Advanced Palladium-Catalyzed Carbonylation for Scalable 1,5-Dihydro-2H-Pyrrole-2-One Production
Introduction to Next-Generation Pyrrol-2-One Synthesis
The structural motif of 1,5-dihydro-2H-pyrrole-2-one serves as a critical pharmacophore embedded within numerous high-value bioactive natural products and therapeutic agents. As illustrated in the chemical landscape, this core skeleton is fundamental to the efficacy of potent antibiotics like Althiomycin, hypoglycemic agents such as Glimepiride, and anticancer candidates like Isomalyngamide A. 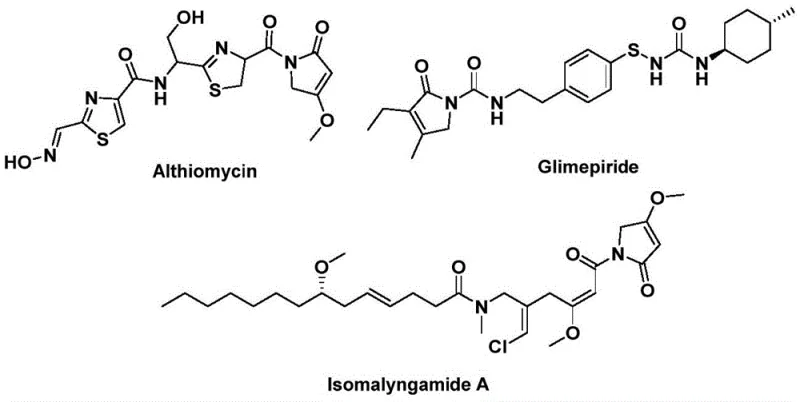 . Recognizing the immense pharmaceutical potential of this scaffold, recent innovations have focused on streamlining its construction. Specifically, patent CN112694430B discloses a groundbreaking preparation method that leverages palladium-catalyzed bis-carbonylation to assemble these complex heterocycles in a single operational step. This technological leap addresses long-standing challenges in organic synthesis by utilizing readily available starting materials—propargyl amines and benzyl chlorides—thereby offering a robust pathway for the reliable pharmaceutical intermediate supplier seeking to optimize their production pipelines.
. Recognizing the immense pharmaceutical potential of this scaffold, recent innovations have focused on streamlining its construction. Specifically, patent CN112694430B discloses a groundbreaking preparation method that leverages palladium-catalyzed bis-carbonylation to assemble these complex heterocycles in a single operational step. This technological leap addresses long-standing challenges in organic synthesis by utilizing readily available starting materials—propargyl amines and benzyl chlorides—thereby offering a robust pathway for the reliable pharmaceutical intermediate supplier seeking to optimize their production pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of the 1,5-dihydro-2H-pyrrol-2-one backbone has been fraught with synthetic inefficiencies that hinder large-scale manufacturing. Classical approaches often rely on multi-step sequences involving harsh cyclization conditions, unstable intermediates, or the use of toxic gaseous carbon monoxide under high pressure. These legacy methods frequently suffer from poor atom economy and limited functional group tolerance, necessitating extensive protection and deprotection strategies that inflate both cost and waste. Furthermore, the reliance on gaseous CO introduces significant safety hazards and engineering complexities, requiring specialized high-pressure reactors that are not universally available in standard fine chemical facilities. Consequently, the commercial scale-up of complex pharmaceutical intermediates via these traditional routes often encounters bottlenecks related to safety compliance and process reproducibility.
The Novel Approach
In stark contrast, the novel methodology outlined in the patent data revolutionizes this synthesis by employing a palladium-catalyzed bis-carbonylation strategy that operates under remarkably mild and safe conditions. By utilizing a solid carbon monoxide surrogate, specifically 1,3,5-tricarboxylic acid phenol ester (TFBen), the process completely circumvents the need for handling hazardous CO gas. This innovation allows the reaction to proceed efficiently in standard glassware or reactors at temperatures between 100°C and 120°C. The one-pot nature of the transformation means that the formation of the five-membered lactam ring occurs concomitantly with the installation of two carbonyl groups, drastically reducing the number of unit operations required. This streamlined approach not only enhances reaction efficiency but also broadens the utility of the method, making it an ideal candidate for cost reduction in API manufacturing where simplicity and safety are paramount.
Mechanistic Insights into Palladium-Catalyzed Bis-Carbonylation
The elegance of this synthesis lies in its sophisticated yet efficient catalytic cycle, which orchestrates the assembly of the pyrrol-2-one core through a series of well-defined organometallic steps. The reaction initiates with the oxidative addition of the palladium catalyst into the carbon-chlorine bond of the benzyl chloride substrate, generating a reactive benzyl-palladium intermediate. Subsequently, carbon monoxide, which is liberated in situ from the phenol 1,3,5-tricarboxylate surrogate, inserts into this intermediate to form an acyl-palladium species. This acyl complex then engages with the propargyl amine nucleophile, triggering a cyclization event that constructs the initial five-membered ring framework coordinated to the metal center.
Following the initial ring closure, the mechanism proceeds through a second, crucial carbonylation step. A second molecule of carbon monoxide inserts into the palladium-carbon bond of the cyclic intermediate, expanding the coordination sphere and setting the stage for the final lactam formation. The catalytic cycle concludes with a reductive elimination step that releases the final 1,5-dihydro-2H-pyrrol-2-one product and regenerates the active palladium(0) species for the next turnover. 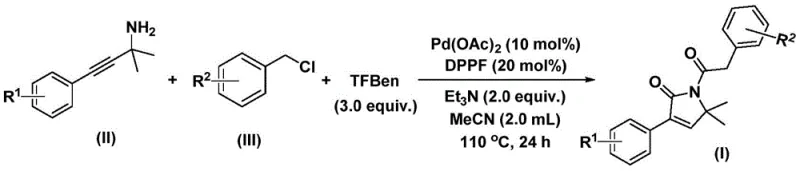 . This mechanistic pathway ensures high regioselectivity and minimizes the formation of oligomeric byproducts, thereby simplifying the impurity profile and facilitating downstream purification efforts essential for maintaining stringent purity specifications.
. This mechanistic pathway ensures high regioselectivity and minimizes the formation of oligomeric byproducts, thereby simplifying the impurity profile and facilitating downstream purification efforts essential for maintaining stringent purity specifications.
How to Synthesize 1,5-Dihydro-2H-Pyrrole-2-One Efficiently
The practical execution of this synthesis is designed for ease of operation, requiring minimal specialized equipment while delivering high conversion rates. The protocol involves charging a reaction vessel with the palladium catalyst, the diphosphine ligand, the solid CO source, base, and the two primary substrates in an organic solvent such as acetonitrile. The detailed standardized synthesis steps for replicating this high-efficiency route are provided in the guide below.
- Combine palladium acetate, DPPP ligand, solid CO surrogate (TFBen), triethylamine, propargyl amine, and benzyl chloride in acetonitrile.
- Heat the reaction mixture to 110°C and stir for 24 hours under inert atmosphere to facilitate bis-carbonylation and cyclization.
- Filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the target pyrrol-2-one compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates into tangible strategic benefits that extend beyond mere chemical yield. The shift towards this catalytic system resolves several critical pain points associated with traditional heterocycle synthesis, particularly regarding raw material security and operational expenditure. By replacing hazardous gases with stable solids and utilizing commodity chemicals as starting blocks, the process de-risks the supply chain and stabilizes production costs against volatile market fluctuations.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the utilization of inexpensive and commercially abundant starting materials, specifically benzyl chlorides and propargyl amines, which are available in bulk quantities from major chemical distributors. Furthermore, the elimination of high-pressure gas infrastructure reduces capital expenditure (CAPEX) requirements for reactor setups, while the high atom efficiency of the bis-carbonylation minimizes raw material waste. The use of a solid CO surrogate also negates the need for expensive gas scrubbing systems, leading to substantial cost savings in both equipment maintenance and regulatory compliance.
- Enhanced Supply Chain Reliability: From a logistics perspective, the reliance on shelf-stable solid reagents rather than compressed gases significantly simplifies storage and transportation protocols. This stability ensures that production schedules are not disrupted by gas delivery delays or cylinder shortages, thereby enhancing the overall reliability of the supply chain. Additionally, the broad substrate compatibility demonstrated in the patent data implies that a single manufacturing line can be adapted to produce a diverse library of derivatives, allowing for agile responses to changing market demands without the need for extensive process re-validation.
- Scalability and Environmental Compliance: The operational simplicity of running the reaction at atmospheric pressure in common solvents like acetonitrile facilitates seamless scale-up from laboratory benchtop to multi-ton commercial production. The reduced hazard profile inherently lowers the environmental footprint of the manufacturing process, aligning with modern green chemistry principles and easing the burden on waste treatment facilities. This compliance advantage is critical for maintaining uninterrupted operations in regions with stringent environmental regulations, ensuring long-term business continuity.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific applications, we have compiled answers to common inquiries regarding the reaction parameters and scope. These insights are derived directly from the experimental data and mechanistic understanding of the patented process, providing a clear picture of its capabilities and limitations in a production environment.
Q: What is the primary advantage of using TFBen over gaseous CO in this synthesis?
A: Using 1,3,5-tricarboxylic acid phenol ester (TFBen) as a solid carbon monoxide surrogate eliminates the need for high-pressure gas equipment, significantly enhancing operational safety and simplifying the reaction setup for scale-up.
Q: Does this method tolerate electron-withdrawing groups on the benzyl chloride substrate?
A: Yes, the protocol demonstrates excellent substrate compatibility, successfully accommodating various substituents including halogens (F, Cl, Br), trifluoromethyl groups, and cyano groups with high yields.
Q: What represents the typical purity profile for compounds synthesized via this route?
A: The reaction proceeds with high selectivity, minimizing side products. Following standard silica gel purification, the resulting 1,5-dihydro-2H-pyrrole-2-one derivatives typically exhibit high purity suitable for downstream pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Dihydro-2H-Pyrrole-2-One Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires more than just chemical knowledge; it demands engineering excellence and rigorous quality control. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields observed in the lab are faithfully reproduced on an industrial scale. We are committed to delivering high-purity 1,5-dihydro-2H-pyrrole-2-one intermediates that meet the most demanding stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation.
We invite forward-thinking partners to collaborate with us to leverage this advanced synthetic technology for their drug development programs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data for our existing inventory or to discuss route feasibility assessments for your proprietary targets, ensuring a secure and efficient supply of these critical building blocks.
