Scalable One-Pot Synthesis of Isoprispirin Derivatives for Commercial Antitumor Drug Development
The pharmaceutical industry is constantly seeking robust and scalable synthetic routes for complex marine alkaloid analogs, particularly those exhibiting potent antitumor properties. Patent CN112225745B, titled "A kind of isoprispirin compound with antitumor activity, preparation method and use," introduces a groundbreaking methodology for the construction of isoprispirin scaffolds. This patent details a novel iodine-catalyzed, one-pot tandem reaction that efficiently couples substituted o-acetphenoxy acrylates with tetrahydroisoquinolines or tetrahydropyridoindoles. Unlike traditional multi-step syntheses that often suffer from low overall yields and cumbersome purification protocols, this innovative approach leverages elemental iodine as a mild oxidant to drive the cyclization process under acidic conditions. The significance of this technology lies in its ability to generate structurally diverse isoprispirin derivatives, which are critical intermediates in the development of next-generation anticancer therapeutics, with high atom economy and operational simplicity.
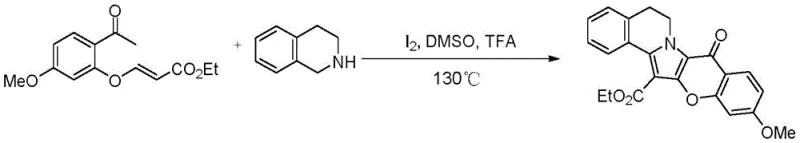
For R&D directors and process chemists, understanding the evolution of synthetic strategies for lamellarin-type alkaloids is crucial for optimizing supply chains. Historically, the construction of the pyrrole-2-one core found in isoprispirins has been challenging. Early methodologies, such as those reported by Thasana's group, relied on copper-catalyzed microwave-assisted reactions to build the isoprispirin A skeleton. While effective on a small scale, the reliance on transition metals like copper introduces significant regulatory hurdles for pharmaceutical applications due to strict limits on heavy metal residues in active pharmaceutical ingredients (APIs). Furthermore, microwave assistance, while rapid, often presents difficulties in linear scale-up for large-scale industrial reactors.
Subsequent advancements by Yang's research group utilized base-catalyzed [3+2] cycloaddition reactions between 4-chloro-3-formylcoumarins and tetrahydroisoquinolines. Although this represented a shift away from microwave dependence, it still required specific halogenated precursors and strong bases, which can limit substrate scope and generate hazardous waste streams. In stark contrast, the novel approach detailed in patent CN112225745B utilizes a metal-free (regarding transition metals) oxidative cyclization strategy. By employing elemental iodine in conjunction with organic acids like trifluoroacetic acid (TFA), the reaction proceeds smoothly in polar aprotic solvents such as DMSO at moderate temperatures around 130°C. This shift not only broadens the functional group tolerance but also drastically simplifies the workup procedure, as the absence of transition metals eliminates the need for specialized scavenging resins or complex chelation steps during purification.
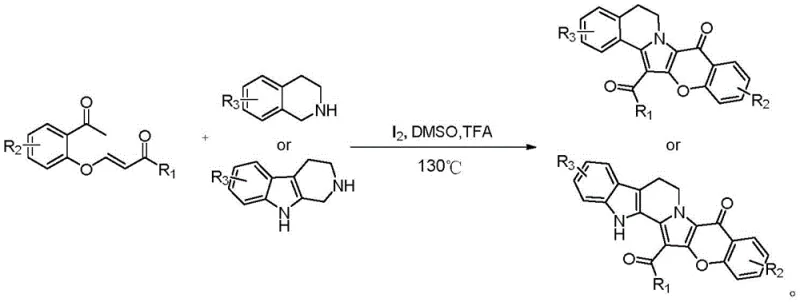
The mechanistic insights into this iodine-catalyzed transformation reveal a sophisticated interplay of electrophilic activation and oxidative aromatization. The reaction initiates with the activation of the enolizable ketone moiety in the o-acetphenoxy acrylate precursor by the acidic medium. Elemental iodine acts as a soft electrophile, facilitating the formation of an iodonium intermediate or promoting single-electron transfer processes that activate the nucleophilic nitrogen of the tetrahydroisoquinoline ring. This triggers an intramolecular cyclization cascade where the carbon-nitrogen bond is formed concurrently with the oxidation of the dihydro-pyridine ring to the fully aromatic pyridinium or pyrrole system, depending on the specific substrate. The presence of the acid catalyst is paramount; it not only protonates intermediate species to enhance their electrophilicity but also helps in the final elimination of HI to restore aromaticity. This self-organized one-pot sequence effectively merges multiple bond-forming events—C-N coupling, C-O bond retention, and oxidative dehydrogenation—into a single operational step.
From an impurity control perspective, this mechanism offers distinct advantages. Traditional stepwise syntheses often accumulate byproducts from each isolation stage, leading to complex impurity profiles that are difficult to characterize and remove. The concerted nature of this iodine-mediated cyclization minimizes the lifetime of reactive intermediates, thereby suppressing side reactions such as polymerization or over-oxidation. The primary byproducts are typically inorganic salts and reduced iodine species, which are easily removed during the aqueous workup and subsequent column chromatography. This results in a cleaner crude product profile, significantly reducing the burden on quality control laboratories and ensuring that the final pharmaceutical intermediates meet stringent purity specifications required for clinical trial material production.
How to Synthesize Isoprispirin Derivatives Efficiently
The practical implementation of this synthesis route is designed for maximum efficiency and reproducibility in a GMP-compliant environment. The protocol involves dissolving the substituted o-acetphenoxy acrylate and the amine component (tetrahydroisoquinoline or tetrahydropyridoindole) in a solvent system such as dimethyl sulfoxide (DMSO). To this mixture, stoichiometric amounts of elemental iodine and a Brønsted acid like trifluoroacetic acid are added. The reaction vessel is then sealed and heated to approximately 130°C for a duration of 8 to 15 hours. Upon completion, the reaction mixture undergoes a standard extractive workup followed by purification via silica gel chromatography. The detailed standardized synthesis steps, including precise molar ratios and safety precautions for handling iodine vapors, are outlined below.
- Mix substituted o-acetphenoxy acrylate and tetrahydroisoquinoline derivatives in an organic solvent like DMSO.
- Add elemental iodine and an acid catalyst such as trifluoroacetic acid to the reaction mixture.
- Heat the closed vessel to 130°C for 10 hours to facilitate the one-pot oxidative cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in patent CN112225745B translates directly into tangible operational efficiencies and risk mitigation. The most significant advantage is the drastic simplification of the manufacturing process. By consolidating what would traditionally be a multi-step sequence into a single one-pot reaction, the number of unit operations is significantly reduced. This reduction eliminates the need for intermediate isolation, drying, and re-dissolution steps, which are often the most time-consuming and labor-intensive parts of chemical manufacturing. Consequently, the overall cycle time for producing these high-value antitumor intermediates is compressed, allowing for faster response to market demands and shorter lead times for custom synthesis projects.
- Cost Reduction in Manufacturing: The economic impact of this synthetic route is profound. By removing the requirement for expensive transition metal catalysts such as palladium, copper, or rhodium, the direct material costs are lowered. More importantly, the elimination of heavy metals removes the necessity for costly purification technologies like preparative HPLC with metal-scavenging columns or extensive recrystallization processes designed to meet ICH Q3D guidelines for elemental impurities. The use of commodity chemicals like iodine and TFA, which are inexpensive and readily available in bulk quantities, further stabilizes the cost of goods sold (COGS). Additionally, the high yields reported in the patent examples, often exceeding 70-80%, ensure better material throughput and less waste generation, contributing to substantial cost savings in raw material procurement.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of robust and widely available starting materials. The substrates required for this reaction, such as substituted phenols and acrylates, are common building blocks in the fine chemical industry with established global supply networks. This reduces the risk of supply disruptions that often plague projects relying on exotic or proprietary reagents. Furthermore, the reaction conditions are relatively mild and do not require specialized high-pressure equipment or cryogenic cooling, meaning the process can be executed in standard glass-lined or stainless steel reactors available at most contract development and manufacturing organizations (CDMOs). This flexibility allows for easier technology transfer between sites and ensures continuity of supply even if one manufacturing location faces operational challenges.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method aligns well with green chemistry principles. The one-pot nature inherently reduces solvent consumption and waste generation compared to multi-step linear syntheses. The avoidance of toxic heavy metals simplifies wastewater treatment and disposal protocols, lowering the environmental compliance burden. The reaction generates minimal hazardous byproducts, primarily inorganic salts, which are easier to manage than complex organic waste streams. This eco-friendly profile not only reduces disposal costs but also enhances the sustainability credentials of the final pharmaceutical product, a factor that is increasingly important for major pharmaceutical buyers who are committed to reducing their carbon footprint and environmental impact.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these isoprispirin derivatives. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for potential partners and licensees.
Q: What are the advantages of the iodine-catalyzed method over traditional copper-catalyzed routes?
A: The iodine-catalyzed method described in patent CN112225745B eliminates the need for expensive transition metal catalysts like copper, reducing heavy metal residue concerns and simplifying downstream purification processes for pharmaceutical grade intermediates.
Q: Can this synthesis method be scaled for commercial production?
A: Yes, the one-pot nature of the reaction, combined with mild conditions (130°C) and commercially available starting materials, makes it highly suitable for scale-up from laboratory to multi-ton commercial manufacturing without complex intermediate isolation steps.
Q: What is the biological activity profile of these isoprispirin derivatives?
A: The synthesized compounds exhibit significant antitumor activity, specifically demonstrating inhibitory effects against human oral epidermoid cancer cells (KBV) and human gastric cancer cells (MKN-45), making them valuable lead compounds for oncology drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isoprispirin Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the iodine-catalyzed synthesis route for isoprispirin compounds in the field of oncology drug discovery. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate this patented laboratory methodology into a robust commercial manufacturing process. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from preclinical research to clinical supply and beyond. We are equipped with state-of-the-art rigorous QC labs and analytical instruments capable of verifying stringent purity specifications, guaranteeing that every batch of isoprispirin intermediate meets the highest international standards for pharmaceutical use.
We invite you to collaborate with us to leverage this advanced synthetic technology for your antitumor drug pipeline. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Please contact us today to request specific COA data for our reference standards and comprehensive route feasibility assessments. Let us help you accelerate your drug development program with reliable, high-quality, and cost-effective isoprispirin derivatives manufactured with precision and care.
