Advanced One-Pot Synthesis of Symmetrical 1,2,3-Triazole Derivatives for Pharmaceutical Applications
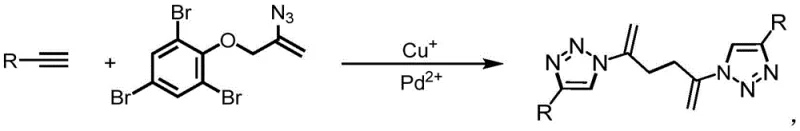
The pharmaceutical industry continuously seeks robust synthetic methodologies that balance structural complexity with manufacturing efficiency. Patent CN113105401B introduces a significant advancement in this domain by disclosing a novel preparation method for symmetrical 1,2,3-triazole derivatives. These compounds are pivotal in modern medicinal chemistry, often serving as bioisosteres for amide bonds or acting as key pharmacophores in anti-inflammatory and anti-tumor agents. The core innovation lies in a bimetallic catalytic system utilizing monovalent copper and divalent palladium salts to facilitate a one-pot coupling reaction between terminal alkynes and aryl azide compounds. This approach directly addresses the longstanding challenges of multi-step syntheses, offering a streamlined pathway to high-value pharmaceutical intermediates with exceptional purity profiles.
Historically, the construction of 1,2,3-triazole scaffolds has relied heavily on classical click chemistry protocols or stepwise cyclizations that often suffer from operational inefficiencies. Conventional methods frequently require the pre-formation of unstable diazonium salts or the use of stoichiometric amounts of hazardous reagents, leading to complex workup procedures and significant safety risks. Furthermore, achieving structural symmetry in triazole derivatives typically demands convergent strategies that multiply the number of unit operations, thereby inflating production costs and extending lead times. The accumulation of by-products in these traditional routes often necessitates rigorous purification steps, such as repeated recrystallization or preparative HPLC, which severely impact the overall mass balance and commercial viability of the final active pharmaceutical ingredient.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The legacy technologies for synthesizing symmetrical triazole derivatives are plagued by inherent thermodynamic and kinetic barriers that hinder industrial adoption. Standard protocols often operate under extreme conditions, requiring cryogenic temperatures for diazotization or elevated pressures for cycloaddition, which imposes heavy demands on reactor infrastructure and energy consumption. Additionally, the reliance on single-metal catalysis, such as copper-only systems, can sometimes lead to incomplete conversions or the formation of regio-isomeric mixtures that are difficult to separate. The handling of free azide intermediates in batch processes poses a severe safety hazard due to their potential for explosive decomposition upon thermal or mechanical shock. Consequently, manufacturers face substantial regulatory hurdles and insurance costs when scaling these dangerous chemistries, limiting the supply continuity of critical drug substances.
The Novel Approach
In stark contrast, the methodology described in CN113105401B leverages a synergistic bimetallic catalytic cycle to overcome these barriers efficiently. By employing a combination of copper and palladium species, the reaction achieves high selectivity and conversion under remarkably mild conditions, typically around 80°C in polar aprotic solvents like DMSO. This one-pot strategy merges the formation of the triazole rings with the coupling of the central linker, effectively collapsing multiple synthetic steps into a single operation. The result is a dramatic simplification of the process flow, where raw materials are converted directly into the target symmetrical structure with minimal generation of waste. This technological leap not only enhances the safety profile by avoiding the isolation of energetic intermediates but also ensures a consistent impurity profile that is easier to control during quality assurance testing.
Mechanistic Insights into Cu/Pd Bimetallic Catalyzed Cyclization
The success of this synthetic route hinges on the intricate interplay between the monovalent copper and divalent palladium catalysts within the reaction medium. The copper species, particularly when sourced from cuprous sulfide or cuprous iodide, acts as the primary activator for the terminal alkyne, facilitating the formation of a copper-acetylide intermediate. Simultaneously, the palladium component, often introduced as palladium acetate, likely assists in the activation of the aryl azide or stabilizes the transition state during the cycloaddition event. This dual-activation mechanism lowers the activation energy barrier for the 1,3-dipolar cycloaddition, allowing the reaction to proceed rapidly even at moderate temperatures. The presence of additives such as DBU (1,8-Diazabicyclo[5.4.0]undec-7-ene) further promotes the deprotonation of the alkyne and maintains the basicity required to sustain the catalytic cycle without degrading the sensitive azide functionality.
From an impurity control perspective, this mechanism offers distinct advantages over traditional acid-catalyzed or thermal methods. The high specificity of the metal-ligand interactions ensures that the cycloaddition occurs exclusively at the terminal position of the alkyne, preventing the formation of unwanted 1,4-disubstituted isomers or polymeric by-products. The use of DMSO as a solvent plays a crucial role in solubilizing both the organic substrates and the inorganic catalyst salts, creating a homogeneous reaction environment that minimizes local hot spots and side reactions. Post-reaction analysis indicates that the major impurities are easily removable via standard silica gel chromatography, yielding products with purity levels suitable for downstream biological testing. This clean reaction profile is essential for pharmaceutical applications, where strict limits on genotoxic impurities and heavy metal residues must be maintained.
How to Synthesize Symmetrical 1,2,3-Triazole Derivatives Efficiently
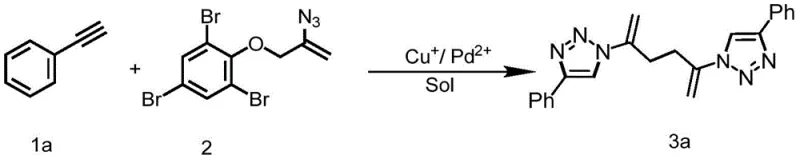
The practical implementation of this patented technology involves a straightforward procedure that is well-suited for both laboratory optimization and plant-scale manufacturing. The process begins with the dissolution of the terminal alkyne and the phenol-substituted alkenyl azide compound in a suitable solvent, followed by the sequential addition of the base and the bimetallic catalyst system. Maintaining an inert atmosphere, typically nitrogen, is recommended to prevent oxidation of the copper catalyst and ensure reproducible yields. The reaction mixture is then heated to the optimal temperature range of 60-100°C, with 80°C identified as the sweet spot for balancing reaction rate and selectivity. Upon completion, indicated by the disappearance of the starting materials via TLC, the product is isolated through a simple aqueous workup and extraction sequence.
- Dissolve terminal alkyne and phenol-substituted alkenyl azide compound in DMSO solvent with DBU additive.
- Add monovalent copper salt (e.g., Cu2S) and divalent palladium salt (e.g., Pd(OAc)2) under nitrogen protection.
- Heat the mixture to 80°C for 4 hours, then perform extraction and silica gel column chromatography for purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this bimetallic synthesis route translates into tangible strategic benefits that extend beyond mere technical feasibility. The consolidation of multiple reaction steps into a single pot significantly reduces the consumption of solvents, reagents, and energy, leading to a lower cost of goods sold (COGS). Furthermore, the use of commercially available and relatively inexpensive starting materials, such as substituted phenylacetylenes and simple azide precursors, mitigates the risk of raw material shortages. The robustness of the reaction conditions allows for flexible scheduling and reduced downtime, ensuring a reliable flow of intermediates to support continuous manufacturing campaigns.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation steps removes the need for extensive drying, filtration, and storage infrastructure, directly lowering capital expenditure and operational overhead. The high catalytic efficiency means that lower loading of precious metals is required, and the ability to use cost-effective copper salts like cuprous sulfide further drives down material costs. Additionally, the high yields reported in the patent examples minimize the loss of valuable starting materials, maximizing the return on investment for every batch produced.
- Enhanced Supply Chain Reliability: By simplifying the synthetic pathway, the potential for batch failures due to human error or equipment malfunction is substantially reduced. The mild reaction conditions are compatible with standard glass-lined or stainless steel reactors found in most multipurpose chemical plants, removing the need for specialized high-pressure or cryogenic equipment. This universality allows for easier technology transfer between sites and the qualification of multiple backup suppliers, thereby strengthening the resilience of the global supply network against disruptions.
- Scalability and Environmental Compliance: The process generates fewer waste streams compared to traditional multi-step syntheses, aligning with green chemistry principles and reducing the burden on wastewater treatment facilities. The solvents used, primarily DMSO, are widely recycled in the industry, and the absence of highly toxic reagents simplifies regulatory compliance and environmental permitting. This eco-friendly profile not only reduces disposal costs but also enhances the corporate sustainability metrics of the manufacturing organization, which is increasingly important for partnerships with major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific experimental data and beneficial effects outlined in the patent documentation, providing clarity on safety, scalability, and application potential. Understanding these aspects is crucial for R&D teams evaluating the route for new drug candidates and for operations teams planning for commercial production.
Q: How does this bimetallic method improve safety compared to traditional azide reactions?
A: Traditional methods often involve unstable diazonium intermediates or harsh conditions. This patent utilizes a mild one-pot protocol at 80°C with stable catalysts, significantly reducing the risk of explosive decomposition associated with free azides while maintaining high conversion rates.
Q: What represents the primary cost advantage of this synthesis route for procurement teams?
A: The process eliminates the need for multi-step isolation of intermediates and uses inexpensive, earth-abundant copper salts alongside palladium. The high yield (up to 91%) and simplified workup (direct extraction and chromatography) drastically reduce solvent consumption and labor costs per kilogram of API intermediate produced.
Q: Is this synthetic route scalable for industrial manufacturing?
A: Yes, the reaction operates at moderate temperatures (60-100°C) in common polar aprotic solvents like DMSO. The robustness of the Cu/Pd catalytic system allows for consistent performance across different substrate scopes, making it highly suitable for commercial scale-up from pilot plants to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,3-Triazole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN113105401B for accelerating drug discovery and development. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the bench to the market. Our state-of-the-art facilities are equipped to handle bimetallic catalytic reactions with precision, supported by stringent purity specifications and rigorous QC labs that guarantee the quality of every batch of high-purity pharmaceutical intermediates we deliver.
We invite you to collaborate with our technical team to explore how this efficient synthesis route can optimize your specific project requirements. By leveraging our expertise, you can obtain a Customized Cost-Saving Analysis tailored to your volume needs and timeline. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
