Advanced Palladium-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Commercial Scale-Up
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for safer, more efficient, and cost-effective synthetic routes. A significant breakthrough in this domain is documented in Chinese Patent CN115353511A, which discloses a sophisticated multi-component method for synthesizing carbonyl-bridged biheterocyclic compounds. This technology represents a paradigm shift in constructing complex molecular architectures, specifically targeting the fusion of indolinone and imidazole motifs, which are prevalent in numerous bioactive natural products and therapeutic agents. By leveraging a transition metal palladium-catalyzed carbonylation cascade, this invention circumvents the traditional reliance on hazardous gaseous carbon monoxide, instead utilizing a benign in situ generation system. For R&D directors and procurement specialists alike, this development signals a new era of accessible, high-purity heterocyclic building blocks that can be produced with reduced environmental impact and enhanced operational safety profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of biheterocyclic systems containing carbonyl bridges has been fraught with synthetic challenges that hinder efficient commercial production. Traditional methodologies often rely on the direct coupling of two pre-formed heterocyclic substrates, a strategy that frequently suffers from low atom economy and poor regioselectivity, leading to difficult purification processes and diminished overall yields. Alternatively, oxidative cyclization reactions involving bis-nucleophilic substrates and activated methyl-substituted heterocycles typically necessitate harsh reaction conditions, including elevated temperatures and strong oxidants, which can degrade sensitive functional groups and limit the scope of applicable substrates. Furthermore, classical carbonylation approaches invariably require the use of pressurized carbon monoxide gas, introducing severe safety hazards and necessitating specialized high-pressure reactor infrastructure that drastically increases capital expenditure and operational complexity for fine chemical manufacturers.
The Novel Approach
In stark contrast to these legacy techniques, the methodology described in patent CN115353511A introduces a streamlined, one-pot tandem reaction that efficiently assembles the target biheterocyclic framework from simple, commercially available precursors. This innovative route employs trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives as the foundational building blocks, reacting them in the presence of a palladium catalyst and a unique carbon monoxide surrogate system composed of formic acid and acetic anhydride. 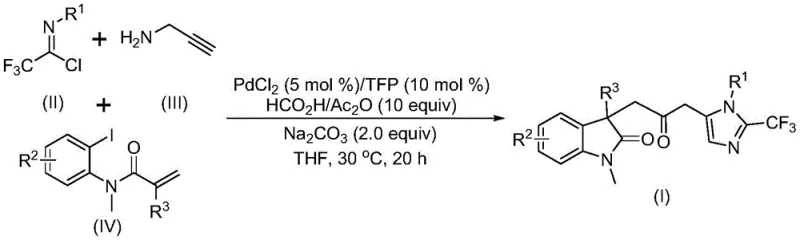 The reaction proceeds under remarkably mild conditions, typically at 30°C, eliminating the thermal stress associated with older methods. This approach not only simplifies the operational workflow by avoiding the handling of toxic gases but also demonstrates exceptional substrate compatibility, allowing for the introduction of diverse functional groups such as halogens, alkyls, and nitro groups without compromising reaction efficiency, thereby offering a versatile platform for the rapid generation of chemical libraries.
The reaction proceeds under remarkably mild conditions, typically at 30°C, eliminating the thermal stress associated with older methods. This approach not only simplifies the operational workflow by avoiding the handling of toxic gases but also demonstrates exceptional substrate compatibility, allowing for the introduction of diverse functional groups such as halogens, alkyls, and nitro groups without compromising reaction efficiency, thereby offering a versatile platform for the rapid generation of chemical libraries.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
The mechanistic elegance of this transformation lies in its intricate catalytic cycle, which orchestrates multiple bond-forming events in a single operation. The process is initiated by the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate, generating a reactive organopalladium intermediate. This species subsequently undergoes an intramolecular Heck-type cyclization to form a five-membered indolinone ring system, establishing the first heterocyclic core. Crucially, the carbon monoxide required for the bridging carbonyl group is generated in situ through the dehydration of formic acid mediated by acetic anhydride, which then inserts into the palladium-carbon bond to form an acyl-palladium complex. This acyl intermediate is then poised to engage with the second heterocyclic component, which is formed concurrently through the base-promoted condensation of trifluoroethylimidoyl chloride and propargylamine to yield a trifluoroacetamidine species.
Following the formation of the trifluoroacetamidine, an isomerization event occurs, rendering the molecule susceptible to nucleophilic attack or insertion by the activated acyl-palladium species. This final cyclization step closes the second ring, forging the imidazole moiety and completing the construction of the carbonyl-bridged biheterocyclic architecture. The use of trifurylphosphine (TFP) as a ligand is instrumental in stabilizing the palladium center throughout this complex sequence, ensuring high turnover numbers and minimizing catalyst deactivation. From a purity perspective, this concerted mechanism minimizes the formation of side products often associated with stepwise syntheses, as the reactive intermediates are consumed immediately upon formation, resulting in a cleaner crude reaction profile that simplifies downstream purification and enhances the overall quality of the final pharmaceutical intermediate.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
Executing this synthesis requires precise control over stoichiometry and reaction parameters to maximize yield and purity. The standard protocol involves charging a reaction vessel with palladium chloride and trifurylphosphine in a molar ratio optimized for catalytic activity, typically around 5 mol% for the metal and 10 mol% for the ligand. To this mixture, sodium carbonate is added as a base to facilitate the deprotonation steps essential for the cyclization events. The carbon monoxide source is introduced as a mixture of formic acid and acetic anhydride, which reacts gently to release CO at the required rate. The substrates—trifluoroethylimidoyl chloride, propargylamine, and the specific acrylamide derivative—are then introduced into an aprotic organic solvent such as tetrahydrofuran (THF), which provides the ideal solubility profile for all components.
- Combine palladium chloride catalyst, trifurylphosphine ligand, sodium carbonate base, and a formic acid/acetic anhydride mixture in an organic solvent such as THF.
- Add the substrates: trifluoroethylimidoyl chloride, propargylamine, and the specific acrylamide derivative to the reaction mixture under stirring.
- Maintain the reaction at 30°C for 12 to 20 hours, then filter, purify via silica gel column chromatography to isolate the final biheterocyclic product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers profound strategic advantages that extend beyond mere chemical novelty. The elimination of toxic carbon monoxide gas from the supply chain removes a significant logistical and safety burden, as there is no longer a need to procure, store, and monitor high-pressure gas cylinders, which are subject to stringent regulatory oversight and transportation restrictions. This shift inherently reduces the insurance premiums and safety compliance costs associated with the manufacturing facility, while simultaneously mitigating the risk of production stoppages due to gas supply disruptions or safety audits. Furthermore, the reliance on liquid reagents that are stable and easy to handle streamlines the raw material intake process, allowing for more flexible inventory management and just-in-time manufacturing strategies.
- Cost Reduction in Manufacturing: The economic implications of this technology are substantial, primarily driven by the use of inexpensive and readily available starting materials such as propargylamine and simple acrylamides, which are commodity chemicals with stable global pricing. By consolidating multiple synthetic steps into a single one-pot operation, the process drastically reduces the consumption of solvents, energy, and labor hours that would otherwise be expended on isolating and purifying intermediates in a stepwise approach. The mild reaction temperature of 30°C further contributes to cost efficiency by lowering the energy demand for heating or cooling, contrasting sharply with energy-intensive high-temperature protocols. Additionally, the high catalytic efficiency means that expensive palladium loading can be kept to a minimum while still achieving high conversion rates, optimizing the cost-per-kilogram of the final active pharmaceutical ingredient precursor.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the broad substrate scope of this reaction, which allows manufacturers to source alternative starting materials if specific suppliers face shortages, without needing to revalidate an entirely new synthetic route. The robustness of the reaction conditions ensures consistent batch-to-batch reproducibility, a critical factor for maintaining long-term contracts with multinational pharmaceutical clients who demand rigorous quality standards. Because the process does not rely on specialized high-pressure equipment, it can be implemented in a wider range of existing manufacturing facilities, increasing the available capacity and reducing the lead time for scaling up production to meet sudden spikes in market demand.
- Scalability and Environmental Compliance: The transition from laboratory scale to commercial production is facilitated by the simplicity of the workup procedure, which involves standard filtration and column chromatography techniques familiar to plant operators. The avoidance of toxic gases aligns perfectly with increasingly stringent environmental, health, and safety (EHS) regulations globally, positioning manufacturers as leaders in green chemistry initiatives. This environmental stewardship not only avoids potential fines and remediation costs but also enhances the brand reputation of the supplier among eco-conscious partners. The ability to scale this reaction to gram levels and beyond without loss of efficiency demonstrates a clear path to multi-ton production, ensuring that the supply of these valuable biheterocyclic intermediates can grow in tandem with the clinical and commercial success of the drugs they support.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the experimental data and background analysis of the patent documentation. These insights are intended to clarify the operational feasibility and strategic value of adopting this method for the production of high-value pharmaceutical intermediates.
Q: What is the primary safety advantage of this carbonylation method compared to traditional protocols?
A: Unlike conventional carbonylation reactions that require handling toxic and hazardous carbon monoxide gas cylinders, this patented method utilizes a formic acid and acetic anhydride mixture to generate carbon monoxide in situ, significantly enhancing operational safety and reducing regulatory compliance burdens.
Q: Does this synthesis route support diverse substrate modifications for drug discovery?
A: Yes, the method demonstrates excellent functional group tolerance, accommodating various substituents on the aryl rings including halogens, alkyl groups, alkoxy groups, and nitro groups, allowing medicinal chemists to rapidly generate diverse libraries of biheterocyclic scaffolds.
Q: Is this process suitable for large-scale industrial manufacturing?
A: The protocol has been validated at the gram scale with high efficiency and uses commercially available, inexpensive starting materials, indicating strong potential for seamless translation to kilogram and multi-ton commercial production without requiring exotic high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the palladium-catalyzed carbonylation cascade described in CN115353511A for the next generation of therapeutic agents. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market-ready supply is seamless and secure. Our state-of-the-art facilities are equipped to handle complex multi-component reactions with the utmost precision, adhering to stringent purity specifications and operating within rigorous QC labs that guarantee every batch meets the highest international standards for pharmaceutical intermediates.
We invite you to leverage our technical expertise to optimize your supply chain for these critical biheterocyclic building blocks. By collaborating with our team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this novel route can improve your bottom line. We encourage you to contact our technical procurement team today to request specific COA data for our catalog items or to discuss route feasibility assessments for your proprietary targets, ensuring a reliable and competitive supply for your global operations.
