Advanced Rhodium-Catalyzed Synthesis of Indolo[2,1-a]isoquinoline Intermediates for Commercial Scale-Up
Introduction to Next-Generation Heterocycle Synthesis
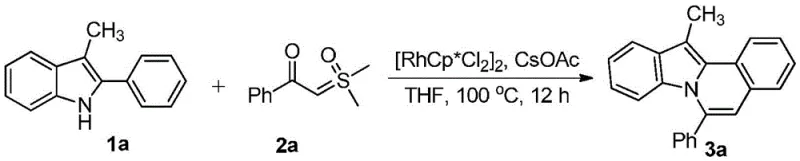
The development of efficient synthetic routes for complex nitrogen-containing fused heterocycles remains a cornerstone challenge in modern medicinal chemistry. Patent CN108640917B introduces a transformative methodology for constructing the indolo[2,1-a]isoquinoline scaffold, a privileged structure found in numerous bioactive natural products and pharmaceutical candidates. This innovation leverages a rhodium(III)-catalyzed tandem reaction between readily available 2-aryl-3-alkylindoles and sulfur ylides, bypassing the multi-step sequences typically required for such tetracyclic systems. By enabling a direct one-pot annulation, this technology addresses critical bottlenecks in process chemistry, offering a streamlined pathway that aligns with the principles of green chemistry and sustainable manufacturing.
![General reaction scheme for Rh-catalyzed synthesis of indolo[2,1-a]isoquinolines from 2-aryl-3-alkylindoles and sulfur ylides](/insights/img/indolo-isoquinoline-synthesis-rh-catalysis-pharma-supplier-20260302221947-01.webp)
The strategic value of this approach lies in its operational simplicity and robust substrate tolerance. Unlike traditional methods that often demand cryogenic temperatures or highly sensitive organometallic reagents, this protocol operates under relatively mild thermal conditions ranging from 80°C to 140°C in common solvents like tetrahydrofuran. For R&D teams focused on rapid library generation, the ability to access diverse derivatives through simple substitution patterns on the indole or the sulfur ylide component represents a significant acceleration in lead optimization cycles. Furthermore, the use of air-stable catalyst precursors simplifies the handling requirements, reducing the barrier to entry for laboratories without specialized inert atmosphere infrastructure.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the indolo[2,1-a]isoquinoline core has relied on classical cyclization strategies that are fraught with inefficiencies. Traditional routes often involve the preparation of highly functionalized precursors, such as halogenated indoles or activated alkynes, which require multiple synthetic steps to prepare. These multi-step sequences inevitably lead to cumulative yield losses and generate substantial quantities of chemical waste, driving up the cost of goods sold (COGS) for the final intermediate. Additionally, many conventional methods employ harsh reaction conditions, including strong acids or bases and elevated temperatures exceeding 150°C, which can compromise the integrity of sensitive functional groups often present in drug-like molecules. The necessity for rigorous purification between steps further exacerbates the timeline and resource consumption, making these legacy processes ill-suited for the agile demands of modern pharmaceutical supply chains.
The Novel Approach
In stark contrast, the methodology disclosed in CN108640917B utilizes a direct C-H functionalization strategy that fundamentally reshapes the synthetic logic. By employing a cationic rhodium(III) species generated in situ from [RhCp*Cl2]2 and cesium acetate, the reaction activates the inert C-H bond at the C2 position of the indole ring directly. This activation allows for the immediate insertion of the sulfur ylide, followed by a cascade of elimination and cyclization events that forge the new C-C and C-N bonds in a single operational step. This "one-pot" philosophy not only drastically reduces the number of unit operations but also minimizes the exposure of reactive intermediates to degradation. The result is a process that delivers the target tetracyclic framework with high atom economy, transforming simple, commodity-grade starting materials into high-value scaffolds with minimal environmental footprint.
Mechanistic Insights into Rhodium(III)-Catalyzed C-H Activation
The success of this transformation hinges on the unique reactivity profile of the pentamethylcyclopentadienyl rhodium(III) catalyst. The mechanism initiates with the coordination of the indole nitrogen to the rhodium center, directing the metal to the proximal C-H bond on the aryl ring or the indole core, depending on the specific substrate geometry. This directed metallation forms a stable five-membered rhodacycle intermediate, a key species that lowers the activation energy for subsequent bond formation. The sulfur ylide then acts as a specialized two-carbon synthon; upon coordination to the metal center, it undergoes migratory insertion into the Rh-C bond. This step is critical as it establishes the carbon skeleton of the new ring system while retaining the necessary functionality for the final closure.
Following the insertion event, the elimination of the dimethyl sulfoxide (DMSO) or sulfide moiety drives the reaction forward thermodynamically, regenerating the aromaticity of the system and releasing the active catalyst for the next turnover. From an impurity control perspective, this mechanism is highly advantageous. The specificity of the C-H activation step ensures that side reactions, such as non-directed polymerization of the ylide or decomposition of the indole, are kinetically suppressed. The mild basicity of the cesium acetate additive further aids in maintaining a clean reaction profile by neutralizing acidic byproducts without promoting hydrolysis of sensitive ester or amide groups that might be present on the substrate. This precise control over the reaction trajectory is essential for achieving the high purity levels required for GMP manufacturing of pharmaceutical intermediates.
How to Synthesize Indolo[2,1-a]isoquinoline Derivatives Efficiently
To implement this technology effectively, process chemists should adhere to the optimized parameters established in the patent examples. The standard protocol involves charging a reaction vessel with the 2-aryl-3-alkylindole substrate and the sulfur ylide in a molar ratio of approximately 1:1.5 to ensure complete conversion of the limiting indole reagent. Tetrahydrofuran (THF) is identified as the preferred solvent due to its ability to solubilize both organic substrates and the ionic catalyst additives while maintaining stability at reflux temperatures. The addition of the catalyst system, specifically the rhodium dimer and cesium acetate, should be performed sequentially to ensure proper formation of the active cationic species before heating commences.
- Dissolve the 2-aryl-3-alkylindole substrate and sulfur ylide reagent in tetrahydrofuran (THF) within a sealed reaction vessel.
- Add the rhodium catalyst dimer [RhCp*Cl2]2 and cesium acetate additive under air conditions.
- Heat the mixture to 100°C for 12 hours, then perform aqueous workup and silica gel chromatography to isolate the product.
The reaction is typically conducted at 100°C for a duration of 12 hours, a timeframe that balances complete conversion with energy efficiency. Upon completion, the workup procedure is remarkably straightforward, involving a simple aqueous quench followed by extraction with ethyl acetate. The crude product can then be purified via standard silica gel chromatography using a petroleum ether and ethyl acetate gradient. This simplicity in downstream processing is a major benefit for scale-up, as it avoids the need for complex crystallization protocols or preparative HPLC, which are often cost-prohibitive at the kilogram scale. For detailed standard operating procedures and safety data sheets regarding the specific reagents, please refer to the technical documentation provided below.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits beyond mere chemical elegance. The primary driver for cost reduction lies in the consolidation of multiple synthetic steps into a single pot. By eliminating the isolation and purification of intermediates, manufacturers can significantly reduce solvent consumption, labor hours, and equipment occupancy time. This intensification of the process directly translates to lower operational expenditures and a reduced carbon footprint, aligning with corporate sustainability goals. Furthermore, the reliance on commercially available starting materials, such as substituted indoles and simple ketone-derived sulfur ylides, mitigates supply chain risks associated with custom-synthesized building blocks that may have long lead times or single-source dependencies.
- Cost Reduction in Manufacturing: The elimination of pre-functionalization steps, such as halogenation or lithiation, removes the need for expensive reagents and cryogenic cooling infrastructure. This simplification allows for the use of standard glass-lined reactors and off-the-shelf utilities, drastically lowering the capital expenditure required for production. Additionally, the high atom economy of the tandem reaction ensures that a greater proportion of the raw material mass is incorporated into the final product, minimizing waste disposal costs which are a significant factor in the total cost of ownership for fine chemical manufacturing.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to higher batch-to-batch consistency, a critical metric for maintaining supply continuity. The tolerance for air and moisture during the setup phase reduces the risk of batch failure due to minor deviations in inert gas quality or sealing integrity. Moreover, the broad substrate scope means that if a specific substituted indole becomes unavailable, alternative analogs can often be processed using the same platform technology without extensive re-optimization, providing flexibility in sourcing strategies and protecting against raw material shortages.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram quantities is facilitated by the absence of hazardous reagents like organolithiums or pyrophoric catalysts. The exothermic profile is manageable under standard reflux conditions, reducing the engineering controls needed for heat dissipation. From an environmental compliance standpoint, the generation of benign byproducts and the use of recyclable solvents like THF simplify wastewater treatment and solvent recovery processes. This ease of compliance accelerates regulatory approval timelines for new drug applications, getting products to market faster.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this rhodium-catalyzed technology. These answers are derived from the experimental data and scope limitations defined in the patent literature, providing a realistic expectation of the process capabilities. Understanding these nuances is vital for project planning and risk assessment when integrating this chemistry into your development pipeline.
Q: What are the primary advantages of this Rh-catalyzed method over traditional cyclization routes?
A: This method utilizes a direct C-H activation strategy that eliminates the need for pre-functionalized halide substrates, significantly improving atom economy and reducing waste generation compared to classical cross-coupling approaches.
Q: Is the process scalable for industrial production of API intermediates?
A: Yes, the patent explicitly highlights the simplicity of operation and mild reaction conditions (80-140°C) as key factors making it suitable for industrial production, avoiding harsh reagents that complicate scale-up.
Q: What is the substrate scope regarding electronic properties on the indole ring?
A: The methodology demonstrates broad tolerance, successfully accommodating electron-withdrawing groups like fluorine and chlorine, as well as electron-donating alkyl and alkoxy groups, ensuring versatility for diverse drug discovery programs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indolo[2,1-a]isoquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from bench-scale discovery to commercial supply requires more than just a promising patent; it demands deep process engineering expertise. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the efficiencies demonstrated in the laboratory are faithfully reproduced in the plant. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to monitor impurity profiles and ensure that every batch of indolo[2,1-a]isoquinoline intermediate meets the exacting standards required for API synthesis. Our commitment to quality assurance guarantees that your supply chain remains uninterrupted and compliant with global regulatory frameworks.
We invite you to leverage our technical capabilities to optimize your specific project requirements. Whether you need to adapt this rhodium-catalyzed route for a novel derivative or require a Customized Cost-Saving Analysis comparing this method to your current legacy process, our experts are ready to assist. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your custom synthesis needs. Let us partner with you to accelerate your drug development timeline through superior chemical manufacturing solutions.
