Advanced Mo/Cu Co-Catalyzed Route for Scalable 3-Trifluoromethyl-1,2,4-Triazole Production
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct nitrogen-containing heterocycles, particularly those bearing trifluoromethyl groups which are pivotal for enhancing metabolic stability and lipophilicity in drug candidates. A groundbreaking development in this domain is detailed in Chinese Patent CN113307778A, which discloses a novel preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. This technology represents a significant leap forward, utilizing a synergistic molybdenum and copper co-catalytic system to drive the cycloaddition of trifluoroethylimidoyl chloride with functionalized isonitriles. The relevance of this chemistry is underscored by the prevalence of the 1,2,4-triazole scaffold in blockbuster medications; for instance, Sitagliptin, a widely prescribed antidiabetic agent, relies on similar heterocyclic motifs for its biological activity.
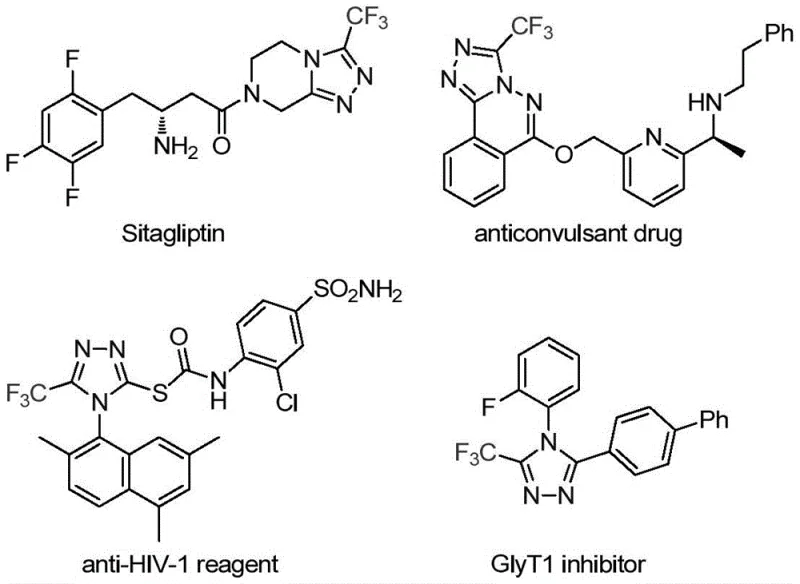
For R&D directors and process chemists, the ability to access these scaffolds through a streamlined, one-pot procedure is invaluable. The patent highlights that this method not only operates under mild conditions but also boasts a broad substrate scope, allowing for the introduction of various functional groups without compromising yield. This flexibility is crucial for medicinal chemists aiming to rapidly generate libraries of analogs for structure-activity relationship (SAR) studies. Furthermore, the use of readily available starting materials positions this technology as a highly practical solution for both early-stage discovery and late-stage process optimization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted 1,2,4-triazoles has been fraught with challenges that hinder efficient manufacturing. Traditional routes often involve the cyclization of trifluoroacetyl hydrazine with amidine compounds, a process that frequently requires harsh reaction conditions and generates significant amounts of waste. Another common approach involves the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinone, which adds unnecessary synthetic steps and reduces overall atom economy. Additionally, methods employing diazonium salts and trifluorodiazoethane pose serious safety concerns due to the explosive nature of diazo compounds, making them less desirable for large-scale operations. These legacy methods often suffer from poor functional group tolerance, limiting the diversity of molecules that can be accessed and increasing the cost of goods for complex API intermediates.
The Novel Approach
In stark contrast, the methodology described in CN113307778A offers a transformative alternative by leveraging a Mo(CO)6 and CuOAc co-catalytic system. This innovative route utilizes trifluoroethylimidoyl chloride and functionalized isonitriles as key building blocks, reacting them in the presence of triethylamine and molecular sieves. The reaction proceeds smoothly at moderate temperatures ranging from 70°C to 90°C, eliminating the need for extreme thermal inputs or cryogenic conditions. As illustrated in the general reaction scheme below, the process efficiently constructs the triazole ring while tolerating a wide array of substituents, including electron-donating and electron-withdrawing groups on the aromatic ring.
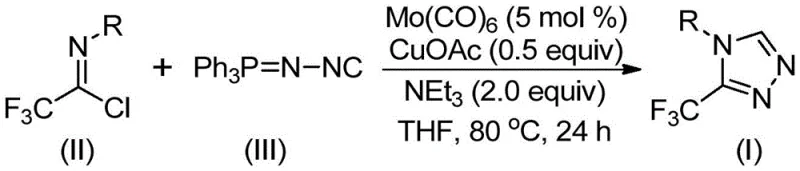
This new approach drastically simplifies the synthetic workflow, reducing the number of unit operations required to reach the final product. The use of stable, non-explosive reagents enhances operational safety, a critical factor for procurement and supply chain teams managing risk in chemical manufacturing. Moreover, the high reaction efficiency reported in the patent examples suggests that this method can significantly reduce raw material consumption and waste generation, aligning with modern green chemistry principles and sustainability goals.
Mechanistic Insights into Mo/Cu Co-Catalyzed Cycloaddition
Understanding the mechanistic underpinnings of this transformation is essential for optimizing the process for commercial scale-up. The reaction is believed to initiate with the activation of the functionalized isonitrile by molybdenum hexacarbonyl, forming a reactive metal-isocyanide complex. This activation lowers the energy barrier for the subsequent nucleophilic attack. Following this, a copper-promoted [3+2] cycloaddition occurs between the activated isonitrile species and the trifluoroethylimidoyl chloride. This step is critical as it forms the five-membered triazole ring intermediate with high regioselectivity. The presence of copper acetate acts as a Lewis acid catalyst, facilitating the cyclization and ensuring that the reaction proceeds with minimal formation of regioisomeric byproducts.
Once the cyclic intermediate is formed, the final step involves the elimination of triphenylphosphine oxide, driven by the presence of water within the reaction system (often introduced via the molecular sieves or ambient moisture control). This elimination step restores aromaticity to the triazole ring, yielding the stable 3-trifluoromethyl-1,2,4-triazole product. The robustness of this mechanism is evidenced by the high yields observed across diverse substrates, such as the 99% yield achieved with 4-methylthio-substituted phenyl groups. For quality control teams, this predictable mechanism implies a cleaner impurity profile, as the pathway avoids the formation of complex polymeric side products often seen in radical-based triazole syntheses.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires precise control over stoichiometry and reaction parameters to maximize yield and purity. The patent outlines a straightforward protocol where the catalysts and reagents are combined in an aprotic solvent, with THF being the preferred medium due to its superior solubilizing properties and reaction promotion capabilities. The molar ratio of reactants is optimized to ensure complete conversion, typically employing a slight excess of the functionalized isonitrile relative to the imidoyl chloride. Detailed standard operating procedures for executing this synthesis, including specific workup and purification techniques, are provided in the technical guide below.
- Combine molybdenum hexacarbonyl, cuprous acetate, triethylamine, molecular sieves, trifluoroethylimidoyl chloride, and functionalized isonitrile in an organic solvent such as THF.
- Heat the reaction mixture to a temperature range of 70°C to 90°C and maintain stirring for a duration of 18 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix the crude sample with silica gel, and purify via column chromatography to isolate the final 3-trifluoromethyl-substituted 1,2,4-triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers compelling advantages that directly address the pain points of cost and supply chain reliability in the pharmaceutical intermediate sector. By shifting away from hazardous diazonium chemistry and multi-step hydrazinolysis routes, manufacturers can achieve substantial cost savings through simplified processing and reduced safety infrastructure requirements. The reliance on commodity chemicals like cuprous acetate and triethylamine, rather than exotic precious metal catalysts, further drives down the raw material costs, making the final API intermediate more price-competitive in the global market.
- Cost Reduction in Manufacturing: The elimination of expensive transition metals and the use of a one-pot reaction sequence significantly lower the operational expenditure associated with production. The mild reaction conditions reduce energy consumption for heating and cooling, while the simplified workup—often requiring only filtration and standard chromatography—minimizes solvent usage and waste disposal costs. This streamlined process translates to a lower cost of goods sold (COGS), providing a strategic margin advantage for generic drug manufacturers and CDMOs.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including various substituted anilines and trifluoroacetic acid derivatives, are widely available from multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions ensures consistent batch-to-batch reproducibility, which is critical for maintaining uninterrupted supply lines for downstream API production. This reliability allows supply chain managers to forecast inventory needs with greater confidence and reduce the safety stock levels required to buffer against production variability.
- Scalability and Environmental Compliance: The method has been demonstrated to be scalable from milligram to gram quantities without loss of efficiency, indicating a clear path toward kilogram and ton-scale production. The avoidance of explosive reagents and the generation of less hazardous waste streams simplify regulatory compliance and environmental permitting processes. This 'green' profile not only reduces the environmental footprint but also future-proofs the manufacturing process against increasingly stringent global environmental regulations.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the viability of this technology for their specific applications, we have compiled answers to common inquiries regarding the reaction scope, safety, and scalability. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, ensuring accuracy and relevance for process development teams.
Q: What are the primary advantages of this Mo/Cu co-catalyzed method over traditional triazole synthesis?
A: Unlike traditional methods that often rely on harsh cyclization conditions or unstable diazonium salts, this novel approach utilizes mild temperatures (70-90°C) and commercially available starting materials. It eliminates the need for complex multi-step sequences, significantly simplifying the purification process and improving overall atom economy.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the patent explicitly demonstrates that the reaction conditions are robust and can be extended from milligram scales to gram-level reactions without significant loss in efficiency. The use of inexpensive catalysts like cuprous acetate and standard solvents like THF makes the process highly amenable to commercial scale-up.
Q: What types of substituents are tolerated in this trifluoromethyl triazole synthesis?
A: The method exhibits excellent substrate tolerance, accommodating a wide range of functional groups on the aryl ring, including alkyl, alkoxy, halogen, and nitro groups. This versatility allows for the synthesis of diverse derivatives crucial for SAR studies in drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of next-generation therapeutics. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the bench to the plant. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 3-trifluoromethyl-1,2,4-triazole meets the highest industry standards, supporting your regulatory filings and clinical trial timelines with unwavering consistency.
We invite you to collaborate with us to leverage this advanced synthetic technology for your upcoming projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. Please contact us today to request specific COA data and comprehensive route feasibility assessments, and let us demonstrate how our commitment to innovation can drive value and efficiency in your supply chain.
