Advanced Manufacturing of Edoxaban Chiral Amine Intermediates via Safe DPPA Chemistry
The pharmaceutical industry continuously seeks robust synthetic pathways for anticoagulant agents, particularly for the direct factor Xa inhibitor Edoxaban. A pivotal advancement in this domain is documented in Chinese Patent CN111606827B, which discloses a novel, safe, and industrially viable method for preparing the key chiral amine intermediate N-[(1R,2S,5S)-2-amino-5-[(dimethylamino)carbonyl]cyclohexyl]carbamic acid tert-butyl ester. This technology addresses critical safety and purity bottlenecks associated with legacy manufacturing processes by replacing hazardous metal azides with diphenylphosphoryl azide (DPPA). For global procurement leaders and R&D directors, this patent represents a significant opportunity to enhance supply chain resilience while mitigating regulatory risks associated with genotoxic impurities and nitrosamines. The following analysis details the technical superiority and commercial viability of this approach for large-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
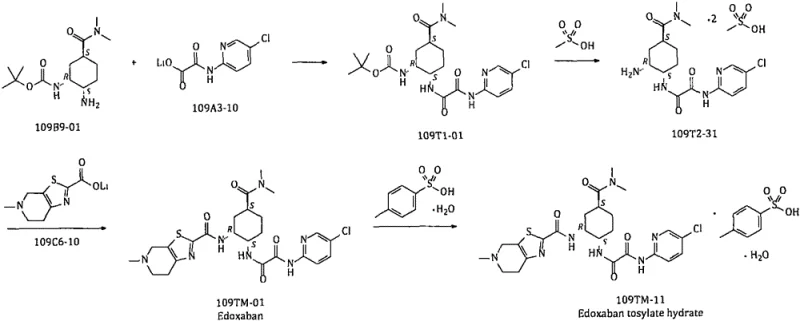
Historically, the synthesis of Edoxaban intermediates has relied on routes that introduce significant operational hazards and cost inefficiencies, as illustrated in the conventional pathway above. Traditional methods frequently employ highly explosive metal azide salts, such as sodium azide, which require stringent safety protocols and specialized containment infrastructure to prevent catastrophic accidents during storage and reaction. Furthermore, these legacy routes often necessitate the use of expensive noble metal catalysts like Palladium on Carbon (Pd/C) for hydrogenation steps, which not only increases raw material costs but also introduces the risk of heavy metal residue in the final active pharmaceutical ingredient. The reliance on polar aprotic solvents like DMF or DMAc in these older processes further exacerbates the risk of forming carcinogenic nitrosamine impurities, creating a complex purification burden that delays time-to-market and compromises batch consistency.
The Novel Approach
In stark contrast, the methodology outlined in CN111606827B fundamentally reengineers the synthetic logic to prioritize safety and scalability without compromising yield. By utilizing diphenylphosphoryl azide (DPPA) in the presence of DBU, the process generates the necessary azide functionality in situ, effectively eliminating the need to handle bulk quantities of unstable metal azide salts. This strategic shift allows the reaction to proceed in safer hydrocarbon solvents such as toluene and n-heptane, which inherently suppresses the formation of nitrosamine byproducts that are commonly associated with amide solvents. Additionally, the replacement of catalytic hydrogenation with a triphenylphosphine-mediated reduction step removes the dependency on high-pressure equipment and precious metals, resulting in a streamlined workflow that is far more conducive to continuous manufacturing and rapid commercial scale-up.
Mechanistic Insights into DPPA-Mediated Azidation and Reduction
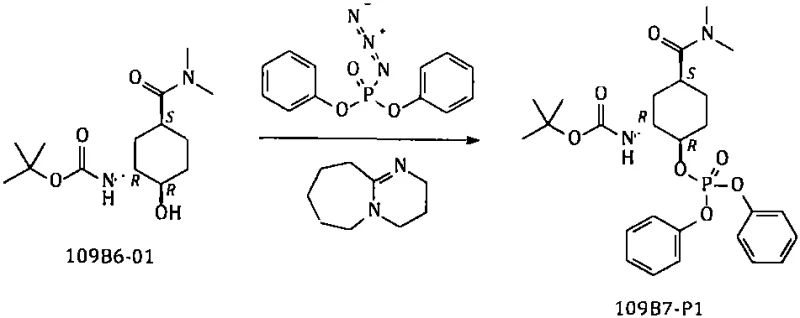
The core innovation of this technology lies in the precise mechanistic execution of the azidation step, where the hydroxyl group of the Boc-protected amino alcohol is activated and substituted under mild thermal conditions. As depicted in the reaction scheme, the interaction between the substrate, DPPA, and DBU initially forms a diphenoxyphosphoryl oxy intermediate, which serves as a superior leaving group compared to traditional sulfonates. This intermediate is then subjected to nucleophilic substitution by azide ions generated within the system upon the addition of a base, driving the conversion to the desired azido compound with high stereochemical fidelity. The use of DBU as a non-nucleophilic base ensures that side reactions are minimized, while the specific solvent choice of toluene facilitates the easy removal of byproducts through aqueous workup, thereby enhancing the overall purity profile of the crude material before crystallization.
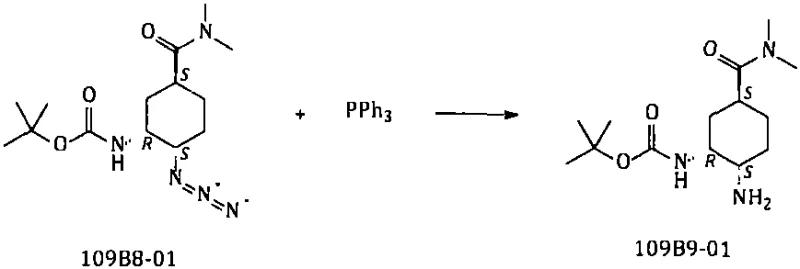
Following the azidation, the reduction of the azide group to the primary amine is achieved through a Staudinger-type reduction using triphenylphosphine, which offers distinct advantages over catalytic methods. This chemical reduction proceeds smoothly in a tetrahydrofuran and water mixture at moderate temperatures, avoiding the high energy input and safety risks associated with high-pressure hydrogen gas. The mechanism involves the formation of an aza-ylide intermediate which is subsequently hydrolyzed to release the free amine and triphenylphosphine oxide, a byproduct that is easily separated during the extraction and crystallization phases. This approach not only guarantees the absence of palladium residues, simplifying the downstream purification process, but also ensures that the chiral integrity of the cyclohexyl ring is maintained throughout the transformation, delivering a product that meets stringent enantiomeric excess specifications required for API synthesis.
How to Synthesize Edoxaban Chiral Amine Efficiently
To implement this advanced synthetic route in a production environment, operators must adhere to specific parameter controls regarding temperature and stoichiometry to maximize yield and safety. The process begins with the ammonolysis of the epoxy precursor followed by Boc protection, setting the stage for the critical DPPA substitution which requires careful monitoring of the exotherm during base addition. Detailed standard operating procedures dictate the precise addition rates of DBU and the maintenance of reaction temperatures between 45°C and 105°C depending on the specific stage, ensuring complete conversion while preventing thermal degradation of the sensitive azide intermediate. The final isolation involves crystallization from ethyl acetate and n-heptane mixtures, a technique that leverages solubility differences to purge impurities effectively.
- React Boc-protected amino alcohol with Diphenylphosphoryl Azide (DPPA) and DBU in toluene to form the phosphate intermediate.
- Add base and heat to substitute the phosphate group with an azide group, forming the azido intermediate without metal salts.
- Reduce the azide group to an amine using Triphenylphosphine in THF/Water, avoiding expensive Pd/C hydrogenation.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this patent-protected methodology offers substantial advantages in terms of cost structure stability and supply continuity for pharmaceutical manufacturers. By eliminating the requirement for explosive metal azides and expensive palladium catalysts, the process significantly reduces the volatility associated with raw material pricing and availability, allowing for more accurate long-term budget forecasting. The simplified safety profile means that production can be executed in standard multipurpose reactors without the need for specialized blast-proof facilities, thereby lowering capital expenditure barriers and increasing the pool of qualified contract manufacturing organizations capable of executing the campaign. Furthermore, the avoidance of genotoxic sulfonates and nitrosamine-prone solvents reduces the regulatory burden and testing costs associated with batch release, accelerating the speed at which products can reach the market.
- Cost Reduction in Manufacturing: The elimination of noble metal catalysts such as Pd/C directly translates to lower variable costs per kilogram, as there is no need for costly catalyst recovery systems or extensive metal scavenging steps. Additionally, the use of commodity hydrocarbon solvents like toluene and heptane instead of specialized polar aprotic solvents reduces raw material expenses and simplifies solvent recovery distillation trains. The higher throughput achievable due to improved safety margins allows for larger batch sizes, which spreads fixed operational costs over a greater volume of output, driving down the unit cost of the intermediate significantly.
- Enhanced Supply Chain Reliability: Sourcing diphenylphosphoryl azide and triphenylphosphine is generally more stable and less regulated than procuring bulk sodium azide, which is subject to strict transportation and storage restrictions globally. This flexibility enables suppliers to maintain healthier inventory levels and respond more rapidly to demand surges without facing logistical bottlenecks related to hazardous material shipping. The robustness of the chemistry also implies fewer batch failures due to safety interlocks or contamination issues, ensuring a consistent flow of material to downstream API synthesis sites and preventing production stoppages.
- Scalability and Environmental Compliance: The process generates waste streams that are easier to treat, as the absence of heavy metals and persistent organic pollutants simplifies effluent management and reduces environmental compliance costs. The ability to run the reaction at atmospheric pressure during the reduction phase removes the need for complex high-pressure hydrogenation infrastructure, making it easier to scale from pilot plant to multi-ton commercial production without significant engineering redesigns. This inherent scalability ensures that the supply chain can grow in tandem with the market demand for Edoxaban, providing a secure foundation for long-term commercial partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route, derived directly from the patent specifications and process data. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer and for procurement officers assessing supplier capabilities. The answers reflect the specific advantages of the DPPA and triphenylphosphine methodology over traditional approaches, highlighting its suitability for modern GMP manufacturing environments.
Q: Why is the DPPA route safer than traditional azide methods?
A: Traditional methods use explosive metal azides like sodium azide. This patent uses Diphenylphosphoryl Azide (DPPA), which generates azide in situ, significantly lowering explosion risks and improving handling safety.
Q: How does this process reduce nitrosamine risks?
A: The process avoids amide solvents like DMF or DMAc and nitrite sources. By using hydrocarbon solvents like toluene and heptane, the formation of carcinogenic nitrosamine impurities is drastically minimized.
Q: What are the cost benefits of using Triphenylphosphine reduction?
A: Replacing Pd/C catalytic hydrogenation with Triphenylphosphine reduction eliminates the need for expensive noble metal catalysts and high-pressure hydrogenation equipment, simplifying the operational setup and reducing capital expenditure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Edoxaban Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer, more efficient synthetic routes requires a partner with deep technical expertise and proven manufacturing capacity. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN111606827B are fully realized in practical, large-scale operations. We maintain stringent purity specifications across all our facilities, supported by rigorous QC labs equipped to detect trace impurities including potential nitrosamines and residual metals, guaranteeing that every batch meets the exacting standards required by global regulatory agencies.
We invite you to engage with our technical procurement team to discuss how this advanced manufacturing platform can optimize your supply chain for Edoxaban intermediates. By requesting a Customized Cost-Saving Analysis, you can gain visibility into the specific economic benefits of switching to this DPPA-based route for your project. We encourage potential partners to contact us directly to obtain specific COA data from our recent pilot runs and to schedule a comprehensive review of route feasibility assessments tailored to your specific volume requirements and timeline constraints.
