Advanced Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for heterocyclic scaffolds that serve as core structures in bioactive molecules. Patent CN114920707B introduces a groundbreaking preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds, addressing critical bottlenecks in current manufacturing processes. This innovation leverages the ubiquitous solvent N,N-dimethylformamide (DMF) not merely as a medium but as a vital carbon source, facilitating a tandem cyclization reaction promoted by molecular iodine. The significance of this chemical transformation cannot be overstated, as trifluoromethyl-substituted triazoles are pivotal motifs found in numerous high-value therapeutic agents, including antiviral drugs, kinase inhibitors, and metabolic regulators. By streamlining the synthesis through the dual functionality of DMF, this technology offers a pathway to enhance the efficiency of producing complex pharmaceutical intermediates while simultaneously reducing the reliance on specialized reagents.
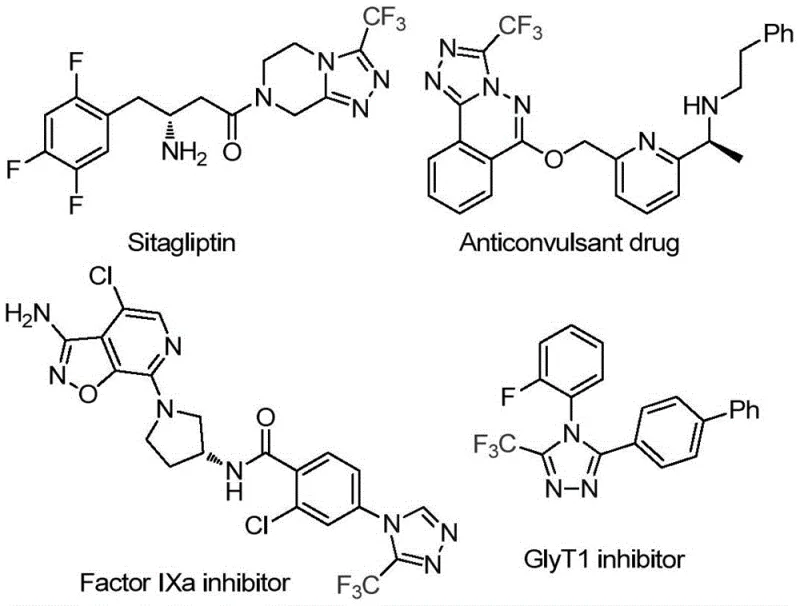
For R&D directors and process chemists, the structural versatility demonstrated in this patent is particularly compelling. The ability to incorporate diverse functional groups onto the triazole ring allows for the rapid generation of analog libraries essential for structure-activity relationship (SAR) studies. As illustrated in the provided documentation, compounds such as Sitagliptin and various Factor IXa inhibitors rely on similar heterocyclic cores, underscoring the commercial relevance of mastering this specific chemical architecture. The method described in CN114920707B provides a reliable foundation for synthesizing these high-purity pharmaceutical intermediates, ensuring that downstream drug development programs are supported by a consistent and high-quality supply of key building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing trifluoromethyl-substituted 1,2,4-triazoles often suffer from significant operational complexities and economic inefficiencies that hinder large-scale production. Conventional methodologies frequently necessitate the use of expensive transition metal catalysts, stringent anhydrous conditions, and multi-step sequences that accumulate impurities and reduce overall yield. Furthermore, many existing protocols require specialized carbon sources or hazardous reagents that pose safety risks and increase waste disposal costs, creating substantial barriers for cost reduction in API manufacturing. The reliance on inert atmospheres and moisture-sensitive reagents also demands sophisticated reactor infrastructure, which limits the flexibility of contract development and manufacturing organizations (CDMOs) when scaling up from laboratory to commercial production volumes.
The Novel Approach
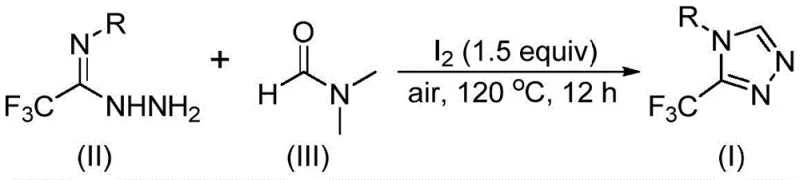
In stark contrast, the novel approach detailed in the patent utilizes a remarkably simple yet effective strategy where DMF acts as both the solvent and the reactant, fundamentally altering the economics of the synthesis. By employing molecular iodine as a promoter under aerobic conditions, the reaction proceeds smoothly at temperatures between 110°C and 130°C without the need for exotic catalysts or protective atmospheres. This iodine-promoted tandem cyclization enables the direct conversion of trifluoroethyliminohydrazide derivatives into the desired triazole products with high efficiency. The elimination of complex pre-functionalization steps and the use of commodity chemicals like DMF drastically simplify the process flow, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The mechanistic pathway of this transformation reveals a sophisticated interplay between the iodine promoter and the DMF solvent, offering deep insights into how simple reagents can achieve complex bond formations. In this catalytic cycle, molecular iodine activates the DMF molecule, facilitating the cleavage of either the formyl group or the N-methyl group to serve as the carbon source for the triazole ring construction. When the formyl group participates, it undergoes condensation with the hydrazide to form a hydrazone intermediate, which subsequently cyclizes with the elimination of dimethylamine. Alternatively, the N-methyl pathway involves the formation of an amine salt followed by nucleophilic addition and oxidative aromatization. Understanding these dual pathways is crucial for optimizing reaction parameters and ensuring high purity specifications in the final product.
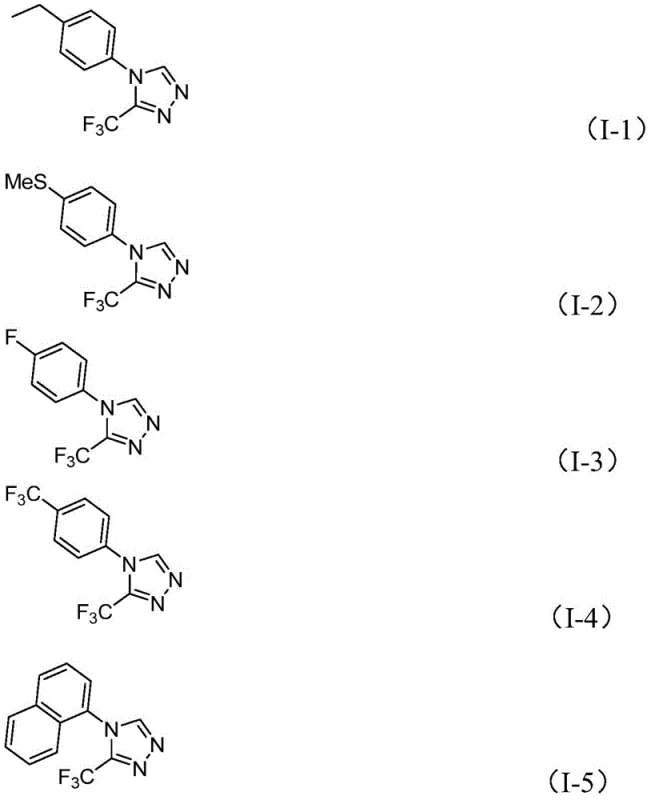
From an impurity control perspective, the simplicity of the reaction mechanism translates directly into a cleaner crude profile, which is a major advantage for downstream processing. The absence of heavy metal catalysts eliminates the need for costly and time-consuming metal scavenging steps, which are often required to meet strict regulatory limits for residual metals in active pharmaceutical ingredients. Furthermore, the reaction conditions are mild enough to tolerate a wide range of functional groups on the aromatic ring, including electron-withdrawing and electron-donating substituents, without causing decomposition or side reactions. This robustness ensures that the impurity profile remains manageable even when synthesizing diverse analogs, thereby supporting the rigorous quality control standards demanded by global regulatory agencies.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazoles Efficiently
The practical implementation of this synthesis route is designed for ease of execution, allowing chemical manufacturers to adopt the technology with minimal retooling or training. The process begins with the straightforward mixing of readily available starting materials in a standard reaction vessel, followed by heating under ambient air pressure. Detailed standard operating procedures regarding stoichiometry, temperature ramping, and workup protocols are essential for maximizing yield and reproducibility. For teams looking to implement this chemistry immediately, the following guide outlines the critical operational parameters derived directly from the patent data to ensure successful batch production.
- Combine molecular iodine, trifluoroethyliminohydrazide, and organic solvent DMF in a reaction vessel under an air atmosphere.
- Heat the reaction mixture to a temperature range of 110-130°C and maintain stirring for 10-15 hours to ensure complete conversion.
- Upon completion, perform post-treatment including filtration, washing, drying, and column chromatography purification to isolate the target triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic methodology presents a compelling value proposition centered on cost stability and operational resilience. By replacing expensive, specialized reagents with commodity solvents like DMF, the raw material cost structure is significantly optimized, leading to substantial cost savings over the lifecycle of the product. The elimination of noble metal catalysts not only reduces direct material costs but also mitigates the supply chain risks associated with the volatility of precious metal markets. Additionally, the simplified workflow reduces the consumption of utilities and labor hours per kilogram of product, further enhancing the overall economic efficiency of the manufacturing process.
- Cost Reduction in Manufacturing: The strategic use of DMF as a dual-purpose reagent eliminates the need for purchasing separate carbon donors, which are often costly and logistically challenging to source in bulk quantities. This consolidation of reagents simplifies inventory management and reduces the total number of SKUs required for production, leading to streamlined procurement operations. Moreover, the avoidance of expensive transition metal catalysts removes the financial burden of metal recovery and waste treatment, contributing to a leaner and more cost-effective production model that improves margin potential for high-volume contracts.
- Enhanced Supply Chain Reliability: The reliance on widely available, commodity-grade chemicals such as iodine and DMF ensures a robust supply chain that is less susceptible to disruptions compared to processes dependent on niche or imported specialty reagents. Since these materials are produced globally in massive quantities, the risk of shortage is minimized, guaranteeing continuous production schedules and reliable delivery timelines for clients. This stability is critical for maintaining long-term partnerships with pharmaceutical companies that require uninterrupted supply of key intermediates to support their own clinical and commercial manufacturing timelines.
- Scalability and Environmental Compliance: The operational simplicity of running the reaction under air without stringent moisture control makes this process highly scalable from pilot plant to multi-ton commercial production. The reduced generation of hazardous waste, particularly the absence of heavy metal residues, simplifies environmental compliance and lowers the costs associated with effluent treatment and disposal. This alignment with green chemistry principles not only satisfies regulatory requirements but also enhances the corporate sustainability profile, which is increasingly important for securing contracts with environmentally conscious multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These answers are derived from the specific experimental data and beneficial effects reported in the patent documentation, providing clarity on the feasibility and advantages of the method. Understanding these details helps stakeholders make informed decisions about integrating this chemistry into their existing manufacturing portfolios.
Q: What is the primary advantage of using DMF in this synthesis?
A: DMF serves a dual role as both the reaction solvent and the carbon source (C1 synthon), eliminating the need for separate, expensive carbon donors and simplifying the reaction system significantly.
Q: Does this process require strict anhydrous or anaerobic conditions?
A: No, the method operates effectively under an air atmosphere without the need for rigorous anhydrous or oxygen-free environments, greatly reducing operational complexity and equipment costs.
Q: What types of substituents are tolerated on the aromatic ring?
A: The methodology demonstrates broad substrate scope, accommodating various substituents such as alkyl, alkoxy, alkylthio, halogens, and trifluoromethyl groups at ortho, meta, or para positions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iodine-promoted synthesis for producing high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-trifluoromethyl-1,2,4-triazole delivered meets the highest international standards for quality and consistency.
We invite you to collaborate with our technical team to explore how this efficient synthetic route can optimize your supply chain and reduce overall production costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can accelerate your drug development timeline and secure your supply of critical intermediates.
