Advanced Palladium-Catalyzed Carbonylation for Scalable 1,5-Dihydro-2H-Pyrrole-2-One Production
Introduction to Next-Generation Pyrrol-2-One Synthesis
The structural motif of 1,5-dihydro-2H-pyrrole-2-one serves as a critical pharmacophore found in a diverse array of bioactive natural products and therapeutic agents. As illustrated in the reference structures, this core skeleton is integral to the efficacy of potent antibiotics like Althiomycin, hypoglycemic agents such as Glimepiride, and anticancer candidates like Isomalyngamide A. Recognizing the immense value of this scaffold, recent advancements in organic synthesis have sought to streamline its construction. Specifically, patent CN112694430B discloses a groundbreaking preparation method that leverages palladium-catalyzed bis-carbonylation to construct these complex heterocycles in a single operational step. This technological leap addresses long-standing challenges in heterocyclic chemistry by utilizing readily available starting materials—propargyl amines and benzyl chlorides—thereby offering a robust platform for the rapid generation of diverse chemical libraries.
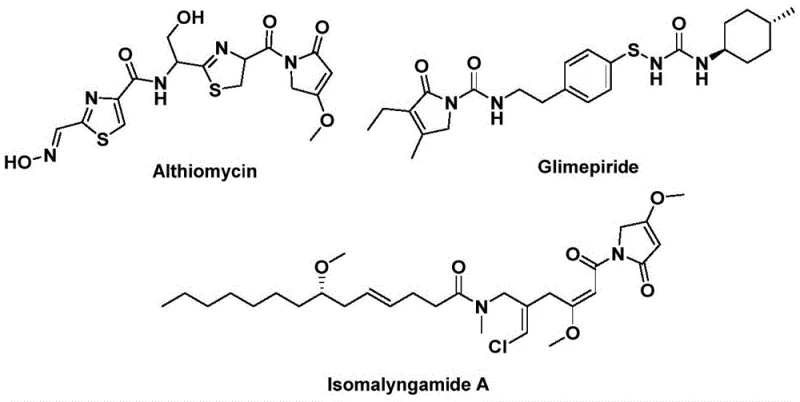
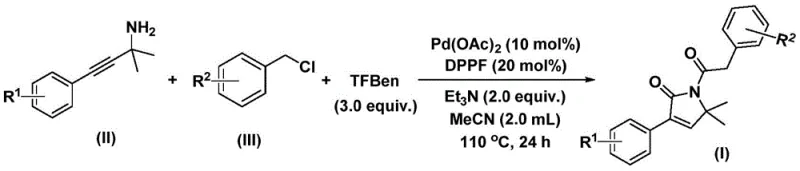
For research and development teams focused on drug discovery, the ability to access this scaffold efficiently is paramount. Traditional routes often suffer from multi-step sequences, harsh reaction conditions, or poor atom economy. The novel approach detailed in the patent data circumvents these limitations by employing a sophisticated catalytic cycle that integrates carbonylation and cyclization seamlessly. By replacing hazardous gaseous carbon monoxide with a solid surrogate, the process not only enhances safety profiles but also improves reproducibility, making it an ideal candidate for both laboratory-scale optimization and eventual commercial manufacturing of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,5-dihydro-2H-pyrrol-2-one derivatives has relied on classical condensation reactions or multi-step cyclization protocols that often require stringent anhydrous conditions and expensive reagents. Conventional carbonylation strategies frequently necessitate the use of high-pressure carbon monoxide gas, which introduces significant safety hazards and requires specialized autoclave equipment that is not universally available in standard synthetic laboratories. Furthermore, traditional methods often exhibit limited substrate scope, struggling to accommodate sensitive functional groups or sterically hindered substrates without significant degradation in yield. These inefficiencies result in prolonged development timelines and increased costs, creating bottlenecks for procurement managers seeking reliable sources of complex intermediates.
The Novel Approach
In stark contrast, the methodology described in patent CN112694430B utilizes a palladium-catalyzed system that operates under relatively mild thermal conditions (100–120°C) using acetonitrile as a solvent. This innovative route employs 1,3,5-tricarboxylic acid phenol ester (TFBen) as a solid carbon monoxide substitute, effectively decoupling the synthesis from the logistical and safety constraints of gaseous CO. The reaction tolerates a wide array of functional groups, including methoxy, halogen, and trifluoromethyl substituents, demonstrating exceptional versatility. By merging the carbonylation and ring-closing events into a single pot, this approach drastically reduces waste generation and simplifies the workup procedure, aligning perfectly with modern green chemistry principles and cost-reduction strategies in fine chemical manufacturing.
Mechanistic Insights into Palladium-Catalyzed Bis-Carbonylation
The elegance of this synthesis lies in its intricate catalytic cycle, which orchestrates the assembly of the five-membered lactam ring through a series of well-defined organometallic transformations. The reaction initiates with the oxidative addition of the palladium(0) species into the carbon-chlorine bond of the benzyl chloride substrate, generating a reactive benzyl-palladium intermediate. Subsequently, carbon monoxide, released in situ from the thermal decomposition of the phenol ester surrogate, inserts into the palladium-carbon bond to form an acyl-palladium species. This acyl intermediate then undergoes nucleophilic attack by the propargyl amine, setting the stage for the crucial cyclization event.
Following the initial ring closure, a second molecule of carbon monoxide inserts into the metallacycle, expanding the coordination sphere before final reductive elimination releases the target 1,5-dihydro-2H-pyrrole-2-one product and regenerates the active catalyst. This bis-carbonylation mechanism is highly efficient, minimizing the formation of linear byproducts that often plague simpler carbonylation reactions. For quality control teams, understanding this mechanism is vital for impurity profiling; the high selectivity of the palladium cycle ensures that the crude reaction mixture is clean, facilitating easier purification and ensuring that the final API intermediate meets stringent regulatory specifications for residual metals and organic impurities.
How to Synthesize 1,5-Dihydro-2H-Pyrrole-2-One Efficiently
The practical execution of this synthesis is designed for simplicity and scalability, requiring standard laboratory glassware and commercially available reagents. The protocol involves charging a reaction vessel with palladium acetate, a diphosphine ligand (DPPF), the solid CO source, triethylamine base, and the respective amine and chloride substrates in acetonitrile. The mixture is then heated to promote the catalytic turnover. Detailed standard operating procedures regarding stoichiometry, specific temperature ramps, and purification workflows are essential for consistent results.
- Combine palladium acetate, DPPP ligand, solid CO surrogate (TFBen), triethylamine, propargyl amine, and benzyl chloride in acetonitrile.
- Heat the reaction mixture to 110°C and stir for 24 hours under inert atmosphere to facilitate bis-carbonylation and cyclization.
- Filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the target pyrrol-2-one compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented technology offers compelling advantages that directly impact the bottom line and supply chain resilience. The shift towards a one-pot synthesis using stable, solid reagents significantly de-risks the manufacturing process. By eliminating the dependency on high-pressure gas infrastructure, facilities can produce these valuable intermediates with lower capital expenditure and reduced insurance liabilities. Furthermore, the use of commodity chemicals like benzyl chlorides and simple propargyl amines ensures that raw material costs remain low and supply is continuous, mitigating the risk of shortages that often affect exotic starting materials.
- Cost Reduction in Manufacturing: The consolidation of multiple synthetic steps into a single catalytic operation inherently reduces labor costs, solvent consumption, and energy usage. The elimination of expensive transition metal removal steps, often required in less selective couplings, further drives down the cost of goods sold (COGS). Additionally, the high atom economy of the carbonylation process ensures that a maximum proportion of the starting mass is converted into the desired product, minimizing waste disposal fees and maximizing overall process efficiency.
- Enhanced Supply Chain Reliability: The robustness of this method against varying electronic properties of substrates means that a single manufacturing line can produce a diverse portfolio of derivatives simply by swapping the starting benzyl chloride or amine. This flexibility allows suppliers to respond rapidly to changing demand patterns for different analogs without retooling entire production lines. The reliance on shelf-stable solids rather than pressurized gases also simplifies logistics and storage, ensuring uninterrupted production schedules even during global supply chain disruptions.
- Scalability and Environmental Compliance: The reaction conditions (100–120°C) are well within the operational range of standard industrial reactors, facilitating seamless scale-up from kilogram to tonne quantities. The use of acetonitrile, a common and recoverable solvent, combined with the absence of toxic gaseous emissions, simplifies environmental compliance and waste treatment. This alignment with green chemistry metrics makes the process attractive for companies aiming to reduce their carbon footprint and meet increasingly strict regulatory standards for pharmaceutical manufacturing.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology for their specific pipelines, we have compiled answers to common inquiries regarding the reaction parameters and scope. These insights are derived directly from the experimental data and mechanistic understanding of the palladium-catalyzed system.
Q: What is the primary advantage of using TFBen over gaseous CO in this synthesis?
A: Using 1,3,5-tricarboxylic acid phenol ester (TFBen) as a solid carbon monoxide surrogate eliminates the need for high-pressure gas equipment, significantly enhancing operational safety and simplifying the reaction setup for scale-up.
Q: Does this method tolerate electron-withdrawing groups on the benzyl chloride substrate?
A: Yes, the protocol demonstrates excellent substrate compatibility, successfully accommodating various substituents including halogens (F, Cl, Br), trifluoromethyl groups, and cyano groups with high yields.
Q: What represents the typical purity profile for compounds synthesized via this route?
A: The reaction proceeds with high selectivity, minimizing side products. Following standard silica gel purification, the resulting 1,5-dihydro-2H-pyrrole-2-one derivatives typically exhibit high purity suitable for downstream pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Dihydro-2H-Pyrrole-2-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the pivotal role that efficient synthetic methodologies play in accelerating drug development timelines. Our team of expert chemists has thoroughly analyzed the potential of this palladium-catalyzed route and is prepared to leverage it for your custom synthesis needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you require gram-scale samples for SAR studies or metric tonnes for clinical trials, our output remains consistent and reliable. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 1,5-dihydro-2H-pyrrole-2-one intermediate delivered meets the highest industry standards.
We invite you to collaborate with us to optimize your supply chain for these critical building blocks. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to contact us directly to obtain specific COA data for related analogs and to discuss route feasibility assessments for your proprietary targets, ensuring a seamless transition from benchtop discovery to commercial reality.
