimidazole to form the final imidazolium salt ligand.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this imidazolium-supported chiral ligand technology offers substantial advantages in cost reduction and supply chain resilience. The primary economic driver is the drastic reduction in catalyst consumption. Because the catalyst can be recovered and reused multiple times without significant loss of activity, the effective cost per kilogram of the final product is significantly lowered. This eliminates the need for purchasing large quantities of expensive chiral ligands for single-use batches, directly impacting the bottom line for procurement managers. Additionally, the simplified workup procedure reduces the consumption of solvents and silica gel typically required for catalyst removal, further decreasing operational expenditures and environmental footprint.
- Cost Reduction in Manufacturing: The ability to recycle the catalyst up to fifteen times means that the amortized cost of the chiral ligand becomes negligible compared to traditional single-use systems. By eliminating the need for complex chromatographic purification to remove metal residues, manufacturers can save significantly on stationary phase costs and solvent usage. The high selectivity of the catalyst also minimizes the formation of unwanted enantiomers, reducing the material loss associated with recrystallization or chiral separation steps, leading to a more efficient overall mass balance.
- Enhanced Supply Chain Reliability: The synthetic route for these ligands utilizes common industrial reagents such as butyllithium, imidazole derivatives, and standard alkylating agents, ensuring a stable and secure supply of raw materials. The robustness of the catalyst system reduces the risk of batch failures due to catalyst degradation, ensuring consistent production schedules. For supply chain heads, this reliability is crucial for maintaining continuous manufacturing operations and meeting strict delivery deadlines for downstream pharmaceutical clients without unexpected interruptions.
- Scalability and Environmental Compliance: The process is designed for scalability, moving seamlessly from gram-scale laboratory optimization to multi-kilogram production. The use of ionic liquid-like properties facilitates safer handling of reactive metal species and reduces the volatility of organic solvents required for extraction. This aligns with increasingly stringent environmental regulations regarding heavy metal discharge and solvent emissions. The simplified separation process generates less hazardous waste, making it easier for facilities to maintain compliance with environmental standards while scaling up production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral ligand technology in industrial settings. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the system's capabilities and limitations for potential adopters.
Q: How many times can the imidazolium-based chiral catalyst be recycled?
A: According to patent data, the catalyst maintains its activity and enantioselectivity even after being recovered and reused fifteen times without significant degradation.
Q: Which metals are compatible with this bisoxazoline ligand system?
A: The ligand forms stable and active complexes with a variety of transition metals including copper, zinc, magnesium, iron, and nickel for different asymmetric transformations.
Q: What specific asymmetric reactions does this catalyst support?
A: The catalyst system is specifically validated for asymmetric Diels-Alder reactions and asymmetric Henry reactions, showing high ee values and yields.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Bisoxazoline Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the imidazolium-functionalized bisoxazoline ligands described in CN102040594A. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory concepts are successfully translated into robust industrial processes. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of chiral intermediate meets the highest global standards for pharmaceutical applications. We are committed to delivering high-purity chiral ligands and catalysts that empower your synthesis teams to achieve superior enantioselectivity.
We invite you to collaborate with our technical team to explore how this recyclable catalyst system can optimize your specific manufacturing challenges. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our supply solutions can enhance your operational efficiency and reduce time-to-market for your critical drug candidates.
