Advanced CuCl-Catalyzed Synthesis of Asymmetric Azoxybenzenes for Commercial Scale-Up
The landscape of fine chemical manufacturing is constantly evolving, driven by the urgent need for greener, more cost-effective synthetic routes for high-value intermediates. A pivotal advancement in this domain is detailed in patent CN107353233B, which discloses a robust method for the catalytic synthesis of asymmetric azoxybenzene compounds. These compounds serve as critical building blocks in the production of liquid crystals for electronic displays, agrochemicals, and various pharmaceutical active ingredients. The patented technology leverages a composite catalytic system comprising Cuprous Chloride (CuCl) and an auxiliary base, operating under mild aerobic conditions to achieve high selectivity and yield. For global procurement teams and R&D directors, this represents a significant opportunity to optimize supply chains for complex aromatic intermediates while adhering to stricter environmental regulations.
Traditionally, the synthesis of azoxybenzene derivatives has been fraught with significant technical and economic challenges that hinder large-scale commercial adoption. Conventional methodologies often rely on the reduction of aromatic nitro compounds or the oxidation of aromatic amines using stoichiometric amounts of hazardous reagents. Historical processes frequently employ heavy metal salts such as lead, mercury, or manganese, which pose severe environmental risks and require costly waste treatment protocols. Furthermore, alternative oxidative systems utilizing tert-butyl hydroperoxide (TBHP) or noble metal catalysts like gold or silver on supports introduce prohibitive raw material costs and safety concerns regarding peroxide handling. These legacy methods often suffer from poor atom economy and generate substantial toxic byproducts, making them increasingly untenable for modern sustainable manufacturing frameworks.
In stark contrast, the novel approach outlined in the patent utilizes a synergistic CuCl-pyridine catalytic system that fundamentally alters the reaction economics and safety profile. By employing molecular oxygen (O2) as the terminal oxidant, the process eliminates the need for expensive and dangerous stoichiometric oxidants. The use of earth-abundant copper, specifically in the +1 oxidation state, combined with a simple nitrogenous ligand like pyridine, creates a highly active catalytic species capable of facilitating dehydrogenative condensation under mild temperatures ranging from 55°C to 85°C. This shift not only drastically reduces the cost of goods sold (COGS) by replacing precious metals with commodity chemicals but also simplifies the reactor setup, allowing for safer operation at near-atmospheric pressures. The result is a streamlined process that delivers high-purity asymmetric azoxybenzenes with excellent functional group tolerance.
Mechanistic Insights into CuCl-Catalyzed Aerobic Oxidative Coupling
The efficacy of this synthetic route lies in the precise interplay between the copper center and the auxiliary ligand, which governs the activation of molecular oxygen and the subsequent coupling of the amine and nitroso partners. Mechanistically, the CuCl species, coordinated by pyridine, likely facilitates the formation of a reactive copper-oxygen intermediate that mediates the hydrogen abstraction from the aromatic amine. This generates a transient nitrogen-centered radical or a copper-amido species that rapidly couples with the electrophilic nitrosobenzene. The presence of the pyridine ligand is critical; comparative data indicates that without this specific auxiliary, or when substituted with structurally similar bases like triethylamine, the reaction efficiency drops precipitously. This suggests that the ligand stabilizes the active copper oxidation state and prevents the formation of inactive copper aggregates, ensuring a sustained catalytic cycle throughout the reaction duration.
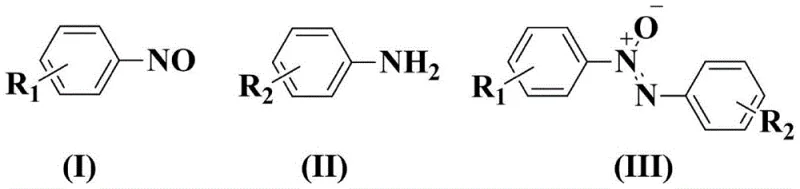
Furthermore, the chemoselectivity of this system is a paramount advantage for R&D teams targeting complex molecules. The mild oxidative conditions prevent the over-oxidation of sensitive functional groups often present in pharmaceutical intermediates, such as sulfides or electron-rich aromatics, which might be degraded by harsher oxidants like peracetic acid. The reaction proceeds through a controlled dehydrogenation pathway that selectively forms the N-N(O) bond while leaving other moieties intact. This high level of control minimizes the formation of side products like azo compounds or hydrazobenzenes, thereby simplifying the downstream purification process. The ability to tolerate diverse substituents, including halogens and esters, as demonstrated in the specific examples, underscores the versatility of this mechanism for generating a broad library of analogues required for drug discovery and material science applications.
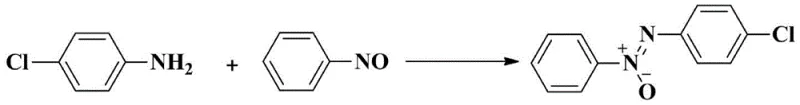
How to Synthesize Asymmetric Azoxybenzene Efficiently
The operational simplicity of this patented method makes it highly attractive for technology transfer from laboratory to pilot plant scales. The procedure involves charging a reaction vessel with the aromatic amine and nitrosobenzene substrates in a suitable organic solvent such as toluene, followed by the addition of the CuCl catalyst and pyridine auxiliary. The system is then pressurized with oxygen and heated to moderate temperatures, typically around 65°C, for a period of 24 to 48 hours. Upon completion, the workup is straightforward, involving concentration and standard silica gel chromatography, avoiding complex aqueous quenching steps often associated with metal-mediated reactions. For detailed standard operating procedures and specific molar ratios optimized for different substrates, please refer to the technical guide below.
- Charge a reaction vessel with aromatic amine, nitrosobenzene, CuCl catalyst (3mol%), pyridine auxiliary, and toluene solvent.
- Purge the system with Oxygen (approx. 1 bar) and heat the mixture to 65°C with stirring for 24 hours.
- Upon completion, concentrate the mixture, adsorb onto silica gel, and purify via column chromatography using petroleum ether/ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this CuCl-catalyzed methodology offers profound advantages in terms of cost stability and supply chain resilience. The primary driver for cost reduction is the substitution of expensive noble metal catalysts and hazardous stoichiometric oxidants with inexpensive, commodity-grade copper salts and atmospheric oxygen. This fundamental change in the bill of materials (BOM) leads to substantial cost savings in raw material procurement, which is particularly impactful when manufacturing at multi-ton scales. Additionally, the elimination of toxic heavy metal waste streams significantly lowers the operational expenditures related to environmental compliance and waste disposal, further enhancing the overall economic viability of the process for commercial partners.
- Cost Reduction in Manufacturing: The transition from precious metal catalysts like gold or palladium to earth-abundant copper chloride represents a drastic decrease in catalyst loading costs. Since copper is widely available and priced significantly lower than noble metals, the direct material cost for the catalytic system is minimized. Moreover, the use of oxygen from air as the oxidant removes the recurring expense of purchasing specialized chemical oxidants like TBHP or hypervalent iodine reagents. This combination of low-cost catalyst and free oxidant creates a highly efficient economic model that allows for competitive pricing of the final asymmetric azoxybenzene intermediates in the global market.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials ensures a robust and uninterrupted supply chain. Aromatic amines and nitrosobenzenes are bulk chemicals produced by numerous suppliers worldwide, reducing the risk of single-source dependency. The catalyst components, CuCl and pyridine, are also standard industrial chemicals with stable global supply networks. This accessibility means that production schedules are less likely to be disrupted by raw material shortages, providing procurement managers with greater certainty in delivery timelines and inventory planning for long-term projects.
- Scalability and Environmental Compliance: The mild reaction conditions, operating at temperatures below 100°C and near-atmospheric pressure, facilitate easy scale-up in standard glass-lined or stainless steel reactors without requiring specialized high-pressure equipment. This lowers the capital expenditure barrier for manufacturing partners. Furthermore, the green chemistry profile of the process, characterized by the absence of toxic heavy metals and the generation of water as the primary byproduct of oxidation, aligns perfectly with increasingly stringent global environmental regulations. This compliance reduces the regulatory burden and potential liabilities associated with hazardous waste management, making the process sustainable for long-term industrial application.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic technology. These insights are derived directly from the experimental data and comparative studies presented in the patent documentation, providing clarity on the scope and limitations of the method for potential licensees and manufacturing partners.
Q: Why is CuCl preferred over other copper salts for this transformation?
A: According to patent data, CuCl demonstrates superior catalytic activity compared to CuSO4, CuO, or CuBr, likely due to optimal solubility and coordination geometry with the pyridine ligand under aerobic conditions.
Q: What is the substrate scope for this asymmetric azoxybenzene synthesis?
A: The method tolerates a wide range of substituents including halogens (F, Cl, Br, I), alkyl groups, alkoxy groups, and esters, making it highly versatile for diverse pharmaceutical intermediate production.
Q: How does this method improve environmental compliance?
A: By utilizing molecular oxygen as the terminal oxidant and avoiding stoichiometric heavy metal oxidants like lead or mercury salts, the process significantly reduces toxic waste generation and simplifies downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Asymmetric Azoxybenzene Supplier
The technological breakthroughs encapsulated in patent CN107353233B highlight the immense potential of copper-catalyzed aerobic oxidation for producing high-value fine chemicals. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our facility is equipped with rigorous QC labs and advanced analytical capabilities to meet stringent purity specifications required by the pharmaceutical and electronic materials sectors. We are committed to delivering high-purity asymmetric azoxybenzene intermediates that adhere to the highest quality standards, supporting your R&D and commercial manufacturing needs with reliability and precision.
We invite you to collaborate with us to leverage this efficient synthetic route for your specific project requirements. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating how this CuCl-catalyzed method can optimize your production budget. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your custom synthesis projects. Let us partner together to drive innovation and efficiency in your supply chain.
