Advanced Chalcogen Catalysis for Scalable Pyrrolidine Skeleton Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct nitrogen-containing heterocycles, which serve as critical scaffolds in drug discovery. Patent CN112608269B discloses a groundbreaking method for the catalytic synthesis of pyrrolidine skeletons utilizing chalcogen weak interactions. This technology represents a significant paradigm shift from traditional Lewis acid catalysis to a more sophisticated sulfur-bonding activation mode. By employing specialized sulfur bond catalysts, such as those derived from 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene (XP) and phenylselenium chloride, the process enables the cycloaddition of aziridine derivatives with olefin compounds under remarkably mild conditions. This innovation not only simplifies the synthetic route but also enhances the feasibility of producing high-purity pharmaceutical intermediates on a commercial scale, addressing long-standing challenges in organic synthesis regarding reaction severity and catalyst stability.
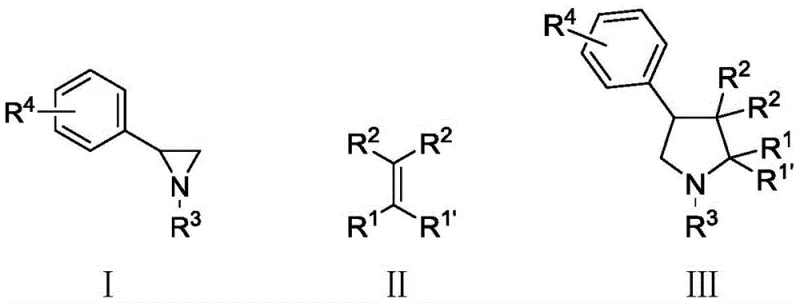
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of pyrrolidine rings from aziridine precursors has relied heavily on aggressive activation strategies that pose significant operational and economic burdens. For instance, prior art methodologies often necessitated the use of stoichiometric amounts of strong Lewis acids like boron trifluoride diethyl etherate (BF3·OEt2), which required cryogenic reaction temperatures as low as -78°C to control selectivity and prevent decomposition. Such extreme conditions demand specialized refrigeration equipment and result in substantial energy costs, making large-scale production economically unviable. Furthermore, alternative approaches utilizing Metal-Organic Frameworks (MOFs) often operated at elevated temperatures around 100°C and required complex metal ligand systems, introducing risks of heavy metal contamination that are strictly regulated in pharmaceutical manufacturing. These conventional limitations create bottlenecks in supply chain reliability and increase the overall cost of goods sold for nitrogen heterocycle intermediates.
The Novel Approach
In stark contrast, the novel chalcogen weak interaction catalysis described in the patent offers a streamlined solution that operates under significantly milder thermal conditions, typically ranging from 0°C to 95°C, with optimal performance observed at 50°C. This method eliminates the need for cryogenic cooling or excessive heating, thereby drastically reducing energy consumption and simplifying reactor requirements. The use of a sulfur bond catalyst facilitates the generation of a stable dipolar transition state from the aziridine derivative, allowing it to react efficiently with unactivated electron-rich olefins in a single step. This one-step cycloaddition process not only shortens the synthetic timeline but also minimizes the formation of by-products, leading to cleaner reaction profiles. The operational simplicity and mild conditions make this approach highly attractive for industrial adoption, providing a robust pathway for the cost reduction in pharmaceutical intermediate manufacturing while maintaining high standards of product quality.
Mechanistic Insights into Chalcogen Weak Interaction Catalysis
The core innovation of this technology lies in the unique activation mechanism driven by chalcogen bonding, specifically involving selenium-phosphorus interactions within the catalyst structure. The sulfur bond catalyst, such as XP-Se BArF4, functions by interacting with the nitrogen atom of the aziridine ring, effectively polarizing the C-N bond and lowering the activation energy for ring opening. This interaction generates a highly reactive yet stable dipolar transition state that is electron-deficient, enabling it to undergo [3+2] cycloaddition with a wide variety of olefin partners without the need for additional activators. The catalyst design incorporates bulky phosphine ligands and non-coordinating anions like BArF4-, which stabilize the cationic selenium species and prevent catalyst deactivation. This precise electronic tuning ensures that the catalytic cycle proceeds efficiently, allowing for the transformation of diverse substrates including those with sensitive functional groups that might degrade under harsher Lewis acidic conditions.
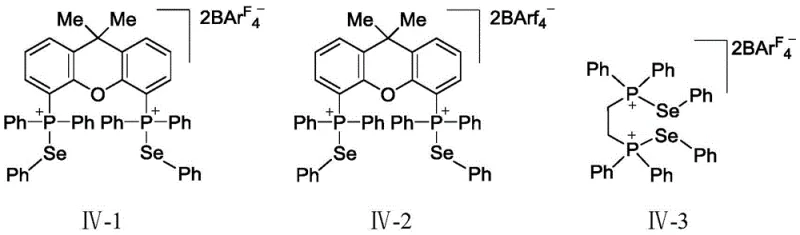
Impurity control is inherently superior in this system due to the high selectivity of the chalcogen interaction. Unlike traditional metal catalysts that may promote side reactions such as polymerization of the olefin or over-alkylation, the sulfur bond catalyst directs the reaction specifically towards the desired pyrrolidine skeleton. The stability of the catalyst throughout the reaction course, as confirmed by 31P NMR spectroscopy showing no significant signal changes before and after the reaction, indicates that the active species does not decompose into reactive fragments that could generate complex impurity profiles. This stability is crucial for downstream processing, as it reduces the burden on purification steps like column chromatography. Consequently, the final product exhibits a cleaner impurity spectrum, which is a critical quality attribute for R&D directors focusing on regulatory compliance and process robustness in API intermediate production.
How to Synthesize Pyrrolidine Derivatives Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible pathway for generating diverse pyrrolidine derivatives suitable for drug development. The process begins with the preparation of the sulfur bond catalyst, followed by the cycloaddition reaction in a common organic solvent like 1,2-dichloroethane (DCE). The reaction is typically conducted under an inert argon atmosphere to prevent moisture interference, with reactants mixed at room temperature before heating to the optimal range. This methodology supports a broad substrate scope, accommodating various substituents on both the aziridine and olefin components, which allows for the rapid generation of chemical libraries. For detailed operational parameters and specific stoichiometric ratios required to achieve optimal yields, please refer to the standardized synthesis guide below.
- Prepare the sulfur bond catalyst (e.g., XP-Se BArF4) by reacting PhSeCl with TMSOTf and a phosphine ligand, followed by anion exchange with NaBArF4.
- In an inert atmosphere, mix the aziridine derivative and the prepared catalyst in a solvent such as 1,2-dichloroethane (DCE).
- Add the olefin compound dropwise and heat the mixture to 50°C for approximately 1 hour, then purify the resulting pyrrolidine via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this chalcogen catalysis technology offers transformative benefits that directly impact the bottom line and operational resilience. The elimination of extreme temperature requirements removes the dependency on specialized cryogenic infrastructure, allowing production to occur in standard reactors available in most multipurpose chemical facilities. This flexibility significantly enhances supply chain reliability by reducing the risk of production delays associated with equipment maintenance or availability. Furthermore, the simplified workup procedure, which involves basic solvent removal and chromatography, streamlines the manufacturing workflow, leading to faster turnaround times for batch production. These operational efficiencies translate into substantial cost savings and improved agility in responding to market demands for high-value nitrogen heterocycles.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the drastic reduction in energy consumption and equipment overhead. By avoiding the need for -78°C cooling baths or high-temperature MOF synthesis, the utility costs per kilogram of product are significantly lowered. Additionally, the catalyst system does not rely on expensive transition metals like palladium or rhodium, which are subject to volatile market pricing and strict residual limits in pharmaceutical products. The use of organoselenium-based catalysts, which can be synthesized from readily available precursors, provides a more stable and predictable cost structure. This economic advantage allows for competitive pricing strategies while maintaining healthy margins, making it an ideal choice for cost-sensitive large-scale manufacturing campaigns.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes directly to supply continuity. Since the reagents and solvents used, such as DCE and common olefins, are commodity chemicals with established global supply chains, the risk of raw material shortages is minimized. The catalyst itself is stable and can be prepared in advance, ensuring that production schedules are not disrupted by catalyst preparation delays. Moreover, the tolerance of the method to various functional groups means that a single platform technology can be used to produce a wide array of intermediates, reducing the need for multiple specialized production lines. This versatility strengthens the supply chain by consolidating production capabilities and reducing the complexity of inventory management for diverse chemical portfolios.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the mild reaction conditions and the absence of hazardous heavy metals. The lack of toxic metal residues simplifies waste treatment protocols and reduces the environmental footprint of the manufacturing process, aligning with increasingly stringent global environmental regulations. The one-step nature of the synthesis minimizes solvent usage and waste generation compared to multi-step traditional routes. This green chemistry profile not only ensures compliance with environmental standards but also enhances the corporate sustainability image, which is becoming a key factor in supplier selection for major pharmaceutical companies committed to responsible sourcing and eco-friendly manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chalcogen weak interaction catalysis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical application and advantages of the method for potential partners and stakeholders.
Q: What are the advantages of chalcogen weak interaction catalysis over traditional Lewis acid methods?
A: Unlike traditional methods requiring harsh conditions like -78°C with BF3·OEt2 or high temperatures with MOFs, this method operates under mild conditions (e.g., 50°C) with simpler operation and shorter reaction steps, reducing energy consumption and equipment complexity.
Q: Is the sulfur bond catalyst stable during the reaction process?
A: Yes, comparative experiments utilizing 31P NMR spectroscopy confirm that the sulfur bond catalyst remains chemically stable throughout the reaction cycle, showing no significant signal changes before and after the process, which ensures consistent catalytic performance.
Q: What types of substrates are compatible with this pyrrolidine synthesis method?
A: The method demonstrates broad substrate scope, accommodating various aziridine derivatives with different substituents (e.g., phenyl, halogen, alkyl groups) and diverse olefin compounds, including cyclic and acyclic alkenes, yielding pyrrolidine skeletons with high structural diversity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrrolidine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the chalcogen-catalyzed pyrrolidine synthesis in accelerating drug discovery and development. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are seamlessly translated into robust industrial operations. Our facility is equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of pyrrolidine intermediate meets the highest quality standards required by global regulatory bodies. We are committed to leveraging this cutting-edge technology to deliver superior products that enhance your R&D efficiency.
We invite you to collaborate with us to explore the full potential of this synthesis route for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this method can optimize your budget without compromising quality. Please contact us to request specific COA data for our available pyrrolidine derivatives and to discuss route feasibility assessments for your target molecules. Let us be your partner in achieving scientific excellence and commercial success through innovative chemical manufacturing.
