Scalable Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial API Manufacturing
Scalable Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial API Manufacturing
The pharmaceutical industry continuously seeks robust synthetic routes for nitrogen-containing heterocycles due to their prevalence in bioactive molecules. Patent CN111675662B discloses a significant advancement in the preparation of 2-trifluoromethyl substituted quinazolinone compounds, a scaffold found in numerous drugs exhibiting anti-cancer, anticonvulsant, and anti-inflammatory activities. This technology leverages an earth-abundant iron catalyst to drive the cyclization of readily available isatin derivatives and trifluoroethylimidoyl chlorides. By shifting away from expensive precious metals and harsh fluorinating agents, this method offers a compelling value proposition for a reliable pharmaceutical intermediate supplier aiming to optimize production costs while maintaining high purity standards.
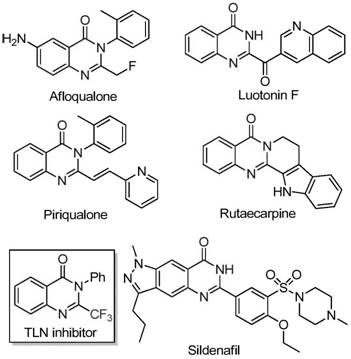
The introduction of the trifluoromethyl group into heterocyclic systems is a critical strategy in modern medicinal chemistry to enhance metabolic stability and lipophilicity. However, traditional methods often rely on costly synthons like trifluoroacetic anhydride or ethyl trifluoroacetate, which can limit the economic feasibility of large-scale manufacturing. The disclosed invention addresses these limitations by utilizing a novel combination of starting materials that are not only commercially accessible but also allow for greater structural diversity. This approach facilitates the rapid generation of compound libraries for drug discovery programs while ensuring that the eventual commercial scale-up of complex pharmaceutical intermediates remains economically viable and environmentally sustainable.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted quinazolinones has been plagued by several operational and economic bottlenecks that hinder efficient manufacturing. Conventional protocols frequently necessitate the use of severe reaction conditions, including strong bases or highly reactive fluorinating agents that pose significant safety hazards in a plant setting. Moreover, the reliance on precious metal catalysts introduces substantial cost volatility and creates stringent regulatory hurdles regarding residual metal limits in final Active Pharmaceutical Ingredients (APIs). These factors collectively contribute to extended production timelines and increased waste generation, making cost reduction in API manufacturing a challenging objective when adhering to legacy synthetic pathways that lack atom economy and operational simplicity.
The Novel Approach
In contrast, the methodology described in patent CN111675662B presents a streamlined alternative that utilizes ferric chloride as a cheap and effective catalyst under aerobic conditions. This novel route employs trifluoroethylimidoyl chloride and isatin as key building blocks, reacting them in a polar aprotic solvent like DMF to achieve high conversion rates. The process operates through a tandem sequence involving initial alkali-promoted bond formation followed by iron-catalyzed decarbonylation and cyclization. This strategic design not only broadens the substrate scope to include various substituted aryl groups but also simplifies the post-reaction workup to basic filtration and chromatography, thereby enhancing the overall throughput and reliability of the supply chain for these valuable heterocyclic intermediates.
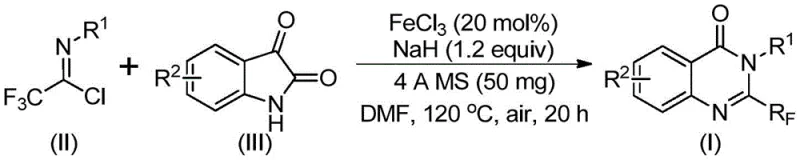
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The core of this synthetic innovation lies in the unique mechanistic pathway facilitated by the iron catalyst, which orchestrates the transformation of linear precursors into the fused quinazolinone ring system. The reaction initiates with the deprotonation of the isatin nitrogen by sodium hydride, generating a nucleophilic species that attacks the imidoyl chloride to form a trifluoroacetamidine intermediate. Subsequently, the ferric chloride coordinates with the carbonyl oxygen, activating the molecule for a crucial decarbonylation step that releases carbon monoxide and drives the cyclization forward. This mechanism is particularly advantageous because it avoids the need for external oxidants or inert atmospheres, allowing the reaction to proceed efficiently in air, which drastically reduces the complexity of the reactor setup and operational overhead for process chemists.
From an impurity control perspective, the use of iron catalysis offers distinct benefits over traditional transition metal methods. Iron salts are generally less prone to forming stable organometallic complexes that persist as difficult-to-remove impurities in the final product. The reaction conditions, specifically the temperature gradient from 40°C to 120°C, are optimized to minimize side reactions such as hydrolysis of the imidoyl chloride or polymerization of the isatin. Furthermore, the inclusion of 4A molecular sieves in the reaction mixture serves to scavenge trace moisture, which is critical for preventing the degradation of the acid chloride functionality. This attention to detail in the reaction design ensures that the resulting high-purity OLED material or pharmaceutical intermediate meets the rigorous quality specifications required for downstream applications without necessitating extensive recrystallization steps.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The practical execution of this synthesis is designed for reproducibility and ease of handling, making it suitable for both laboratory optimization and pilot plant operations. The protocol involves a straightforward addition of reagents into a reaction vessel, followed by a controlled heating profile that maximizes yield while minimizing energy consumption. Operators should note the importance of maintaining the specified molar ratios, particularly the excess of the imidoyl chloride relative to the isatin, to drive the equilibrium towards the desired product. The detailed standardized synthesis steps below outline the precise quantities and conditions required to achieve the reported efficiencies, serving as a foundational guide for process development teams looking to implement this technology.
- Combine ferric chloride (20 mol%), sodium hydride (1.2 equiv), 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin derivative in DMF solvent.
- Stir the reaction mixture at 40°C for approximately 10 hours to initiate alkali-promoted bond formation.
- Heat the mixture to 120°C under air atmosphere for 18-20 hours to complete the iron-catalyzed decarbonylation and cyclization, followed by filtration and chromatographic purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this iron-catalyzed route translates into tangible strategic benefits that extend beyond simple unit cost savings. The shift from precious metals to iron eliminates the supply chain risks associated with geopolitical instability affecting platinum group metal markets. Additionally, the use of commodity chemicals like isatin and simple aromatic amines ensures a stable and diversified source of raw materials, reducing the likelihood of production stoppages due to single-source supplier failures. This resilience is crucial for maintaining continuous manufacturing schedules and meeting the demanding delivery timelines of global pharmaceutical clients who require consistent quality and volume.
- Cost Reduction in Manufacturing: The replacement of expensive catalysts with ferric chloride results in a drastic reduction in direct material costs per kilogram of product. Since iron is abundant and inexpensive, the catalyst cost becomes negligible compared to the organic substrates, allowing for significant margin improvement. Furthermore, the simplified workup procedure reduces the consumption of silica gel and solvents during purification, leading to lower waste disposal fees and reduced operational expenditure on consumables, effectively driving down the total cost of ownership for the manufacturing process.
- Enhanced Supply Chain Reliability: The starting materials, specifically isatin derivatives and trifluoroethylimidoyl chlorides, are widely available from multiple chemical vendors globally. This commoditization of inputs means that procurement teams are not locked into exclusive contracts with niche suppliers, fostering a competitive purchasing environment. The robustness of the reaction conditions also implies that the process is less sensitive to minor variations in raw material quality, further stabilizing the supply chain against fluctuations in feedstock purity and ensuring consistent output quality across different production batches.
- Scalability and Environmental Compliance: The reaction operates under air atmosphere and uses standard organic solvents, eliminating the need for specialized high-pressure equipment or inert gas manifolds that complicate scale-up. From an environmental standpoint, the absence of heavy metals simplifies effluent treatment, as wastewater does not require complex chelation or precipitation steps to meet regulatory discharge limits. This alignment with green chemistry principles facilitates easier permitting for new production lines and supports corporate sustainability goals by minimizing the ecological footprint of the chemical manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing clarity on the operational parameters and expected outcomes. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios or for sourcing teams negotiating supply agreements based on specific process capabilities.
Q: What are the advantages of using FeCl3 over precious metal catalysts for quinazolinone synthesis?
A: Using ferric chloride significantly reduces raw material costs compared to palladium or rhodium catalysts. Furthermore, it eliminates the need for complex heavy metal removal steps required for API compliance, simplifying the downstream purification process and reducing environmental waste.
Q: Is this synthesis method compatible with diverse functional groups on the isatin substrate?
A: Yes, the patent data demonstrates excellent functional group tolerance. The method successfully accommodates substrates with electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as halogens (F, Cl, Br) and nitro groups at various positions on the aromatic ring.
Q: What represents the typical yield range for this iron-catalyzed cyclization?
A: According to the experimental data in patent CN111675662B, the reaction yields are generally high, ranging from approximately 49% to 93% depending on the specific substituents. Electron-deficient substrates often show particularly robust conversion rates under the optimized thermal conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating drug development and commercialization. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries are seamlessly translated into industrial reality. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 2-trifluoromethyl quinazolinone meets the highest international standards for pharmaceutical intermediates, providing our partners with the confidence needed to advance their clinical programs.
We invite you to collaborate with us to leverage this innovative iron-catalyzed technology for your next project. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing expertise can optimize your supply chain and reduce time-to-market for your vital therapeutic candidates.
