Revolutionizing Asymmetric Azoxybenzene Production: A Strategic Analysis of Silver Oxide Catalysis for Global Supply Chains
Introduction to Breakthrough Asymmetric Synthesis Technology
The landscape of fine chemical manufacturing is constantly evolving, driven by the need for more efficient and selective synthetic routes for complex intermediates. A pivotal advancement in this domain is documented in patent CN108689890B, which discloses a robust method for synthesizing asymmetric azoxybenzene compounds. This technology addresses a longstanding challenge in organic synthesis: the selective formation of non-symmetric azoxy structures without generating excessive symmetric byproducts. By leveraging silver oxide as a unique promoter, this process facilitates a dehydrogenative condensation between aromatic amines and nitrosobenzenes. For R&D directors and procurement strategists, this represents a significant opportunity to access high-purity functional materials essential for pharmaceuticals, liquid crystals, and dye industries. The methodology not only simplifies the operational workflow but also aligns with modern green chemistry principles by utilizing commercially available reagents and enabling catalyst recovery.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of azoxybenzene derivatives has relied heavily on two primary pathways: the oxidative coupling of aromatic amines or the reductive coupling of arylnitro compounds. While these methods are well-established, they suffer from inherent structural limitations, predominantly yielding symmetric azoxybenzenes where both aromatic rings are identical. Achieving asymmetry through these traditional routes often requires multi-step sequences, such as the oxidation of asymmetric azobenzenes or the coupling of Grignard reagents with nitrobenzenes. These conventional approaches are plagued by harsh reaction conditions, poor selectivity, and the use of hazardous reagents like hydrazine hydrate or heavy metal oxidants. Furthermore, the separation of symmetric byproducts from the desired asymmetric target is often cumbersome, leading to reduced overall yields and increased waste generation, which poses significant challenges for scalable industrial production and environmental compliance.
The Novel Approach
In stark contrast, the novel approach detailed in the patent introduces a direct cross-coupling strategy that bypasses the symmetry constraints of prior art. By reacting an aromatic amine directly with a nitrosobenzene derivative in the presence of silver oxide, the system achieves high selectivity for the asymmetric product. This method eliminates the need for pre-functionalized intermediates or dangerous reducing agents. The use of silver oxide acts as a mild yet effective promoter that drives the dehydrogenation process efficiently. This shift in synthetic logic allows for the modular assembly of diverse azoxybenzene scaffolds simply by varying the amine and nitroso components. For supply chain managers, this modularity translates to greater flexibility in sourcing raw materials, as a wide range of substituted anilines and nitrosobenzenes are commodity chemicals. The streamlined nature of this single-step condensation significantly reduces processing time and complexity compared to the multi-stage protocols of the past.
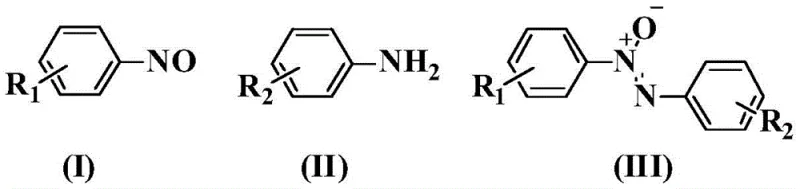
Mechanistic Insights into Ag2O-Promoted Dehydrogenative Condensation
The core of this technological breakthrough lies in the specific role of silver oxide (Ag2O) within the reaction matrix. Unlike traditional transition metal catalysts that might require complex ligand systems, Ag2O functions as a sole promoter to facilitate the removal of hydrogen atoms during the condensation of the amine nitrogen and the nitroso nitrogen. The mechanism likely involves the activation of the N-H bond of the aromatic amine and the subsequent nucleophilic attack on the electrophilic nitrogen of the nitrosobenzene. The silver species assists in the electron transfer processes required to form the N-N bond while simultaneously managing the oxidation state changes necessary to establish the N-oxide functionality. This delicate balance prevents over-oxidation to azo compounds or reduction to hydrazines, ensuring the stability of the azoxy linkage. Understanding this mechanistic pathway is crucial for R&D teams aiming to optimize reaction parameters for specific substrates, as the electronic properties of the substituents on the aromatic rings can influence the rate of condensation and the final yield.
Furthermore, the impurity profile of this reaction is notably cleaner than that of oxidative coupling methods. In traditional peroxide-mediated oxidations, radical side reactions often lead to a complex mixture of azo, hydrazo, and polymeric byproducts that are difficult to separate. The Ag2O-promoted pathway appears to proceed through a more controlled ionic or coordinated mechanism, minimizing radical generation. This results in a crude reaction mixture that is easier to purify, often requiring only standard silica gel chromatography to achieve high purity levels suitable for pharmaceutical applications. The ability to control the regioselectivity and chemoselectivity of the reaction ensures that sensitive functional groups, such as esters or halides, remain intact during the synthesis. This tolerance expands the scope of accessible derivatives, allowing chemists to introduce diverse pharmacophores or material-properties-enhancing groups without protecting group strategies.
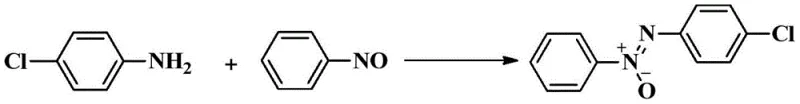
How to Synthesize Asymmetric Azoxybenzene Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize efficiency and safety. The process is designed to be straightforward, utilizing standard glassware and heating equipment without the need for specialized high-pressure reactors. The key to success lies in the precise stoichiometry of the silver oxide promoter and the selection of an appropriate polar aprotic solvent. Detailed standard operating procedures regarding mixing rates, temperature ramping, and work-up protocols are critical for reproducibility. For technical teams looking to adopt this methodology, the following guide outlines the fundamental steps derived from the patent examples to ensure consistent production of high-quality asymmetric azoxybenzene intermediates.
- Prepare the reaction mixture by combining an aromatic amine and a nitrosobenzene derivative in an organic solvent such as DMSO.
- Add silver oxide (Ag2O) as the sole promoter to the reaction vessel, ensuring a molar ratio between 50mol% and 150mol% relative to the substrate.
- Heat the mixture to a temperature between 25°C and 100°C for 24 to 48 hours, then purify the resulting product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this silver oxide-mediated synthesis offers substantial strategic benefits for procurement and supply chain operations. The primary advantage stems from the use of readily available and cost-effective starting materials. Aromatic amines and nitrosobenzenes are produced on a large scale globally, ensuring a stable supply base that is less susceptible to the volatility seen with exotic catalysts or specialized reagents. This accessibility reduces the risk of supply chain disruptions and allows for better long-term planning of raw material inventory. Additionally, the reaction conditions are mild, typically operating between 25°C and 100°C, which significantly lowers energy consumption compared to high-temperature or high-pressure processes. This energy efficiency contributes directly to cost reduction in fine chemical manufacturing, making the final product more competitive in price-sensitive markets.
- Cost Reduction in Manufacturing: The economic viability of this process is enhanced by the recyclability of the silver component. Post-reaction, the silver oxide is converted into elemental silver, which can be recovered and potentially reused or sold, offsetting the initial cost of the promoter. This contrasts sharply with processes using stoichiometric amounts of non-recoverable oxidants. Furthermore, the elimination of complex purification steps reduces solvent usage and waste disposal costs. By simplifying the downstream processing, manufacturers can achieve higher throughput with existing infrastructure, effectively lowering the cost per kilogram of the active intermediate without compromising on quality standards.
- Enhanced Supply Chain Reliability: The robustness of the reaction across a wide range of substrates ensures that supply chains remain flexible. Whether the target molecule requires electron-withdrawing groups like chlorides or electron-donating groups like methoxy, the core synthetic protocol remains largely unchanged. This universality means that procurement teams do not need to qualify entirely new processes for different product variants, streamlining the vendor qualification process. The reliance on commodity chemicals also means that multiple suppliers can be sourced for raw materials, mitigating the risk of single-source dependency and enhancing the overall resilience of the supply network against market fluctuations.
- Scalability and Environmental Compliance: Scaling this reaction from gram to tonnage levels is facilitated by its operational simplicity and safety profile. The absence of explosive reagents like hydrazine or high-pressure hydrogen gas removes significant safety barriers often encountered during scale-up. Moreover, the environmental footprint is minimized due to the atom economy of the condensation reaction and the potential for silver recovery. This aligns with increasingly stringent global environmental regulations, reducing the regulatory burden on manufacturing sites. Companies adopting this technology can market their products as sustainably produced, adding value for end-clients in the pharmaceutical and agrochemical sectors who are prioritizing green supply chains.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology, we have compiled answers to common inquiries regarding the synthesis and application of these asymmetric azoxybenzene compounds. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation. Understanding these nuances is essential for assessing the feasibility of integrating this method into existing production lines or R&D pipelines. The following questions address critical aspects of catalyst performance, substrate scope, and product quality assurance.
Q: What distinguishes this silver oxide method from traditional oxidative coupling?
A: Unlike traditional oxidative coupling of arylamines which predominantly yields symmetric azoxybenzenes, this method utilizes a cross-condensation strategy between distinct aromatic amines and nitrosobenzenes, enabling the precise construction of asymmetric structures with high selectivity.
Q: Is the silver promoter recyclable in this process?
A: Yes, a significant advantage of this protocol is that the silver oxide promoter is converted into elemental silver post-reaction, which can be recovered and recycled, thereby reducing heavy metal waste and lowering long-term material costs.
Q: What are the typical reaction conditions required for this synthesis?
A: The reaction operates under remarkably mild conditions, typically requiring temperatures between 25°C and 100°C in common organic solvents like DMSO or acetonitrile, eliminating the need for extreme pressure or hazardous oxidants like hydrazine.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Asymmetric Azoxybenzene Supplier
NINGBO INNO PHARMCHEM stands at the forefront of implementing advanced synthetic methodologies like the Ag2O-promoted condensation for commercial production. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of asymmetric azoxybenzene meets the exacting standards required for pharmaceutical and electronic material applications. Our commitment to quality ensures that the impurity profiles are tightly controlled, providing our partners with reliable intermediates that facilitate smoother downstream synthesis.
We invite global partners to collaborate with us to leverage this cutting-edge technology for their specific project needs. By engaging with our technical procurement team, clients can request a Customized Cost-Saving Analysis tailored to their volume requirements and target specifications. We encourage you to reach out for specific COA data and route feasibility assessments to determine how this efficient synthesis method can optimize your supply chain and reduce overall manufacturing costs. Let us be your trusted partner in delivering high-performance chemical solutions.
