Advanced Pd-Catalyzed Carbonylation for Scalable 2-Trifluoromethyl Quinazolinone Production
Introduction to Next-Generation Quinazolinone Synthesis
The pharmaceutical industry continuously seeks robust synthetic routes for privileged scaffolds that offer enhanced metabolic stability and bioavailability. Quinazolinone derivatives represent a critical class of fused-ring nitrogen-containing heterocycles found in numerous bioactive molecules, exhibiting potent anti-inflammatory, antiviral, antifungal, and anticancer properties. The strategic introduction of a trifluoromethyl group at the 2-position of the quinazolinone core significantly modulates physicochemical parameters such as electronegativity and lipophilicity, often leading to superior drug candidates. Patent CN112125856A discloses a groundbreaking preparation method for these valuable 2-trifluoromethyl substituted quinazolinone derivatives. This technology represents a paradigm shift from hazardous gas-phase carbonylation to a safer, solid-state carbon monoxide surrogate approach, offering a reliable pharmaceutical intermediate supplier pathway for complex API synthesis.
Traditional methods for constructing the quinazolinone core often rely on harsh cyclization conditions or unstable reagents that limit scalability and safety. In contrast, the disclosed invention utilizes a transition metal palladium-catalyzed carbonylative tandem reaction. By employing 1,3,5-tricarboxylate phenol ester (TFBen) as a solid substitute for toxic carbon monoxide gas, this method circumvents the severe safety hazards associated with high-pressure CO handling. The process is characterized by its operational simplicity, utilizing cheap and readily available starting materials such as o-iodoaniline and trifluoroacetimidoyl chloride. This innovation not only addresses the critical need for cost reduction in API manufacturing but also ensures a safer working environment, making it an ideal candidate for commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-trifluoromethyl substituted quinazolinones has been plagued by significant technical and safety hurdles. Conventional literature reports typically describe cyclization reactions involving anthranilamide with ethyl trifluoroacetate, trifluoroacetic anhydride, or trifluoroacetic acid under varying conditions. Alternative routes involve the reaction of anthranilic esters with unstable trifluoroacetamides or the cyclization of isatoic anhydride with trifluoroacetic anhydride. More recent approaches utilize T3P-promoted tandem reactions of anthranilic acid, trifluoroacetic acid, and amines. However, these existing methodologies generally suffer from severe drawbacks, including苛刻 reaction conditions that require extreme temperatures or pressures, the necessity for expensive or pre-activated substrates that drive up raw material costs, and consistently low yields that hinder economic viability. Furthermore, many of these routes exhibit narrow substrate scopes, failing to tolerate diverse functional groups, which restricts their utility in medicinal chemistry optimization campaigns.
The Novel Approach
The novel methodology presented in the patent data overcomes these historical bottlenecks through a sophisticated palladium-catalyzed carbonylation strategy. This approach leverages the reactivity of o-iodoaniline and trifluoroacetimidoyl chloride in the presence of a solid carbon monoxide source. The use of TFBen allows for the controlled release of CO in situ, facilitating the carbonylation step without the need for external gas cylinders or specialized high-pressure reactors. This method demonstrates exceptional compatibility with various substituents, allowing for the synthesis of derivatives with different groups at multiple positions on the aromatic rings. As illustrated in the specific examples below, the protocol successfully generates a diverse library of compounds, proving its versatility for generating high-purity OLED material precursors or pharmaceutical building blocks.
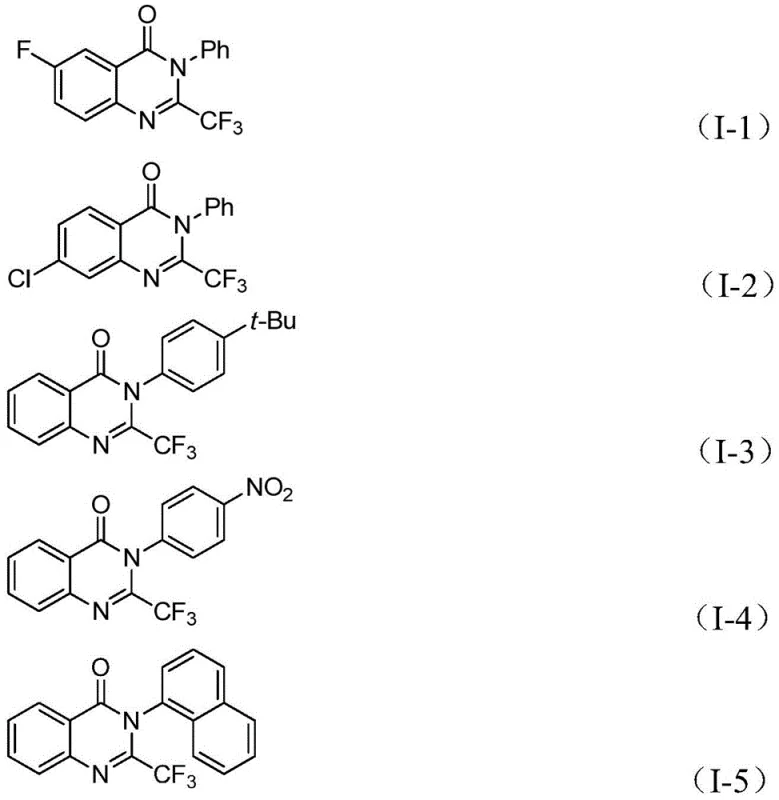
The structural diversity achievable with this method is evident in the successful synthesis of derivatives bearing electron-donating groups like tert-butyl and methyl, as well as electron-withdrawing groups such as nitro and halogens. This broad functional group tolerance is crucial for process chemists aiming to rapidly iterate on lead compounds. The ability to access these structures efficiently translates directly to reduced lead time for high-purity pharmaceutical intermediates, enabling faster progression from discovery to development stages.
Mechanistic Insights into Pd-Catalyzed Carbonylative Cyclization
The reaction mechanism involves a complex catalytic cycle initiated by the interaction of the palladium catalyst with the organic substrates. It is hypothesized that the reaction first undergoes an intermolecular carbon-nitrogen bond coupling promoted by the base, potassium tert-butoxide, to generate a trifluoroacetamidine derivative intermediate. Subsequently, the palladium catalyst inserts into the carbon-iodine bond of the o-iodoaniline moiety, forming a divalent palladium intermediate. Under the heating conditions of 90°C, the solid surrogate TFBen decomposes to release carbon monoxide, which then inserts into the carbon-palladium bond to form a key acyl palladium intermediate. This step is critical as it introduces the carbonyl functionality required for the quinazolinone ring closure.
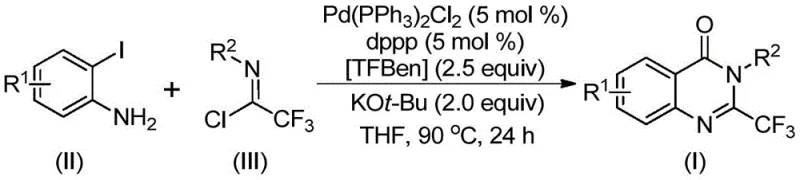
Following the CO insertion, the base promotes the formation of a palladium-nitrogen bond, leading to the generation of a seven-membered ring palladium intermediate. The cycle concludes with a reductive elimination step that releases the final 2-trifluoromethyl substituted quinazolinone derivative and regenerates the active palladium catalyst. Understanding this mechanism is vital for impurity control; by optimizing the ratio of the palladium catalyst, ligand (dppp), and base, side reactions such as homocoupling of the aryl iodide or hydrolysis of the imidoyl chloride can be minimized. The precise stoichiometry, typically maintaining a molar ratio of catalyst to ligand to base at 0.05:0.05:2 relative to the substrate, ensures high conversion rates and simplifies the downstream purification process, resulting in a cleaner crude product profile.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
Implementing this synthesis route requires careful attention to reagent quality and reaction parameters to maximize yield and purity. The process is designed to be operationally simple, utilizing standard laboratory equipment without the need for specialized high-pressure apparatus. The detailed standardized synthesis steps involve mixing the palladium catalyst, ligand, base, solid CO source, and substrates in an aprotic solvent like THF, followed by heating and standard workup procedures. For a comprehensive guide on the exact procedural steps, stoichiometry, and purification techniques validated by our technical team, please refer to the structured protocol below.
- Combine palladium catalyst, dppp ligand, potassium tert-butoxide, TFBen (solid CO source), trifluoroacetimidoyl chloride, and o-iodoaniline in an organic solvent like THF.
- Heat the reaction mixture to 90°C and maintain stirring for 16 to 30 hours to allow the carbonylation tandem reaction to proceed to completion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 2-trifluoromethyl-substituted quinazolinone derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers substantial advantages for procurement managers and supply chain heads focused on cost efficiency and risk mitigation. The shift from gaseous carbon monoxide to a solid surrogate fundamentally alters the safety profile of the manufacturing process, removing the need for expensive gas handling infrastructure and rigorous safety monitoring systems associated with toxic gases. This transition significantly lowers the capital expenditure required for setting up production lines and reduces the ongoing operational costs related to safety compliance and waste disposal. Furthermore, the use of cheap and readily available starting materials like o-iodoaniline and trifluoroacetimidoyl chloride ensures a stable supply chain, minimizing the risk of raw material shortages that could disrupt production schedules.
- Cost Reduction in Manufacturing: The elimination of toxic carbon monoxide gas removes the necessity for specialized high-pressure reactors and complex gas delivery systems, leading to drastic simplification of the plant infrastructure. Additionally, the high atom economy of the tandem reaction and the use of inexpensive, commercially available reagents contribute to substantial cost savings in raw material procurement. The simplified post-treatment process, involving filtration and standard column chromatography, further reduces labor and solvent consumption costs compared to multi-step conventional syntheses.
- Enhanced Supply Chain Reliability: The reliance on stable, solid reagents rather than hazardous gases enhances the overall reliability of the supply chain. Solid surrogates like TFBen are easier to store, transport, and handle than compressed gas cylinders, reducing logistics complexities and potential delays. The broad substrate scope means that a single robust platform technology can be used to produce a wide variety of derivatives, allowing for flexible manufacturing planning and rapid response to changing market demands for different API intermediates.
- Scalability and Environmental Compliance: The mild reaction conditions (90°C) and the absence of high-pressure gas make this process inherently safer and easier to scale from kilogram to tonne quantities. The use of a solid CO source minimizes the release of volatile organic compounds and toxic gases into the environment, aligning with increasingly stringent global environmental regulations. This green chemistry approach not only ensures regulatory compliance but also enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on reaction scope, safety, and operational parameters. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for large-scale production.
Q: What is the primary safety advantage of this synthesis method over traditional carbonylation?
A: This method utilizes 1,3,5-tricarboxylate phenol ester (TFBen) as a solid carbon monoxide surrogate, completely eliminating the need for handling toxic, high-pressure carbon monoxide gas, thereby significantly enhancing operational safety and reducing infrastructure costs.
Q: Does this protocol support diverse functional groups on the aromatic ring?
A: Yes, the patent demonstrates excellent substrate compatibility, successfully tolerating various substituents including halogens (F, Cl, Br), alkyl groups (methyl, tert-butyl), and electron-withdrawing groups (nitro, trifluoromethyl) at ortho, meta, and para positions without significant yield loss.
Q: What are the typical reaction conditions required for this transformation?
A: The reaction typically proceeds in an aprotic solvent such as tetrahydrofuran (THF) at a moderate temperature of 90°C for a duration of 16 to 30 hours, using a palladium catalyst system with dppp ligand and potassium tert-butoxide as the base.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development timelines. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly translated into industrial reality. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to implement complex palladium-catalyzed reactions safely and efficiently positions us as a strategic partner for your most challenging synthesis projects.
We invite you to collaborate with us to leverage this innovative carbonylation technology for your specific pipeline needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this route can optimize your bill of materials. Please contact us today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets, ensuring a secure and efficient supply of critical pharmaceutical building blocks.
