Scalable Synthesis of Antitumor Isoprispirin Derivatives via Metal-Free Iodine Catalysis
Scalable Synthesis of Antitumor Isoprispirin Derivatives via Metal-Free Iodine Catalysis
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for greener, more efficient, and cost-effective synthetic routes. A significant breakthrough in this domain is documented in Chinese Patent CN112225745A, which introduces a novel methodology for synthesizing isoprispirin compounds with potent antitumor activity. This patent outlines a transformative approach that utilizes elemental iodine as a catalyst to construct complex heterocyclic scaffolds traditionally associated with marine alkaloids like Lamellarins. For R&D directors and procurement specialists alike, this development represents a critical opportunity to access high-value anticancer leads through a streamlined, metal-free process that aligns with modern sustainability goals and regulatory standards for impurity control.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the isoprispirin core structure has relied heavily on transition metal catalysis, often involving copper or other heavy metals under rigorous conditions. Previous literature, such as the work by Thasana's group in 2007, utilized copper-catalyzed microwave-assisted methods to achieve the skeleton construction. While effective on a small laboratory scale, these conventional pathways present substantial hurdles for industrial application. The reliance on transition metals introduces significant downstream processing challenges, including the absolute necessity for rigorous metal scavenging steps to meet International Council for Harmonisation (ICH) guidelines on elemental impurities. Furthermore, multi-step sequences requiring intermediate isolation increase solvent consumption, waste generation, and overall production time, thereby inflating the cost of goods sold (COGS) and complicating supply chain logistics for reliable pharmaceutical intermediate supplier operations.
The Novel Approach
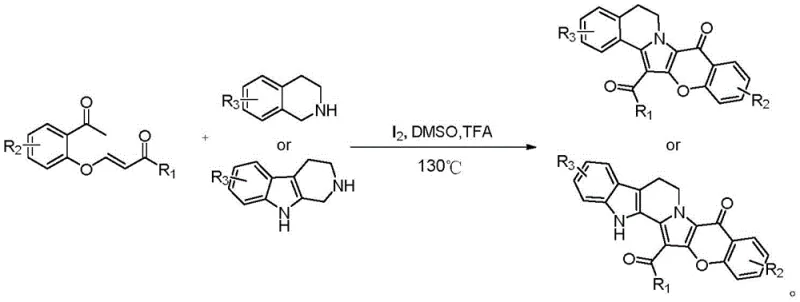
In stark contrast, the methodology disclosed in CN112225745A offers a paradigm shift by employing a self-organized one-pot synthesis driven by elemental iodine. This innovative route allows for the direct coupling of substituted o-acetphenoxy acrylate compounds with tetrahydroisoquinoline or tetrahydropyridoindole derivatives. The reaction proceeds efficiently under acidic conditions without the need for isolating unstable intermediates, effectively collapsing multiple synthetic steps into a single operational unit. This telescoping of reactions not only drastically simplifies the workflow but also enhances the overall atom economy. By eliminating the requirement for toxic heavy metal catalysts, the process inherently reduces the environmental footprint and removes the burden of extensive purification protocols, positioning this technology as a superior choice for cost reduction in API manufacturing.
Mechanistic Insights into Iodine-Catalyzed Oxidative Cyclization
The core of this technological advancement lies in the unique mechanistic role of elemental iodine, which functions dually as an oxidant and a Lewis acid catalyst to facilitate the oxidative cyclization. In this sophisticated transformation, the iodine species activates the electron-rich aromatic systems and the alkene moieties within the acrylate precursors, promoting an intramolecular nucleophilic attack that closes the pyrrole ring system characteristic of the isoprispirin scaffold. The presence of an acid promoter, such as trifluoroacetic acid, further enhances the electrophilicity of the reaction centers, ensuring high conversion rates even at moderate temperatures around 130°C. This mechanistic pathway avoids the formation of radical species often associated with harsher oxidative conditions, thereby minimizing the generation of complex polymeric byproducts and ensuring a cleaner reaction profile that is easier to manage during scale-up.
From an impurity control perspective, this mechanism offers distinct advantages for quality assurance teams. The absence of transition metals means that the primary impurities are organic in nature and can be effectively managed through standard crystallization or chromatography techniques, rather than requiring specialized chelating resins. The mild acidic environment helps to suppress side reactions such as hydrolysis of the ester groups, which is a common issue in base-catalyzed alternatives. Consequently, the resulting crude product typically exhibits higher purity, reducing the load on final purification steps. This robustness in impurity profiling is critical for maintaining batch-to-batch consistency, a key metric for any commercial scale-up of complex pharmaceutical intermediates where regulatory compliance is non-negotiable.
How to Synthesize Isoprispirin Derivatives Efficiently
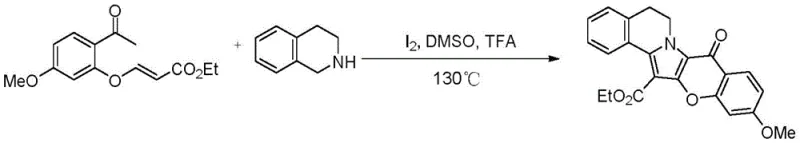
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and purity. The process begins with the precise stoichiometric mixing of the acrylate precursor and the amine component in a polar aprotic solvent like dimethyl sulfoxide (DMSO). The addition of the iodine catalyst and acid promoter must be controlled to initiate the cascade reaction smoothly. As demonstrated in the patent examples, maintaining a sealed environment is crucial to prevent solvent loss and maintain the necessary pressure for the reaction to proceed to completion over a period of approximately 10 hours. The following guide outlines the standardized operational steps derived from the patent data to ensure reproducible results in a pilot or production setting.
- Mix substituted o-acetphenoxy acrylate and tetrahydroisoquinoline derivatives in an organic solvent like DMSO.
- Add elemental iodine catalyst and an acid promoter such as trifluoroacetic acid to the reaction mixture.
- Heat the sealed reaction vessel to 130°C for approximately 10 hours to facilitate oxidative cyclization and isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iodine-catalyzed protocol translates into tangible strategic benefits beyond mere chemical elegance. The elimination of expensive transition metal catalysts directly impacts the raw material cost structure, removing the volatility associated with precious metal pricing. Moreover, the simplification of the purification train reduces the consumption of auxiliary materials such as scavengers and specialized filtration media. This streamlining of the manufacturing process leads to substantial cost savings by shortening the overall cycle time and reducing the labor hours required for monitoring and handling multiple intermediate stages. The ability to source cheap and readily available starting materials further enhances the economic viability of this route, making it an attractive option for long-term supply contracts.
- Cost Reduction in Manufacturing: The removal of transition metal catalysts eliminates the need for costly metal scavenging resins and extensive analytical testing for residual metals, which are significant cost drivers in traditional pharmaceutical synthesis. By utilizing inexpensive elemental iodine and common acids, the direct material costs are significantly lowered. Additionally, the one-pot nature of the reaction reduces solvent usage and energy consumption associated with heating and cooling cycles for multiple steps, leading to a leaner and more cost-efficient production model that improves overall margin potential.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, such as substituted phenols and tetrahydroisoquinolines, are commodity chemicals with robust global supply chains, reducing the risk of bottlenecks associated with specialized reagents. The simplicity of the reaction conditions means that the process can be easily transferred between different manufacturing sites without requiring highly specialized equipment or expertise. This flexibility ensures continuity of supply and allows for rapid scaling to meet market demand fluctuations, providing a secure foundation for reliable pharmaceutical intermediate supplier partnerships.
- Scalability and Environmental Compliance: The process operates under relatively mild thermal conditions and avoids the use of toxic heavy metals, aligning perfectly with green chemistry principles and stringent environmental regulations. The reduction in hazardous waste generation simplifies waste disposal logistics and lowers compliance costs. Furthermore, the high efficiency and selectivity of the reaction minimize the formation of difficult-to-separate byproducts, facilitating easier scale-up from kilogram to tonnage production while maintaining high quality standards and minimizing the environmental impact of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical implementation and potential applications of these novel compounds in drug discovery pipelines.
Q: What are the advantages of using iodine over transition metal catalysts for isoprispirin synthesis?
A: Using elemental iodine eliminates the need for expensive and toxic transition metals like copper, simplifying purification processes and ensuring compliance with strict residual metal limits in pharmaceutical intermediates.
Q: Can this synthesis method be scaled for commercial production?
A: Yes, the one-pot tandem reaction operates under relatively mild conditions without the need for intermediate isolation, making it highly suitable for scale-up and continuous manufacturing processes.
Q: What is the biological activity profile of these isoprispirin derivatives?
A: The synthesized compounds exhibit significant antitumor activity, specifically showing inhibition against KBV oral epidermoid cancer cells and MKN-45 gastric cancer cell lines.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isoprispirin Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of this iodine-catalyzed technology in advancing oncology research and development. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering high-purity isoprispirin derivatives that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee batch consistency and regulatory compliance for your critical projects.
We invite you to collaborate with us to leverage this cutting-edge synthetic route for your antitumor drug development programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your timeline to market while optimizing your overall production costs.
