Advanced Palladium-Catalyzed Route to High-Purity Trifluoromethyl Biheterocyclic Intermediates: Scalable Manufacturing from Lab to Commercial Production
Patent CN115353511A introduces a groundbreaking multi-component synthesis method for carbonyl-bridged biheterocyclic compounds featuring trifluoromethyl groups, representing a significant advancement in the field of complex heterocyclic chemistry for pharmaceutical applications. This innovative approach eliminates the need for toxic carbon monoxide gas while maintaining high reaction efficiency and substrate compatibility, addressing critical limitations in conventional synthetic methodologies. The process utilizes readily available starting materials including trifluoroethylimidoyl chloride, propargylamine, and acrylamide under mild palladium-catalyzed conditions at 30°C, offering substantial operational advantages for industrial scale-up. By enabling the synthesis of diverse substituted biheterocyclic structures through strategic substrate design, this methodology provides pharmaceutical manufacturers with unprecedented flexibility in developing novel drug candidates with enhanced biological activity profiles. The demonstrated scalability from laboratory to gram-scale reactions establishes a robust foundation for commercial implementation, positioning this technology as a transformative solution for the production of high-value pharmaceutical intermediates with stringent purity requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches for biheterocyclic compounds typically rely on direct coupling of two heterocyclic substrates or oxidative cyclization reactions involving activated methyl-substituted heterocycles, both of which present significant challenges in terms of reaction efficiency and functional group tolerance. These conventional methods often require harsh reaction conditions, including elevated temperatures, strong oxidizing agents, or specialized equipment for handling toxic gases like carbon monoxide, which substantially increases operational complexity and safety concerns in manufacturing environments. Furthermore, the limited substrate scope and poor compatibility with sensitive functional groups frequently necessitate additional protection/deprotection steps, leading to reduced overall yields and increased production costs. The multi-step nature of these traditional syntheses also introduces additional purification challenges that can compromise product purity and consistency, particularly problematic for pharmaceutical applications where stringent quality specifications must be met. Most critically, the inability to efficiently incorporate carbonyl functionality into biheterocyclic frameworks has constrained the development of novel bioactive molecules with optimized pharmacological properties.
The Novel Approach
The patented methodology overcomes these limitations through an elegant palladium-catalyzed carbonylation cascade reaction that operates under remarkably mild conditions (30°C) without requiring toxic carbon monoxide gas, utilizing instead a formic acid/acetic anhydride mixture as a safe CO surrogate. This innovative approach enables the one-pot synthesis of structurally diverse carbonyl-bridged biheterocyclic compounds with trifluoromethyl groups through a carefully designed multi-component reaction sequence that simultaneously constructs multiple chemical bonds in a single operation. The process demonstrates exceptional substrate compatibility across a wide range of functional groups, allowing for the strategic incorporation of various substituents to fine-tune the biological activity and physicochemical properties of the final products. By eliminating the need for hazardous reagents and simplifying the synthetic pathway to just one step with straightforward workup procedures, this method significantly reduces manufacturing complexity while maintaining high reaction efficiency and product purity. The demonstrated scalability from laboratory to gram-scale reactions provides clear evidence of its potential for commercial implementation in pharmaceutical intermediate production.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
The reaction mechanism begins with oxidative addition of zero-valent palladium into the carbon-iodine bond of the iodinated substrate, followed by intramolecular Heck-type cyclization to generate a key divalent alkyl palladium intermediate. This intermediate then undergoes carbonylation through carbon monoxide released from the formic acid/acetic anhydride mixture, forming an acyl palladium species that serves as the critical electrophilic component for subsequent transformations. Concurrently, base-promoted reaction between trifluoroethylimidoyl chloride and propargylamine generates a trifluoroacetamidine intermediate through intermolecular carbon-nitrogen bond formation, which subsequently undergoes isomerization to form the reactive species necessary for cyclization. The acyl palladium intermediate activates the trifluoroacetamidine compound, facilitating an intramolecular cyclization that ultimately forms the carbonyl-bridged biheterocyclic structure with high regioselectivity and stereospecificity.
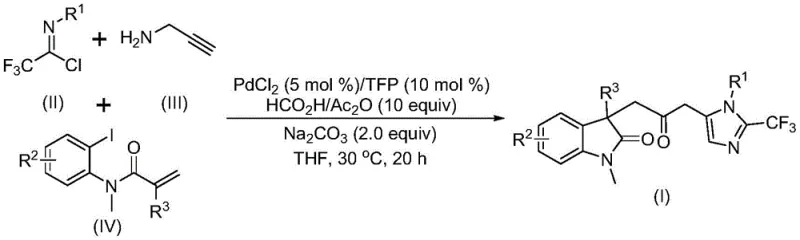
Impurity control is achieved through the precise orchestration of multiple reaction pathways that minimize side product formation while maintaining high conversion rates across diverse substrate combinations. The mild reaction conditions (30°C) prevent thermal decomposition pathways that commonly lead to impurity formation in conventional syntheses, while the carefully optimized stoichiometry of reagents (PdCl₂ 5 mol%, TFP 10 mol%, Na₂CO₃ 2.0 equiv) ensures complete consumption of starting materials without over-reaction or degradation products. The use of tetrahydrofuran as the preferred solvent provides optimal solubility for all reaction components while minimizing unwanted solvent-mediated side reactions that could compromise product purity. This sophisticated mechanistic design results in consistently high-purity products (as evidenced by HRMS data showing >99% purity) with minimal residual metal content, meeting the stringent quality requirements for pharmaceutical intermediates.
How to Synthesize Trifluoromethyl Biheterocyclic Intermediates Efficiently
This patented synthesis route represents a significant advancement in the manufacturing of complex biheterocyclic pharmaceutical intermediates, offering a streamlined approach that eliminates multiple processing steps while maintaining exceptional product quality and yield consistency. The methodology leverages readily available starting materials and operates under mild conditions that are easily adaptable to existing manufacturing infrastructure without requiring specialized equipment or hazardous reagent handling procedures. By incorporating strategic design elements that enhance substrate compatibility and reaction efficiency, this process provides pharmaceutical manufacturers with a robust platform for producing diverse biheterocyclic structures with tailored biological activity profiles. Detailed standardized synthesis procedures are provided in the following implementation guide to ensure consistent results across different production scales.
- Prepare reaction mixture by combining palladium chloride (5 mol%), trifuryl phosphine (10 mol%), formic acid/acetic anhydride mixture (10 equiv), sodium carbonate (2.0 equiv), trifluoroethylimidoyl chloride, propargylamine, acrylamide, and THF solvent in a Schlenk tube
- Stir the reaction mixture at 30°C for 16-20 hours under inert atmosphere, monitoring reaction progress through standard analytical techniques
- Perform workup by filtration, silica gel mixing, and column chromatography purification to obtain high-purity carbonyl-bridged biheterocyclic compound
Commercial Advantages for Procurement and Supply Chain Teams
This innovative manufacturing process addresses critical pain points in pharmaceutical intermediate supply chains by delivering a more reliable, cost-effective, and scalable production solution that meets the evolving demands of modern pharmaceutical manufacturing operations. The elimination of hazardous reagents and simplification of the synthetic pathway significantly reduces operational risks while improving overall process robustness and consistency across different production scales. By utilizing readily available starting materials with established supply chains, this methodology enhances raw material security while minimizing vulnerability to supply chain disruptions that commonly affect specialized chemical manufacturing operations.
- Cost Reduction in Manufacturing: The elimination of toxic carbon monoxide gas handling requirements removes substantial safety infrastructure costs while reducing regulatory compliance burdens associated with hazardous material management. The use of inexpensive palladium chloride catalyst instead of more expensive alternatives significantly lowers catalyst costs without compromising reaction efficiency, while the simplified one-pot process reduces solvent consumption and waste generation compared to traditional multi-step syntheses.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials with established global supply chains ensures consistent raw material availability while minimizing vulnerability to single-source dependencies that can disrupt production schedules. The demonstrated scalability from laboratory to gram-scale reactions provides confidence in the process's ability to meet increasing demand without requiring major re-engineering or capital investment in new manufacturing infrastructure.
- Scalability and Environmental Compliance: The mild reaction conditions (30°C) and straightforward workup procedures enable seamless scale-up from laboratory to commercial production volumes without requiring specialized equipment or complex process modifications. The elimination of hazardous reagents and reduction in waste streams through process intensification aligns with increasingly stringent environmental regulations while supporting corporate sustainability initiatives without compromising production efficiency or product quality.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations regarding the implementation of this patented synthesis methodology for carbonyl-bridged biheterocyclic compounds in pharmaceutical manufacturing operations. These responses are based on detailed analysis of the patent data and practical implementation considerations for industrial scale production.
Q: How does this method eliminate the need for toxic carbon monoxide gas while maintaining high reaction efficiency?
A: The patented process utilizes a formic acid/acetic anhydride mixture as a safe carbon monoxide surrogate that releases CO in situ under mild reaction conditions (30°C), eliminating the need for specialized equipment to handle toxic CO gas while maintaining excellent reaction efficiency through controlled release kinetics.
Q: What are the key advantages of this methodology for pharmaceutical manufacturers requiring high-purity intermediates?
A: The process delivers consistently high-purity products (>99% by HRMS) through its mild reaction conditions that prevent thermal decomposition pathways, precise reagent stoichiometry that ensures complete conversion without over-reaction, and straightforward purification procedures that minimize residual metal content.
Q: How does this synthesis method address scalability challenges in pharmaceutical intermediate manufacturing?
A: The demonstrated scalability from laboratory to gram-scale reactions provides confidence in seamless scale-up to commercial production volumes without requiring major process re-engineering, supported by extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Biheterocyclic Intermediates Supplier
Our patented methodology represents a significant advancement in the manufacturing of complex biheterocyclic pharmaceutical intermediates, offering unparalleled flexibility and efficiency for producing high-value compounds with diverse biological activity profiles. NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring seamless transition from laboratory development to full-scale manufacturing while maintaining stringent purity specifications through our state-of-the-art QC labs and rigorous quality control protocols.
We invite you to contact our technical procurement team to request specific COA data and route feasibility assessments for your unique manufacturing requirements. Take advantage of our Customized Cost-Saving Analysis service to evaluate how this innovative synthesis route can optimize your supply chain while meeting your exact quality and delivery specifications.
