Advanced Synthesis of Tetrahydroquinazoline Derivatives for Commercial Pharmaceutical Production
The pharmaceutical industry continuously seeks robust synthetic pathways for nitrogen-containing heterocyclic compounds due to their prevalence in bioactive molecules. Patent CN109651375B introduces a significant advancement in the synthesis of tetrahydroquinazoline derivatives, which serve as critical scaffolds for various therapeutic agents including anticancer and antihypertensive drugs. This specific intellectual property outlines a novel two-step methodology that overcomes traditional limitations associated with harsh reaction conditions and complex purification processes. By leveraging the unique properties of hexafluoroisopropanol as a solvent, the disclosed method achieves high atom economy and operational safety, making it an attractive candidate for commercial scale-up. The strategic integration of nucleophilic aromatic substitution followed by a mild cyclization step ensures that manufacturers can produce high-purity intermediates efficiently. For R&D directors and procurement managers, understanding the technical nuances of this patent is essential for evaluating potential supply chain partnerships and optimizing manufacturing costs in the competitive landscape of fine chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the tetrahydroquinazoline skeleton has been plagued by several inherent inefficiencies that hinder large-scale production. Conventional synthetic routes often rely on prolonged reaction times and require heating to reflux temperatures, which significantly increases energy consumption and operational risks in a plant setting. Many traditional methods necessitate the use of expensive transition metal catalysts or strong acids, which not only drive up raw material costs but also introduce challenges in removing trace metal impurities from the final active pharmaceutical ingredient. Furthermore, the atom economy of these older processes is frequently suboptimal, generating substantial chemical waste that complicates environmental compliance and waste disposal protocols. The reliance on harsh conditions can also limit the functional group tolerance, restricting the diversity of derivatives that can be synthesized without protecting group strategies. These cumulative factors create bottlenecks in the supply chain, leading to longer lead times and higher overall manufacturing costs for pharmaceutical companies seeking reliable sources of these complex intermediates.
The Novel Approach
In stark contrast to legacy methods, the technology disclosed in CN109651375B offers a streamlined and efficient pathway that addresses these critical pain points directly. The novel approach utilizes a sequential strategy starting with the nucleophilic substitution of substituted ortho-fluorobenzaldehyde with a secondary amine, followed by a one-pot cyclization with an amine compound. A key innovation lies in the use of hexafluoroisopropanol, which facilitates the reaction of weak nucleophiles without the need for additional acidic or metallic catalysts. This solvent choice not only simplifies the reaction setup but also enhances the recovery and recycling potential, contributing to a greener manufacturing profile. The process operates under mild conditions, particularly in the second step where room temperature is sufficient, drastically reducing thermal stress on the equipment and energy requirements. The high yields reported, reaching up to 96%, demonstrate the robustness of this chemistry, ensuring that raw materials are converted into valuable products with minimal loss. 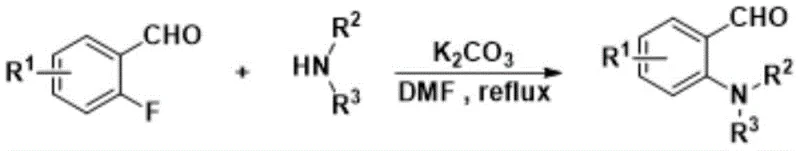
Mechanistic Insights into HFIP-Promoted Cyclization
The core of this synthetic breakthrough lies in the mechanistic role of hexafluoroisopropanol (HFIP) during the cyclization phase. HFIP is characterized by its high hydrogen bond donating ability and low nucleophilicity, which creates a unique microenvironment that stabilizes charged intermediates and transition states. In the context of this reaction, HFIP promotes the dehydration condensation between the ortho-substituted benzaldehyde intermediate and the amine compound to form an imine species. This activation allows the subsequent intramolecular [1,5]-hydrogen transfer and cyclization to proceed smoothly at room temperature, a feat that typically requires elevated temperatures or strong catalysts in other solvents. The ability of HFIP to enhance the electrophilicity of the carbonyl group without consuming stoichiometric reagents is a testament to its efficiency as a reaction medium. This mechanistic advantage translates directly to process simplicity, as it eliminates the need for complex additive systems that often complicate downstream purification and quality control testing in a GMP environment.
Furthermore, the impurity profile generated by this method is significantly cleaner compared to acid-catalyzed alternatives. The mild nature of the HFIP-promoted reaction minimizes side reactions such as polymerization or over-alkylation, which are common pitfalls in the synthesis of nitrogen heterocycles. The resulting crude product typically requires only standard silica gel column chromatography for purification, avoiding the need for specialized resin treatments or extensive recrystallization steps. This high level of chemoselectivity ensures that the final tetrahydroquinazoline derivatives meet stringent purity specifications required for pharmaceutical applications. For technical teams, this means a more predictable manufacturing process with reduced batch-to-batch variability. The structural integrity of the sensitive functional groups on the aromatic ring is preserved, allowing for a broader scope of substituents to be introduced without degradation. 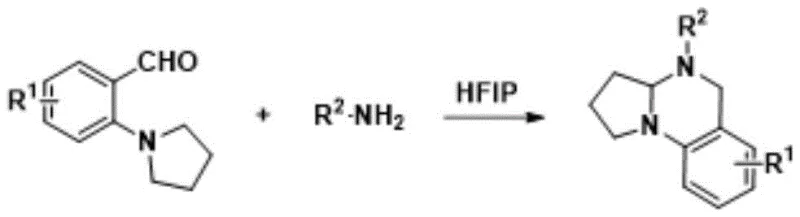
How to Synthesize Tetrahydroquinazoline Derivatives Efficiently
The practical implementation of this synthesis route involves a straightforward two-step protocol that is well-suited for both laboratory optimization and industrial production. The first stage focuses on the preparation of the key ortho-substituted benzaldehyde intermediate through a nucleophilic aromatic substitution reaction using potassium carbonate in DMF. Once this intermediate is isolated and purified, it serves as the substrate for the critical cyclization step in hexafluoroisopropanol. The detailed standardized synthesis steps, including specific molar ratios, temperature controls, and workup procedures, are outlined in the guide below to ensure reproducibility and safety. Adhering to these parameters is crucial for achieving the high yields and purity levels documented in the patent data.
- Perform nucleophilic substitution of substituted o-fluorobenzaldehyde with a secondary amine using K2CO3 in DMF at 140-160°C.
- Isolate the ortho-substituted benzaldehyde intermediate via extraction and silica gel column chromatography.
- React the intermediate with an amine compound in hexafluoroisopropanol at room temperature to form the tetrahydroquinazoline derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers substantial benefits for procurement managers and supply chain heads looking to optimize their vendor networks. The elimination of expensive transition metal catalysts and the reduction in energy consumption due to mild reaction conditions directly contribute to a lower cost of goods sold. This cost efficiency can be passed down through the supply chain, providing a competitive pricing advantage for the final drug product. Additionally, the simplicity of the post-processing steps reduces the turnaround time for batch release, enhancing the overall responsiveness of the supply chain to market demands. The use of readily available and non-toxic raw materials further mitigates supply risks associated with scarce or regulated reagents. These factors collectively build a more resilient and cost-effective manufacturing framework for tetrahydroquinazoline intermediates.
- Cost Reduction in Manufacturing: The process eliminates the need for costly palladium or other transition metal catalysts, which are often subject to price volatility and strict residual limits in pharmaceuticals. By relying on organic solvents and inorganic bases like potassium carbonate, the raw material costs are significantly stabilized and reduced. The high atom economy ensures that a larger proportion of the starting materials ends up in the final product, minimizing waste disposal costs. Furthermore, the ability to recover and reuse the hexafluoroisopropanol solvent adds another layer of long-term economic value to the process. These combined efficiencies result in a leaner manufacturing cost structure without compromising on quality.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as substituted fluorobenzaldehydes and common amines ensures a stable and diverse supply base. Unlike specialized catalysts that may have single-source suppliers, these raw materials are widely available from multiple global vendors, reducing the risk of supply disruptions. The mild reaction conditions also lower the barrier for contract manufacturing organizations to adopt the process, increasing the number of qualified suppliers in the market. This redundancy is critical for maintaining continuous production schedules and meeting the rigorous delivery timelines expected by large pharmaceutical companies. Consequently, the supply chain becomes more agile and capable of scaling up rapidly in response to clinical or commercial demand.
- Scalability and Environmental Compliance: The operational safety of running reactions at room temperature in the second step significantly reduces the engineering controls required for scale-up. This simplifies the transition from pilot plant to commercial production, as there is less need for specialized high-pressure or high-temperature equipment. The reduced generation of hazardous waste and the potential for solvent recycling align with increasingly strict environmental regulations and corporate sustainability goals. A greener process profile not only avoids regulatory hurdles but also enhances the brand reputation of the manufacturing partner. This makes the technology particularly attractive for companies aiming to reduce their carbon footprint while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these tetrahydroquinazoline derivatives. The answers are derived directly from the experimental data and beneficial effects described in the patent documentation. Understanding these details helps stakeholders make informed decisions about process adoption and material sourcing.
Q: What are the advantages of using hexafluoroisopropanol in this synthesis?
A: Hexafluoroisopropanol acts as a solvent with high hydrogen bond donating ability and ionization power, promoting the reaction of weak nucleophiles without needing Lewis or Bronsted acid catalysts, while being easy to recover and environmentally friendly.
Q: What is the maximum yield reported for this tetrahydroquinazoline synthesis method?
A: According to patent CN109651375B, the yield of the tetrahydroquinazoline derivative can reach up to 96% under optimized conditions.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the method features simple operation, mild reaction conditions (room temperature for the second step), and easy post-processing via silica gel column chromatography, making it highly scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydroquinazoline Derivatives Supplier
NINGBO INNO PHARMCHEM stands ready to leverage this advanced synthetic technology to support your pharmaceutical development and commercialization goals. As a seasoned CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of tetrahydroquinazoline derivatives meets the highest industry standards. We understand the critical nature of intermediate supply in the drug development timeline and are committed to providing a seamless and reliable partnership. Our technical team is well-versed in the nuances of HFIP-mediated chemistry and can optimize the process further to suit your specific cost and capacity requirements.
We invite you to engage with our technical procurement team to discuss how this innovative route can enhance your project's economics and timeline. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential financial benefits of switching to this manufacturing method. We encourage you to reach out for specific COA data and route feasibility assessments to validate the compatibility of this chemistry with your existing workflows. Let us help you engineer a more efficient and sustainable supply chain for your next-generation therapeutics.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
