Advanced Metal-Free Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Intermediates for Scalable Pharmaceutical Manufacturing
The landscape of heterocyclic chemistry is continually evolving, driven by the demand for more efficient and sustainable pathways to bioactive scaffolds. Patent CN113307790B introduces a groundbreaking preparation method for 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compounds, a structural motif prevalent in numerous high-value pharmaceutical agents and functional materials. As illustrated in the broader context of medicinal chemistry, 1,2,4-triazole rings serve as critical pharmacophores in drugs such as antifungal agents and kinase inhibitors, often acting as bioisosteres for amide bonds or carboxylic acids to improve metabolic stability. 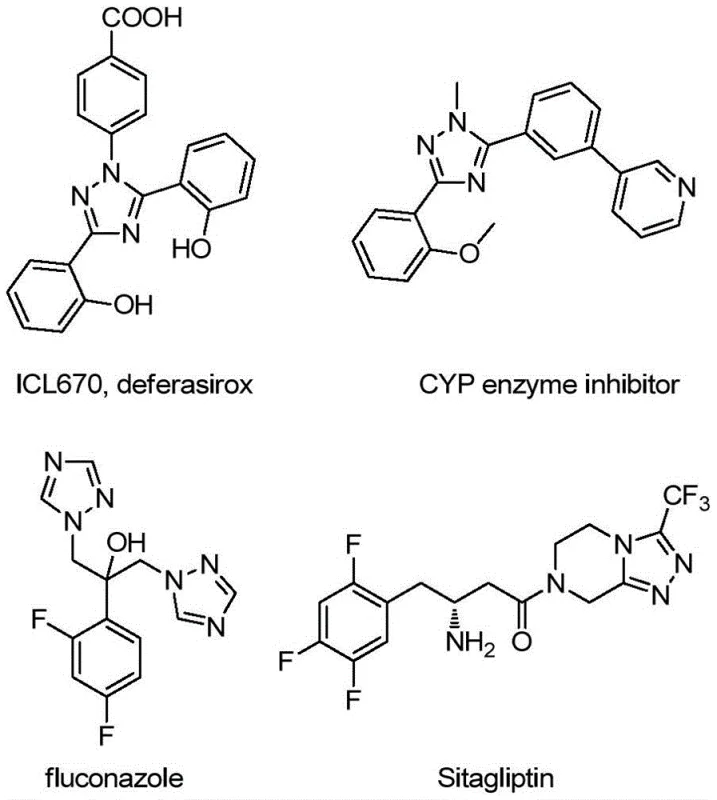 . This specific innovation addresses the long-standing challenges associated with synthesizing these complex fused systems by replacing multi-step, low-yielding sequences with a direct, oxidative cyclization strategy that operates under remarkably mild conditions.
. This specific innovation addresses the long-standing challenges associated with synthesizing these complex fused systems by replacing multi-step, low-yielding sequences with a direct, oxidative cyclization strategy that operates under remarkably mild conditions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of quinolyl-substituted 1,2,4-triazoles has been plagued by inefficiency and operational complexity. Traditional synthetic routes typically rely on quinoline-2-carboxylic acid as the primary starting material, necessitating a cumbersome five-step reaction sequence to achieve the final target structure. This legacy approach suffers from a dismal total yield of approximately 17%, rendering it economically unviable for large-scale commercial manufacturing. Furthermore, these conventional methods often demand severe reaction conditions, including strict anhydrous and anaerobic environments, which significantly increase operational costs and safety risks in an industrial setting. The reliance on such苛刻 conditions not only limits the scope of substrate tolerance but also complicates the supply chain by requiring specialized equipment and handling protocols that are difficult to maintain consistently across different production batches.
The Novel Approach
In stark contrast, the methodology disclosed in CN113307790B utilizes a direct oxidative coupling between readily available 2-methylquinoline derivatives and trifluoroethylimide hydrazides. This transformative approach leverages a catalytic system comprising tetrabutylammonium iodide (TBAI) and tert-butyl peroxide (TBHP) to facilitate the formation of the triazole ring in a single pot. 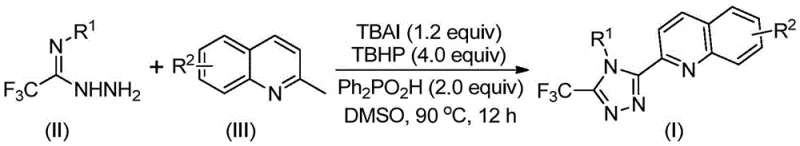 . By bypassing the need for pre-functionalized carboxylic acid precursors, this novel route drastically reduces the number of unit operations and eliminates the accumulation of waste associated with intermediate isolations. The reaction proceeds efficiently in polar aprotic solvents like DMSO at moderate temperatures between 80°C and 100°C, achieving isolated yields as high as 97% for optimized substrates, thereby representing a paradigm shift in the manufacturing of these valuable heterocyclic intermediates.
. By bypassing the need for pre-functionalized carboxylic acid precursors, this novel route drastically reduces the number of unit operations and eliminates the accumulation of waste associated with intermediate isolations. The reaction proceeds efficiently in polar aprotic solvents like DMSO at moderate temperatures between 80°C and 100°C, achieving isolated yields as high as 97% for optimized substrates, thereby representing a paradigm shift in the manufacturing of these valuable heterocyclic intermediates.
Mechanistic Insights into TBAI/TBHP Promoted Oxidative Cyclization
The mechanistic elegance of this transformation lies in the synergistic interaction between the iodide promoter and the peroxide oxidant. The process initiates with the TBAI-catalyzed oxidation of the methyl group on the 2-methylquinoline substrate, effectively converting it in situ into a reactive aldehyde equivalent or an iodinated intermediate. This activated species then undergoes a condensation reaction with the trifluoroethylimide hydrazide to form a dehydrated hydrazone intermediate. Subsequent oxidative iodination and intramolecular electrophilic substitution, promoted by the diphenylphosphinic acid additive, drive the cyclization forward. The final aromatization step yields the stable 3-quinolyl-5-trifluoromethyl-1,2,4-triazole core. This radical-mediated pathway avoids the use of transition metals entirely, relying instead on the redox potential of the organic iodine species to orchestrate the bond-forming events with high precision and selectivity.
From an impurity control perspective, this mechanism offers distinct advantages for R&D teams focused on purity profiles. The absence of heavy metal catalysts means there is no risk of metal leaching into the final product, a critical parameter for pharmaceutical intermediates intended for clinical use. Furthermore, the use of diphenylphosphinic acid helps to suppress side reactions and stabilize the reactive intermediates, leading to a cleaner crude reaction mixture. The broad substrate tolerance observed in the patent examples, where various electron-donating and electron-withdrawing groups on both the quinoline and the aryl hydrazide moieties are well-tolerated, suggests that the electronic demands of the transition state are well-balanced. This robustness ensures that minor variations in raw material quality do not lead to catastrophic failures in conversion, providing a reliable safety margin for process chemists during scale-up activities.
How to Synthesize 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for reproducing these high-yielding transformations in a laboratory or pilot plant setting. The procedure involves charging a reaction vessel with the specific molar ratios of tetrabutylammonium iodide, tert-butyl peroxide aqueous solution, diphenylphosphinic acid, the hydrazide component, and the 2-methylquinoline derivative in a suitable organic solvent. The detailed standardized synthesis steps below outline the precise addition order, temperature ramping, and workup procedures required to maximize yield and purity while minimizing safety hazards associated with peroxide handling.
- Combine tetrabutylammonium iodide (TBAI), tert-butyl peroxide (TBHP) aqueous solution, diphenylphosphinic acid, trifluoroethylimide hydrazide, and 2-methylquinoline derivative in an organic solvent such as DMSO.
- Heat the reaction mixture to a temperature range of 80°C to 100°C and maintain stirring for a duration of 8 to 14 hours to ensure complete oxidative cyclization.
- Upon completion, perform filtration and silica gel column chromatography purification to isolate the final 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole product with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route presents a compelling value proposition centered on cost efficiency and supply continuity. The elimination of precious metal catalysts such as palladium or copper removes a significant cost driver from the bill of materials, while simultaneously simplifying the downstream purification process. Without the need for expensive metal scavengers or complex filtration units to meet strict residual metal specifications, the overall processing time is significantly reduced. This streamlining of the manufacturing workflow translates directly into lower operational expenditures and a reduced carbon footprint, aligning with modern green chemistry initiatives that are increasingly mandated by global regulatory bodies and corporate sustainability goals.
- Cost Reduction in Manufacturing: The economic benefits of this process are substantial, primarily driven by the use of commodity chemicals as starting materials. 2-methylquinoline and aromatic amines used to prepare the hydrazide are widely available in the bulk chemical market at competitive prices, ensuring a stable and predictable cost base. By consolidating a five-step sequence into a single telescoped operation, the method drastically reduces solvent consumption, labor hours, and energy usage associated with heating and cooling cycles for multiple reactors. This intensification of the process allows for higher throughput in existing infrastructure, effectively increasing capacity without the need for capital-intensive expansion of the production facility.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions, which do not require inert atmosphere techniques like nitrogen blanketing or glovebox operations. This tolerance to ambient conditions simplifies logistics and storage requirements for intermediates, reducing the risk of batch rejection due to moisture or oxygen sensitivity. Furthermore, the high yields reported across a diverse range of substrates indicate a flexible platform technology that can be adapted to produce various analogues quickly. This agility allows manufacturers to respond rapidly to changing market demands or custom synthesis requests from clients without lengthy re-optimization periods, ensuring consistent delivery timelines even in volatile market environments.
- Scalability and Environmental Compliance: The environmental profile of this method is superior to traditional approaches, as it avoids the generation of heavy metal waste streams that require specialized and costly disposal procedures. The use of DMSO as a solvent, while requiring careful recovery, is well-established in industrial settings with mature recycling technologies. The reaction's ability to scale from gram to kilogram quantities without loss of efficiency demonstrates its readiness for commercial deployment. This scalability ensures that the supply of these critical pharmaceutical intermediates can be maintained continuously to support downstream API production, mitigating the risk of shortages that can disrupt the entire drug development pipeline.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidative cyclization technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of adopting this method for industrial applications. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of this synthesis method over conventional routes?
A: Unlike traditional methods requiring five steps and yielding only 17%, this novel approach achieves yields up to 97% in a single pot without harsh anhydrous conditions or toxic heavy metal catalysts.
Q: Does this process require expensive transition metal catalysts?
A: No, the process utilizes an organocatalytic system based on tetrabutylammonium iodide (TBAI) and tert-butyl peroxide (TBHP), eliminating the need for costly palladium or copper catalysts and subsequent metal scavenging steps.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the patent explicitly states the reaction is easily expandable to gram-scale and beyond, utilizing cheap, commercially available starting materials and standard organic solvents like DMSO.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the global pharmaceutical supply chain. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering high-purity 3-quinolyl-5-trifluoromethyl-1,2,4-triazole derivatives that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch.
We invite potential partners to engage with our technical procurement team to discuss how this advanced metal-free technology can be leveraged for your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this streamlined process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability as a trusted partner in your journey toward commercial success.
