Advanced Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for Scalable Pharmaceutical Manufacturing
Advanced Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for Scalable Pharmaceutical Manufacturing
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable pathways to access nitrogen-containing heterocycles, particularly those functionalized with trifluoromethyl groups which enhance metabolic stability and lipophilicity. Patent CN110467579B discloses a groundbreaking preparation method for 5-trifluoromethyl substituted 1,2,4-triazole compounds that addresses many of the historical bottlenecks in heterocyclic synthesis. This technology leverages a non-metallic iodine-promoted cyclization strategy, offering a distinct advantage over traditional transition-metal catalyzed routes. For R&D directors and procurement managers alike, this represents a significant opportunity to streamline the supply chain for critical pharmaceutical intermediates. The method utilizes inexpensive starting materials such as hydrazones and trifluoroethylimidoyl chloride, bypassing the need for complex, moisture-sensitive reagents often associated with trifluoromethylation chemistry.
The structural versatility of the 1,2,4-triazole scaffold is well-documented, serving as a core motif in numerous bioactive molecules ranging from antifungal agents to kinase inhibitors. As illustrated in the reference data, compounds like deferasirox and various CYP enzyme inhibitors rely on similar heterocyclic architectures to exert their biological effects. 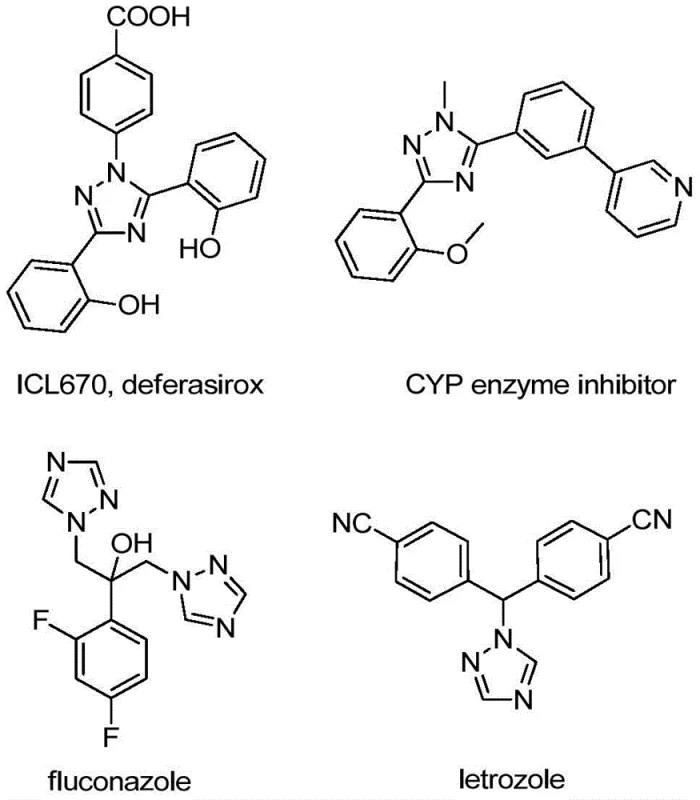 Introducing a trifluoromethyl group at the 5-position of this ring system further amplifies the pharmacological profile by modulating electronic properties and improving binding affinity. Consequently, developing a reliable, cost-effective synthesis for these specific derivatives is of paramount importance for any organization aiming to secure a competitive edge in API manufacturing. The disclosed method not only simplifies the synthetic route but also ensures high purity profiles essential for downstream drug development.
Introducing a trifluoromethyl group at the 5-position of this ring system further amplifies the pharmacological profile by modulating electronic properties and improving binding affinity. Consequently, developing a reliable, cost-effective synthesis for these specific derivatives is of paramount importance for any organization aiming to secure a competitive edge in API manufacturing. The disclosed method not only simplifies the synthetic route but also ensures high purity profiles essential for downstream drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted nitrogen heterocycles has been plagued by significant operational challenges and economic inefficiencies. Conventional strategies typically fall into two categories: the direct trifluoromethylation of pre-synthesized heterocycles or the cyclocondensation of trifluoromethyl-containing synthons. The former often necessitates the use of specialized, expensive trifluoromethylating reagents such as Togni reagents or Umemoto reagents, which drastically inflate the raw material costs. Furthermore, these reactions frequently require stringent anhydrous and anaerobic conditions, demanding specialized equipment and increasing the complexity of scale-up operations. The latter approach, while more direct, has traditionally relied on less versatile synthons like trifluorodiazoethane, which poses safety hazards due to its explosive nature, or trifluoroethylimide acid halides which have seen limited application due to perceived low reactivity or selectivity issues.
The Novel Approach
The methodology described in patent CN110467579B fundamentally shifts the paradigm by utilizing a simple, iodine-promoted cyclization between readily available hydrazones and trifluoroethylimidoyl chlorides. This novel approach eliminates the dependency on precious metal catalysts and hazardous diazo compounds, replacing them with benign elemental iodine and stable imidoyl chlorides. The reaction proceeds efficiently in common organic solvents like dichloroethane (DCE) at moderate temperatures (80°C), without the need for inert atmosphere protection. 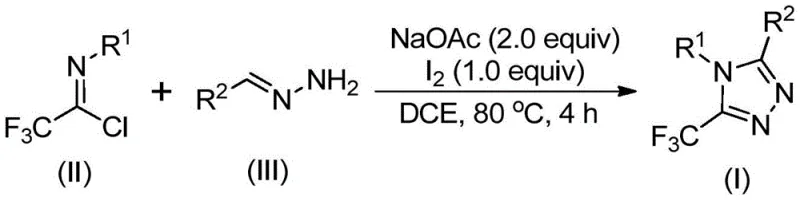 This operational simplicity translates directly into cost reduction in pharmaceutical intermediate manufacturing, as it removes the need for expensive catalyst removal steps (such as scavenger resins) and reduces energy consumption associated with cryogenic or high-pressure conditions. The broad substrate tolerance allows for the synthesis of diverse derivatives, including those with electron-donating or withdrawing groups on the aromatic rings, making it a versatile platform for library synthesis.
This operational simplicity translates directly into cost reduction in pharmaceutical intermediate manufacturing, as it removes the need for expensive catalyst removal steps (such as scavenger resins) and reduces energy consumption associated with cryogenic or high-pressure conditions. The broad substrate tolerance allows for the synthesis of diverse derivatives, including those with electron-donating or withdrawing groups on the aromatic rings, making it a versatile platform for library synthesis.
Mechanistic Insights into Iodine-Promoted Cyclization
From a mechanistic perspective, this transformation is a fascinating example of oxidative heterocyclization driven by a halogen promoter. The reaction likely initiates with a base-promoted intermolecular carbon-nitrogen bond formation between the hydrazone nitrogen and the imidoyl chloride carbon, generating a trifluoroacetamidine intermediate. This step is facilitated by sodium acetate, which acts as a mild base to neutralize the HCl byproduct. Following this initial condensation, the system undergoes an isomerization to align the reactive centers for cyclization. The critical role of elemental iodine becomes apparent in the subsequent oxidative iodination step, where iodine activates the intermediate, possibly forming an iodonium species or facilitating an electrophilic attack. This activation lowers the energy barrier for the final intramolecular electrophilic substitution and aromatization, driving the formation of the stable 1,2,4-triazole ring. Understanding this mechanism is crucial for process chemists aiming to optimize reaction parameters for commercial scale-up of complex heterocycles.
Furthermore, the choice of reagents plays a pivotal role in controlling the impurity profile of the final product. The use of sodium acetate rather than stronger bases minimizes side reactions such as hydrolysis of the imidoyl chloride or decomposition of the hydrazone. The stoichiometric control, specifically maintaining a molar ratio of trifluoroethylimidoyl chloride to hydrazone of roughly 1:2, ensures that the hydrazone is in excess, driving the equilibrium towards the desired product and suppressing the formation of symmetric byproducts. The subsequent addition of iodine after the initial heating period allows the condensation to reach completion before the oxidative cyclization begins, preventing premature oxidation of the starting materials. This sequential addition strategy is a key process parameter that ensures high yields, often exceeding 80% for a wide range of substrates, thereby enhancing the overall atom economy of the process.
How to Synthesize 5-Trifluoromethyl-1,2,4-Triazole Efficiently
The synthesis protocol outlined in the patent provides a clear, reproducible pathway for generating high-purity triazole derivatives suitable for medicinal chemistry campaigns. The procedure involves a straightforward one-pot sequence where reagents are mixed in a solvent like DCE, heated to promote condensation, and then treated with iodine to effect cyclization. This simplicity makes it an ideal candidate for technology transfer from the laboratory to the pilot plant. For detailed operational specifics regarding reagent grades, quenching procedures, and purification techniques, please refer to the standardized synthesis guide below.
- Mix sodium acetate, trifluoroethylimidoyl chloride, and hydrazone in an organic solvent like DCE.
- Heat the mixture to 80°C and react for 2 to 4 hours to form the intermediate.
- Add elemental iodine to the system and continue reacting for 1 to 2 hours, followed by purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the raw material portfolio. By relying on commodity chemicals like hydrazines, aldehydes (for hydrazone prep), and trifluoroacetic acid derivatives, the supply chain becomes far more resilient to market fluctuations compared to routes dependent on proprietary or scarce fluorinating agents. The elimination of heavy metal catalysts not only reduces the cost of goods sold (COGS) by removing the catalyst line item but also significantly de-risks the regulatory filing process, as residual metal limits are a common cause of batch rejection in GMP manufacturing. This leads to substantial cost savings and improved batch consistency.
- Cost Reduction in Manufacturing: The economic impact of this method is profound due to the replacement of expensive transition metal catalysts with inexpensive elemental iodine. Traditional cross-coupling or C-H activation methods often require palladium, copper, or rhodium complexes, which are not only costly to purchase but also require sophisticated recovery systems to meet strict ppm limits in the final API. By switching to an iodine-promoted system, manufacturers can eliminate these capital and operational expenditures entirely. Additionally, the reaction operates at atmospheric pressure and moderate temperatures (80°C), reducing the energy load on reactor systems compared to high-pressure autoclave processes. The simplified workup, involving basic filtration and column chromatography, further reduces labor and solvent costs, contributing to a leaner, more cost-efficient production model.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on single-source suppliers for exotic reagents. This methodology mitigates that risk by utilizing starting materials that are widely available from multiple global chemical vendors. Hydrazones and imidoyl chlorides are standard building blocks in organic synthesis, ensuring that procurement teams can negotiate better pricing and secure long-term contracts without fear of supply disruption. Moreover, the robustness of the reaction conditions—specifically the lack of requirement for anhydrous or oxygen-free environments—means that the process is less sensitive to variations in utility quality or operator error. This inherent stability translates to higher first-pass yields and fewer failed batches, ensuring a steady flow of materials to downstream formulation units.
- Scalability and Environmental Compliance: As the industry moves towards greener chemistry, the environmental footprint of a synthesis route is a critical decision factor. This iodine-promoted cyclization avoids the generation of heavy metal waste streams, which are classified as hazardous and require expensive disposal protocols. The use of dichloroethane (DCE) is noted, but the process efficiency allows for effective solvent recovery and recycling, minimizing overall waste volume. The ability to scale this reaction from gram-scale laboratory experiments to multi-kilogram pilot runs without significant re-optimization demonstrates its maturity for industrial application. This scalability ensures that as demand for the final API grows, the intermediate supply can be ramped up quickly to meet market needs without requiring new process development cycles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this 5-trifluoromethyl-1,2,4-triazole synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for your evaluation of this manufacturing route. Understanding these details is essential for assessing the fit of this technology within your existing production infrastructure.
Q: Does this synthesis require expensive heavy metal catalysts?
A: No, the patented method utilizes elemental iodine as a promoter, completely avoiding the need for toxic and expensive transition metal catalysts, which simplifies purification and reduces costs.
Q: What are the optimal reaction conditions for this triazole formation?
A: The reaction is optimally conducted in dichloroethane (DCE) at 80°C, using a molar ratio of trifluoroethylimidoyl chloride to hydrazone of approximately 1:2, ensuring high conversion rates.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the process does not require strict anhydrous or anaerobic conditions and uses cheap, commercially available starting materials, making it highly scalable for industrial applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to full-scale manufacturing is seamless. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of novel triazole derivatives or bulk supply of established intermediates, our facility is designed to handle complex fluorinated chemistry with the utmost safety and efficiency.
We invite you to collaborate with us to leverage this innovative iodine-promoted synthesis for your next project. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our advanced manufacturing capabilities can accelerate your timeline to market while optimizing your overall production costs.
