Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial API Production
Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial API Production
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable methodologies for constructing nitrogen-containing heterocycles, particularly the quinazolinone scaffold, which serves as a privileged structure in medicinal chemistry. As illustrated in the diverse bioactive molecules shown below, ranging from sedatives like Methaqualone to antifungal agents, the quinazolinone core is ubiquitous in drug discovery. 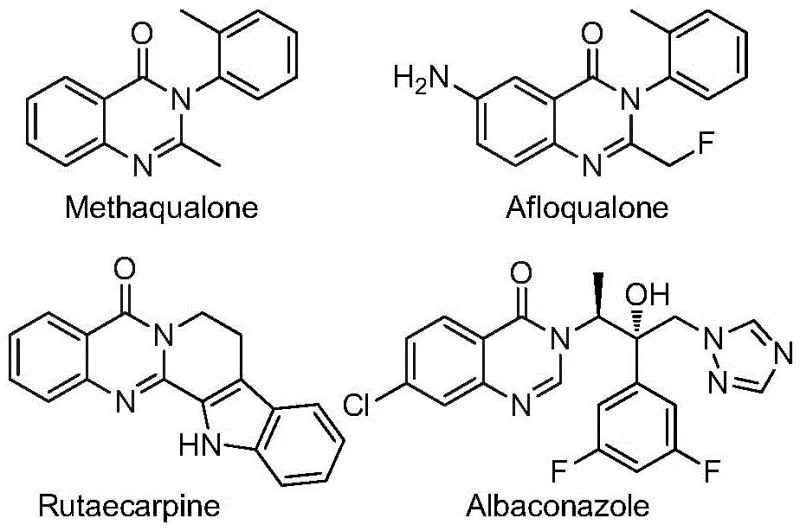 Patent CN112480015B introduces a transformative multi-component one-pot method for synthesizing 2-trifluoromethyl substituted quinazolinones, addressing critical bottlenecks in current manufacturing processes. This innovation leverages a palladium-catalyzed carbonylation cascade that merges cheap nitro compounds with trifluoroethylimidoyl chlorides, offering a streamlined pathway that bypasses the need for hazardous high-pressure carbon monoxide gas. For R&D directors and procurement managers alike, this technology represents a significant leap forward in process efficiency, enabling the rapid generation of complex fluorinated libraries with high atom economy and reduced operational complexity.
Patent CN112480015B introduces a transformative multi-component one-pot method for synthesizing 2-trifluoromethyl substituted quinazolinones, addressing critical bottlenecks in current manufacturing processes. This innovation leverages a palladium-catalyzed carbonylation cascade that merges cheap nitro compounds with trifluoroethylimidoyl chlorides, offering a streamlined pathway that bypasses the need for hazardous high-pressure carbon monoxide gas. For R&D directors and procurement managers alike, this technology represents a significant leap forward in process efficiency, enabling the rapid generation of complex fluorinated libraries with high atom economy and reduced operational complexity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinazolinone ring system has relied on synthetic routes that are fraught with logistical and economic challenges. Traditional methods often necessitate the use of pre-functionalized starting materials, such as 2-bromoformylaniline or 2-iodoaniline derivatives, which are not only expensive to procure but also require additional synthetic steps to prepare, thereby inflating the overall cost of goods. Furthermore, many established protocols depend on the use of gaseous carbon monoxide under high-pressure conditions to facilitate carbonylation, introducing severe safety hazards and requiring specialized, costly reactor infrastructure that limits scalability. Other approaches utilizing iron or ruthenium catalysts frequently suffer from narrow substrate scope, poor functional group tolerance, and low yields, making them unsuitable for the diverse library synthesis required in modern drug discovery. These limitations collectively hinder the ability of supply chain teams to secure reliable, cost-effective sources of high-purity intermediates, often leading to prolonged lead times and supply discontinuities.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in Patent CN112480015B utilizes a sophisticated yet operationally simple palladium-catalyzed system that transforms inexpensive nitro compounds directly into the target heterocycle. As depicted in the general reaction scheme below, the process employs Mo(CO)6 as a solid, safe surrogate for carbon monoxide gas, effectively eliminating the need for high-pressure equipment while maintaining high reaction efficiency. 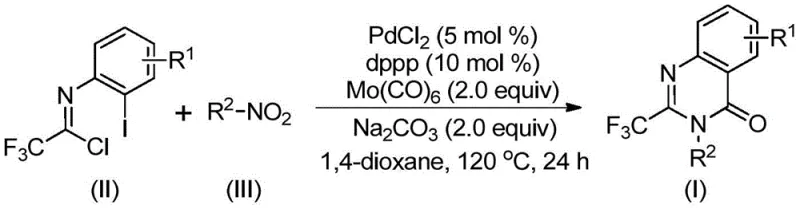 This novel approach allows for the direct coupling of trifluoroethylimidoyl chlorides with a wide variety of nitroarenes in a single pot, significantly reducing waste generation and purification burdens. The use of a PdCl2/dppp catalyst system ensures excellent compatibility with diverse functional groups, including halogens and alkyl chains, enabling the synthesis of a broad spectrum of derivatives without the need for protecting group strategies. This paradigm shift not only simplifies the synthetic workflow but also drastically lowers the barrier to entry for commercial manufacturing, making it an ideal solution for cost reduction in pharmaceutical intermediate manufacturing.
This novel approach allows for the direct coupling of trifluoroethylimidoyl chlorides with a wide variety of nitroarenes in a single pot, significantly reducing waste generation and purification burdens. The use of a PdCl2/dppp catalyst system ensures excellent compatibility with diverse functional groups, including halogens and alkyl chains, enabling the synthesis of a broad spectrum of derivatives without the need for protecting group strategies. This paradigm shift not only simplifies the synthetic workflow but also drastically lowers the barrier to entry for commercial manufacturing, making it an ideal solution for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
The success of this multi-component reaction hinges on a carefully orchestrated catalytic cycle that integrates nitro reduction, amidation, and carbonylative cyclization into a seamless sequence. The mechanism likely initiates with the reduction of the nitro group to an amine by Mo(CO)6, which simultaneously serves as the carbon monoxide source upon thermal decomposition. This in-situ generated amine then undergoes a base-promoted nucleophilic attack on the trifluoroethylimidoyl chloride to form a trifluoroacetamidine intermediate. Subsequently, the palladium catalyst, coordinated by the bidentate dppp ligand, inserts into the carbon-iodine bond of the aromatic ring, forming a reactive organopalladium species. The released carbon monoxide then inserts into the carbon-palladium bond to generate an acyl-palladium intermediate, which is poised for the final cyclization step. Under the influence of the base, an intramolecular nucleophilic attack by the nitrogen atom closes the ring, followed by reductive elimination to release the 2-trifluoromethyl quinazolinone product and regenerate the active palladium catalyst. This intricate interplay of redox and organometallic steps ensures high turnover numbers and minimizes the formation of side products, resulting in a clean impurity profile that is critical for regulatory compliance.
From a quality control perspective, the robustness of this catalytic system provides exceptional control over the impurity spectrum, a key concern for R&D directors overseeing process development. The high selectivity of the Pd/dppp system minimizes homocoupling byproducts and incomplete cyclization species that often plague traditional condensation reactions. Moreover, the tolerance for various substituents on both the nitro compound and the imidoyl chloride allows for the precise tuning of physicochemical properties without compromising yield. As shown in the specific examples below, the method successfully accommodates electron-withdrawing groups like fluorine and chlorine, as well as bulky aryl and cycloalkyl groups, demonstrating remarkable versatility. 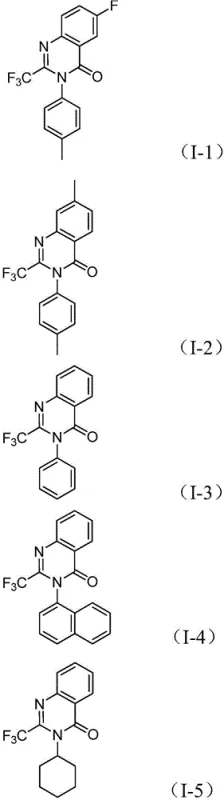 This structural diversity is achieved while maintaining consistent reaction performance, ensuring that the final high-purity pharmaceutical intermediates meet stringent specifications required for downstream biological testing and clinical applications. The ability to predictably manage the reaction outcome across different substrates significantly de-risks the scale-up process.
This structural diversity is achieved while maintaining consistent reaction performance, ensuring that the final high-purity pharmaceutical intermediates meet stringent specifications required for downstream biological testing and clinical applications. The ability to predictably manage the reaction outcome across different substrates significantly de-risks the scale-up process.
How to Synthesize 2-Trifluoromethyl Quinazolinones Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific protocol parameters to maximize yield and purity. The process is designed to be user-friendly, utilizing standard Schlenk techniques and commercially available reagents that do not require specialized handling beyond standard inert atmosphere practices. The reaction typically proceeds in polar aprotic solvents such as 1,4-dioxane, which effectively solubilize both the organic substrates and the inorganic base, facilitating efficient mass transfer. Detailed standardized synthesis steps, including precise molar ratios and workup procedures, are outlined in the structured guide below to ensure reproducibility across different batches and scales.
- Combine palladium chloride, dppp ligand, sodium carbonate, Mo(CO)6, trifluoroethylimidoyl chloride, and nitro compound in an organic solvent like 1,4-dioxane.
- Heat the reaction mixture to 120°C and maintain stirring for 16 to 30 hours to allow for the carbonylation cascade and cyclization to occur.
- Upon completion, filter the mixture, mix with silica gel, and purify the crude product via column chromatography to isolate the target quinazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers tangible strategic benefits that extend far beyond simple chemical transformation. By shifting the starting material basis from expensive, pre-activated amines to commodity-grade nitro compounds, manufacturers can achieve substantial cost savings in raw material acquisition. Nitro compounds are produced on a massive industrial scale globally, ensuring a stable and resilient supply chain that is less susceptible to the volatility often seen with specialized fine chemical intermediates. Furthermore, the elimination of high-pressure carbon monoxide gas removes a significant safety liability and reduces the capital expenditure required for reactor certification and maintenance. This operational simplification translates directly into lower overhead costs and faster turnaround times for production campaigns, enhancing the overall agility of the supply chain.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the replacement of costly reagents with abundant, low-cost alternatives. Utilizing nitro compounds instead of functionalized anilines removes multiple synthetic steps from the upstream supply chain, effectively compressing the value chain and reducing the cumulative cost of goods sold. Additionally, the use of Mo(CO)6 as a solid CO source avoids the logistics and safety costs associated with handling toxic gases, further driving down operational expenses. The high yields reported across a broad substrate scope mean that less raw material is wasted, maximizing the output per batch and optimizing resource utilization for large-scale commercial production.
- Enhanced Supply Chain Reliability: Reliability is paramount in the pharmaceutical industry, and this method strengthens supply security by relying on universally available feedstocks. Since nitro compounds and trifluoroethylimidoyl chlorides are standard catalog items from major chemical suppliers, the risk of supply disruption due to single-source dependency is minimized. The robustness of the reaction conditions also means that production is less sensitive to minor variations in raw material quality, ensuring consistent output even when sourcing from different vendors. This flexibility allows procurement teams to negotiate better terms and maintain continuous inventory levels, safeguarding against market fluctuations and ensuring uninterrupted delivery to downstream clients.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new environmental and safety challenges, but this one-pot method is inherently designed for expansion. The absence of high-pressure gas and the use of standard organic solvents simplify the engineering requirements for moving from gram-scale optimization to multi-kilogram or ton-scale production. From an environmental standpoint, the atom economy of the multi-component reaction is superior to stepwise approaches, generating less chemical waste and reducing the burden on waste treatment facilities. This alignment with green chemistry principles not only lowers disposal costs but also supports corporate sustainability goals, making the process attractive for environmentally conscious stakeholders and regulatory bodies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this method compares to existing standards. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of using nitro compounds over traditional amines in this synthesis?
A: Nitro compounds are significantly cheaper and more readily available than their corresponding amine precursors. Furthermore, this method eliminates the need for pre-activation steps often required for amines, streamlining the supply chain and reducing raw material costs.
Q: How does this method address safety concerns regarding carbon monoxide usage?
A: Traditional carbonylation often requires high-pressure CO gas cylinders, which pose significant safety risks. This patented method utilizes solid Mo(CO)6 as a safe, in-situ CO source, drastically improving operational safety and simplifying reactor requirements for scale-up.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the patent explicitly demonstrates that the method can be expanded to the gram level with high efficiency. The use of robust catalysts and standard solvents like dioxane ensures that the process is adaptable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative academic research into reliable commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent literature are faithfully reproduced at an industrial level. We are committed to delivering high-purity 2-trifluoromethyl quinazolinones that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our expertise in palladium-catalyzed carbonylation allows us to navigate the complexities of this chemistry efficiently, guaranteeing consistent quality and supply continuity for our global partners.
We invite potential collaborators to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits tailored to your volume needs. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
