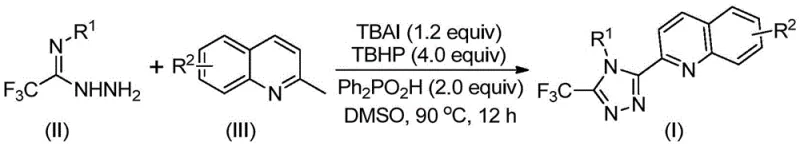
Scalable Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazoles for Advanced Drug Discovery
Scalable Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazoles for Advanced Drug Discovery
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable pathways to access complex heterocyclic scaffolds that serve as critical building blocks for next-generation therapeutics. Patent CN113307790B introduces a groundbreaking preparation method for 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compounds, addressing significant bottlenecks in traditional synthetic routes. This innovation leverages a metal-free oxidative cyclization strategy that not only simplifies the operational workflow but also dramatically improves the economic viability of producing these high-value intermediates. For R&D directors and procurement specialists alike, this technology represents a pivotal shift towards greener, more cost-effective manufacturing processes that do not compromise on purity or structural diversity.
The core of this technological advancement lies in the strategic combination of readily available starting materials, specifically 2-methylquinoline and trifluoroacetohydrazide derivatives. By utilizing a catalytic system composed of tetrabutylammonium iodide (TBAI) and tert-butyl hydroperoxide (TBHP), the method facilitates a direct C-H functionalization and cyclization sequence. This approach bypasses the need for pre-functionalized precursors, which are often expensive and difficult to source in bulk quantities. Furthermore, the reaction conditions are remarkably mild, operating effectively at temperatures between 80°C and 100°C without the stringent requirement for anhydrous or oxygen-free environments, thereby reducing the infrastructure burden on production facilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolyl-substituted 1,2,4-triazoles has been plagued by inefficient multi-step sequences that rely on quinoline-2-carboxylic acid as the primary starting material. This traditional pathway typically necessitates at least five distinct synthetic steps to achieve the final target molecule, resulting in a cumulative total yield that rarely exceeds 17%. Such low efficiency translates directly into exorbitant production costs and significant waste generation, making large-scale application economically unfeasible for most commercial entities. Additionally, the harsh reaction conditions often required for these legacy methods pose safety risks and demand specialized equipment capable of handling extreme temperatures or pressures, further inflating the capital expenditure required for manufacturing setup.
Beyond the economic drawbacks, the conventional reliance on carboxylic acid derivatives often introduces challenges in purification and impurity control. The multiple isolation steps increase the likelihood of product loss and contamination, complicating the regulatory approval process for pharmaceutical intermediates where impurity profiles must be strictly defined and controlled. For supply chain managers, the complexity of a five-step synthesis意味着 longer lead times and higher vulnerability to disruptions in the supply of specific reagents. Consequently, there has been a pressing industry demand for a more convergent, atom-economical approach that can deliver high-purity products with fewer unit operations and reduced environmental impact.
The Novel Approach
In stark contrast to the arduous traditional routes, the novel method disclosed in the patent utilizes a direct oxidative coupling strategy that merges the quinoline and triazole formation into a highly efficient cascade process. By employing 2-methylquinoline and trifluoroacetohydrazide as the foundational building blocks, the synthesis achieves a remarkable convergence, significantly shortening the production timeline. The use of TBAI and TBHP as the promoting system enables the in situ oxidation of the methyl group to an aldehyde equivalent, which subsequently undergoes condensation and cyclization. This telescoped approach eliminates the need for isolating unstable intermediates, thereby streamlining the workflow and maximizing overall throughput.
The operational simplicity of this new method cannot be overstated, as it functions effectively in common organic solvents like DMSO without the need for inert gas protection or rigorous drying of reagents. This robustness makes the process ideally suited for transfer from laboratory discovery to kilogram or ton-scale commercial production. For procurement teams, the shift to cheap and easily obtainable raw materials like 2-methylquinoline represents a substantial opportunity for cost reduction in pharmaceutical intermediate manufacturing. The ability to tolerate a wide range of functional groups on both the quinoline and the hydrazide components further enhances the utility of this method, allowing for the rapid generation of diverse compound libraries for structure-activity relationship (SAR) studies.
Mechanistic Insights into TBAI/TBHP Promoted Oxidative Cyclization
The mechanistic pathway of this transformation is a fascinating example of iodine-catalyzed oxidative functionalization. Initially, the tetrabutylammonium iodide interacts with tert-butyl hydroperoxide to generate reactive iodine species in situ. These species facilitate the oxidation of the methyl group on the 2-methylquinoline substrate, effectively converting it into a 2-quinolinecarbaldehyde intermediate without the need for external oxidants like chromium or manganese reagents. This aldehyde then undergoes a condensation reaction with the trifluoroacetohydrazide to form a hydrazone intermediate, releasing water as the only byproduct of this specific step. The presence of diphenylphosphoric acid plays a crucial role in activating the system and stabilizing the transition states during the subsequent cyclization events.
Following the formation of the hydrazone, the reaction proceeds through an oxidative iodination and intramolecular electrophilic substitution sequence. This critical phase constructs the five-membered 1,2,4-triazole ring fused to the quinoline scaffold. The final aromatization step restores the aromaticity of the triazole ring, driving the reaction to completion and yielding the stable 3-quinolyl-5-trifluoromethyl substituted product. The entire process can also be rationalized through a free radical mechanism, where radical species generated from the TBHP decomposition abstract hydrogen atoms to propagate the oxidation cycle. Understanding this mechanism is vital for process chemists aiming to optimize reaction parameters for specific substrates.
Control over the impurity profile is inherently built into this mechanism due to the high chemoselectivity of the iodine-mediated oxidation. Unlike heavy metal catalysts which can leave persistent residues requiring complex scavenging protocols, the iodine species involved are generally easier to remove during the aqueous workup or silica gel filtration. The use of diphenylphosphoric acid as an additive further suppresses side reactions by modulating the acidity of the medium, ensuring that the condensation and cyclization steps proceed with high fidelity. For quality control laboratories, this translates to cleaner crude reaction mixtures and higher purity final products after standard chromatographic purification, reducing the burden on analytical resources and speeding up batch release times.
How to Synthesize 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazoles Efficiently
Implementing this synthesis in a practical setting requires careful attention to the stoichiometry of the oxidant and the catalyst loading to ensure maximum conversion. The patent data indicates that a molar ratio of approximately 1.5:1 for the hydrazide to the quinoline substrate is optimal, driving the equilibrium towards the product while minimizing the formation of unreacted starting materials. The reaction is typically conducted in dimethyl sulfoxide (DMSO), which provides excellent solubility for both the organic substrates and the ionic catalyst system, although other polar aprotic solvents may also be viable depending on specific solubility constraints. Post-reaction processing is straightforward, involving simple filtration followed by silica gel mixing and column chromatography, techniques that are standard in almost any synthetic chemistry facility.
- Combine tetrabutylammonium iodide, tert-butyl peroxide, diphenylphosphoric acid, trifluoroacetohydrazide, and 2-methylquinoline in DMSO solvent.
- Heat the reaction mixture to 80-100°C and maintain stirring for 8 to 14 hours to ensure complete oxidative cyclization.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the high-purity triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this metal-free oxidative cyclization technology offers profound advantages for supply chain stability and cost management. The elimination of transition metal catalysts removes a significant cost center associated with both the purchase of expensive noble metals and the downstream processing required to meet strict residual metal limits in pharmaceutical products. This simplification of the purification train not only reduces the consumption of scavenging resins and solvents but also shortens the overall cycle time per batch. For procurement managers, the reliance on commodity chemicals like TBAI and TBHP, which are produced globally in vast quantities, ensures a resilient supply chain that is less susceptible to geopolitical disruptions compared to specialized catalysts.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic reduction in step count and the use of inexpensive, bulk-available reagents. By collapsing a traditional five-step sequence into a single-pot operation, manufacturers can significantly lower labor costs, energy consumption, and solvent usage. The avoidance of cryogenic conditions or high-pressure reactors further diminishes the capital intensity of the production line. Additionally, the high yields reported in the patent examples, reaching up to 97% for certain substrates, mean that less raw material is wasted, directly improving the cost of goods sold (COGS) and enhancing the overall margin profile for the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to a more reliable supply of critical intermediates. Since the process does not require strictly anhydrous or anaerobic environments, it can be executed in standard glass-lined reactors without the need for specialized nitrogen blanketing or drying trains, reducing the risk of batch failures due to moisture ingress. The broad substrate scope allows for the flexible production of various analogues using the same core protocol, enabling manufacturers to respond quickly to changing market demands or clinical trial requirements. This flexibility is a key asset for supply chain heads who must manage inventory levels and lead times for multiple drug candidates simultaneously.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram or tonnage levels is facilitated by the homogeneous nature of the reaction mixture and the absence of hazardous gaseous reagents. The use of aqueous TBHP solutions adds a layer of safety by reducing the shock sensitivity often associated with concentrated organic peroxides. From an environmental standpoint, the atom economy of the reaction is superior to classical methods, generating less chemical waste per kilogram of product. This aligns with the increasing regulatory pressure on pharmaceutical companies to adopt greener manufacturing practices, potentially lowering waste disposal costs and improving the company's sustainability metrics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear picture of what partners can expect when integrating this method into their production workflows. Understanding these nuances is essential for making informed decisions about process adoption and resource allocation.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the patented method utilizes a metal-free catalytic system based on tetrabutylammonium iodide (TBAI) and tert-butyl hydroperoxide (TBHP), eliminating the need for costly and toxic heavy metal removal steps.
Q: What are the typical reaction conditions for this oxidative cyclization?
A: The reaction proceeds efficiently in polar aprotic solvents like DMSO at temperatures between 80°C and 100°C for 8 to 14 hours, without requiring strict anhydrous or anaerobic conditions.
Q: Can this method accommodate diverse substituents on the quinoline ring?
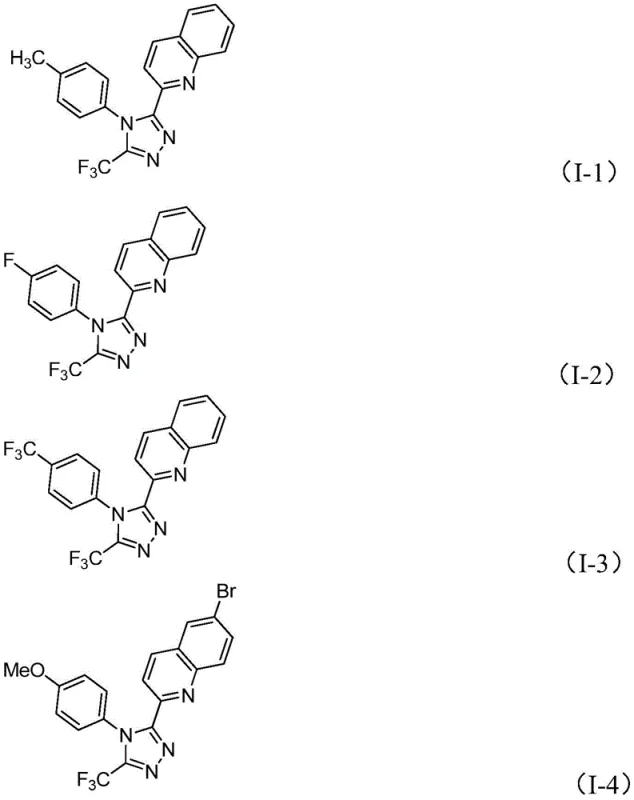
A: Yes, the method demonstrates excellent substrate tolerance, successfully synthesizing derivatives with various substituents such as methyl, methoxy, halogens, and nitro groups on both the quinoline and phenyl rings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free oxidative cyclization technology for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific solvent systems and thermal profiles required for this synthesis, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest international standards for drug substance production.
We invite you to collaborate with us to leverage this innovative route for your specific drug development programs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this method can optimize your budget without compromising quality. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary molecules, and let us help you accelerate your path to market with confidence.
