Advanced Synthetic Route for High-Purity Brivaracetam Intermediates and Commercial Scalability
The pharmaceutical landscape for third-generation antiepileptic drugs continues to evolve, with Brivaracetam standing out as a high-affinity ligand for SV2A. However, the commercial viability of such potent APIs often hinges on the efficiency of their synthetic routes. Patent CN112154140A introduces a groundbreaking approach to synthesizing Brivaracetam bulk drugs via a novel compound of Formula III. This technology addresses critical bottlenecks in existing manufacturing processes by utilizing cheap, easily obtainable raw materials to achieve high optical purity without relying on complex chiral resolution or complicated separation steps. For industry leaders, this represents a significant shift towards more sustainable and cost-effective production methodologies for neurological therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Brivaracetam has been plagued by inefficiencies that drive up costs and complicate supply chains. As illustrated in prior art such as CN1882535A, traditional routes often result in a mixture of the target drug and its diastereoisomer, necessitating separation via chiral solid-phase chromatography using expensive eluents like n-hexane and ethanol. This reliance on chiral HPLC not only limits throughput but also imposes high equipment demands and operational costs. Furthermore, alternative routes disclosed in literature, such as those involving Benoit M., have shown crude product purities ranging only from 83% to 88%, requiring extensive silica gel column separation which is impractical for large-scale industrial application.
Additionally, other known pathways, such as those described in WO2007065634A1, depend on expensive transition metal catalysts like RuCl3, introducing potential heavy metal contamination risks and necessitating rigorous removal steps to meet regulatory standards. Many intermediates in these legacy processes are liquids with weak ultraviolet absorption, making detection and quality control during production exceptionally difficult. These factors collectively create a fragile supply chain vulnerable to delays and quality deviations, highlighting the urgent need for a more robust synthetic strategy.
The Novel Approach
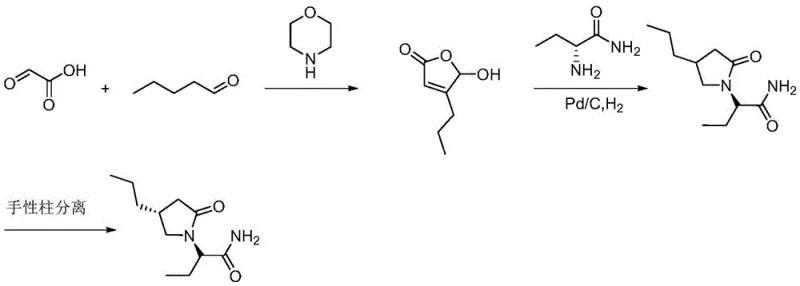
The innovative methodology presented in CN112154140A circumvents these historical challenges through a cleverly designed sequence that prioritizes stereocontrol and ease of purification. The core of this advancement lies in the preparation of a specific compound of Formula III, which serves as a pivotal intermediate. By reacting a chiral sulfonyl-substituted lactone with an optically pure aminobutanamide derivative, the process constructs the pyrrolidinone scaffold with high fidelity. Unlike previous methods that struggle with liquid intermediates, this route generates solid intermediates that can be effectively purified through simple filtration and washing procedures, drastically reducing the need for complex distillation or chromatographic equipment.
This novel approach effectively eliminates the requirement for chiral chromatography in the final stages by embedding the necessary stereochemical information early in the synthesis. The use of accessible reagents like L-2-aminobutanamide hydrochloride and common bases such as sodium methoxide ensures that the raw material supply is stable and cost-effective. Consequently, this pathway offers a streamlined alternative that is inherently more suitable for the commercial scale-up of complex pharmaceutical intermediates, providing a clear competitive advantage in terms of both operational simplicity and economic efficiency.
Mechanistic Insights into Sulfonyl-Pyrrolidinone Cyclization
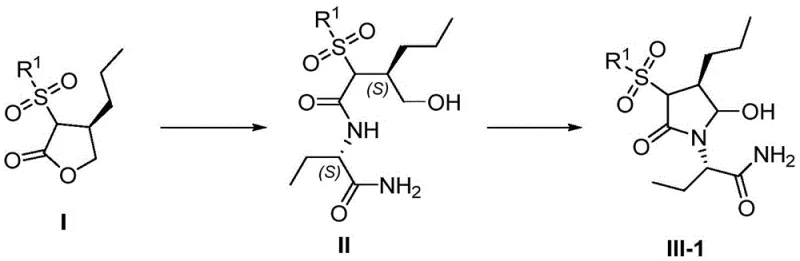
The chemical elegance of this synthesis is rooted in the precise manipulation of the pyrrolidinone ring formation. The process begins with the nucleophilic ring-opening of the chiral dihydrofuran-2-one scaffold by the amino group of the butanamide derivative. This step is critical as it establishes the carbon-nitrogen bond while preserving the stereochemical integrity of the propyl side chain. Following this amidation, the linear intermediate undergoes an oxidative cyclization. Utilizing oxidant systems such as TEMPO/NaOCl or Dess-Martin periodinane, the primary alcohol moiety is oxidized, triggering an intramolecular condensation that closes the five-membered ring. This transformation is highly selective, favoring the formation of the desired (3S, 4S) configuration essential for biological activity.
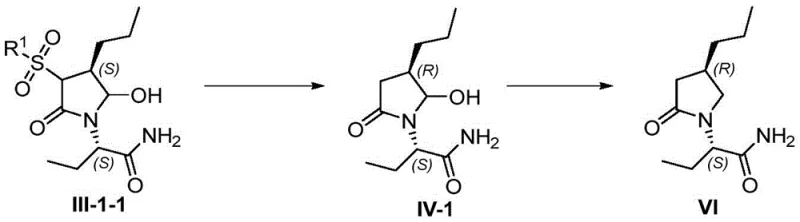
Impurity control is meticulously managed through the choice of the sulfonyl protecting group. The sulfonyl moiety not only activates the lactone for ring-opening but also serves as a robust handle for subsequent de-protection. In the final conversion to Brivaracetam (Formula VI), the sulfonyl group and the hydroxyl group are removed via reductive conditions, often employing magnesium turnings or silane reagents in the presence of acids like trifluoroacetic acid. This de-protection strategy is clean and efficient, avoiding the generation of complex byproduct profiles that typically accompany harsh acidic or basic hydrolysis. The result is a final API with a superior impurity profile, meeting the stringent requirements of global pharmacopeias.
How to Synthesize Brivaracetam Intermediate Efficiently
Implementing this synthesis requires careful attention to reaction conditions to maximize yield and purity. The initial amidation is typically conducted in alcoholic solvents like methanol or ethanol under reflux, ensuring complete conversion of the lactone. The subsequent oxidation step must be monitored closely, with temperature control being paramount to prevent over-oxidation. For the final de-protection, the choice of reducing agent—whether magnesium-based or silane-based—depends on the specific scale and available infrastructure, but both offer viable paths to the target molecule. Detailed standardized operating procedures for each step are essential to maintain consistency across batches.
- React (4S)-4-propyl-3-sulfonyldihydrofuran-2-one with (S)-2-aminobutanamide in the presence of a base like sodium methoxide to form the open-chain amide intermediate.
- Perform oxidative cyclization using a TEMPO-based oxidant system or Dess-Martin reagent to close the pyrrolidinone ring and generate the hydroxy-keto intermediate.
- Execute a reductive de-protection step using magnesium turnings or silane reagents to remove the sulfonyl and hydroxyl groups, yielding the final Brivaracetam API.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route translates into tangible strategic benefits. The primary value proposition lies in the drastic simplification of the manufacturing workflow. By replacing liquid intermediates that require vacuum distillation with solid intermediates amenable to filtration, the process reduces the dependency on specialized glass-lined reactors and high-vacuum systems. This shift not only lowers capital expenditure for new production lines but also enhances the flexibility of existing facilities to switch between different campaigns without extensive cleaning or reconfiguration.
- Cost Reduction in Manufacturing: The elimination of expensive chiral chromatography columns and precious metal catalysts like ruthenium results in substantial cost savings. Traditional methods incur high operational expenses due to the consumption of chiral stationary phases and the energy-intensive nature of recycling solvents from HPLC processes. In contrast, this novel route relies on stoichiometric reagents that are commodity chemicals, driving down the cost of goods sold (COGS) significantly. Furthermore, the higher yields observed in the crystallization steps reduce raw material waste, contributing to a leaner and more economical production model.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved as the key starting materials, such as substituted dihydrofuranones and aminobutanamides, are readily available from multiple global suppliers. The avoidance of proprietary or hard-to-source catalysts mitigates the risk of supply disruptions. Additionally, the robustness of the solid-state purification steps ensures that minor fluctuations in raw material quality do not compromise the final product, leading to more predictable lead times and consistent delivery schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental perspective, this route offers a greener profile by reducing solvent usage associated with chromatographic separations. The ability to perform purifications via slurrying and filtration minimizes the volume of hazardous waste generated, simplifying compliance with increasingly strict environmental regulations. The process is inherently scalable, having been designed with industrial production in mind, allowing for seamless transition from pilot plant quantities to multi-ton commercial output without the need for process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Brivaracetam synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy routes in terms of purity, cost, and operational feasibility.
Q: How does this new route improve upon conventional Brivaracetam synthesis?
A: Unlike prior art methods that rely on expensive ruthenium catalysts or inefficient chiral chromatography for diastereomer separation, this novel route utilizes readily available chiral starting materials and simple crystallization or slurry purification steps, significantly lowering production costs and equipment requirements.
Q: What are the key advantages for supply chain stability?
A: The process avoids liquid intermediates that require difficult vacuum distillation and instead produces solid intermediates that can be purified by filtration and washing. This simplifies quality control and allows for robust scaling from kilogram to multi-ton production without specialized distillation infrastructure.
Q: Is the stereochemical purity maintained throughout the synthesis?
A: Yes, the method leverages the inherent chirality of the starting lactone and the amino acid derivative to control stereocenters at the 3 and 4 positions of the pyrrolidinone ring, ensuring high optical purity without the need for late-stage chiral resolution columns.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Brivaracetam Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the competitive pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical advantages of this patent are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs equipped to handle the specific analytical challenges of chiral intermediates, guaranteeing that every batch meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to optimize your Brivaracetam supply chain. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can drive value and reliability for your organization.
