Revolutionizing Aminothiophene Production: A Scalable, High-Yield Copper-Catalyzed Route for Pharmaceutical Intermediates
Introduction to Advanced Aminothiophene Synthesis
The landscape of pharmaceutical and agrochemical intermediate manufacturing is undergoing a significant transformation driven by the need for safer, more efficient synthetic routes. Patent CN116023359A introduces a groundbreaking methodology for the preparation of aminothiophene compounds, a critical structural motif found in numerous bioactive molecules. This technology pivots away from the historically hazardous nitration-reduction sequences, opting instead for a direct, copper-catalyzed amination strategy. By leveraging a specific catalytic system involving cuprous salts and a carefully selected solvent environment, this invention addresses long-standing challenges in regioselectivity and process safety. For R&D directors and procurement specialists, understanding this shift is vital, as it represents a move towards more sustainable and cost-effective pharmaceutical intermediates production. The core innovation lies in the ability to directly substitute a bromine atom on the thiophene ring with an amino group under relatively mild conditions, thereby streamlining the supply chain for high-value heterocyclic building blocks.
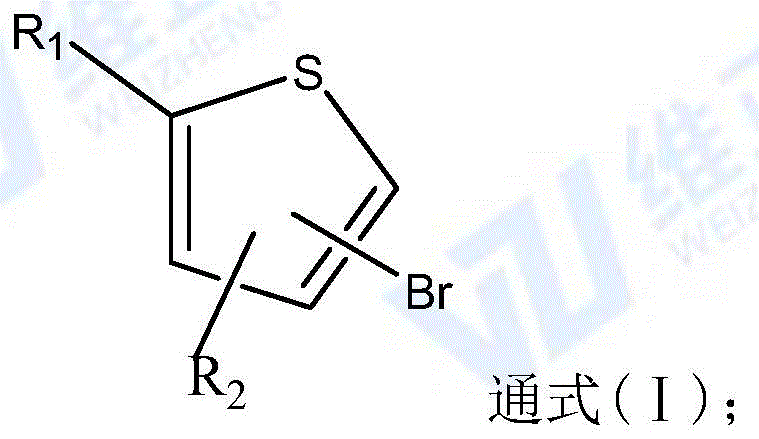
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of aminothiophene derivatives has relied heavily on classical electrophilic aromatic substitution followed by reduction. As illustrated in the comparative data within the patent, the conventional route typically involves the nitration of thiophene carboxylic acids using fuming nitric acid and acetic anhydride at cryogenic temperatures. This approach is fraught with significant drawbacks that impact both operational safety and economic efficiency. Firstly, the nitration step is notoriously non-selective, often generating a complex mixture of regioisomers, such as 4-nitro and 5-nitro derivatives, which are difficult and expensive to separate. Secondly, the subsequent reduction of the nitro group usually requires stoichiometric amounts of metal powders like iron in acidic media, generating substantial quantities of hazardous waste. The cumulative yield of such multi-step processes is often compromised; for instance, the isolation of specific nitro-intermediates can suffer from yields as low as 19.3%, creating a bottleneck that drastically inflates the cost of goods sold (COGS) for the final active pharmaceutical ingredient (API).

The Novel Approach
In stark contrast, the novel methodology disclosed in CN116023359A employs a transition-metal catalyzed cross-coupling strategy that bypasses the nitration step entirely. By utilizing a compound of general formula (I), where a bromine atom serves as the leaving group, the process achieves direct amination using ammonia. This route operates under nitrogen protection with cuprous oxide (Cu2O) as the catalyst and dioxane as the solvent. The simplicity of this one-pot transformation is a game-changer for cost reduction in pharmaceutical intermediates manufacturing. Instead of dealing with explosive nitrating agents and heavy metal sludge from reductions, manufacturers can utilize a cleaner catalytic cycle. The reaction conditions are remarkably mild, typically requiring temperatures between 80-110°C, which reduces energy consumption and equipment stress. Furthermore, the selectivity is inherent to the starting material; since the bromine position is predefined in the precursor, there is no risk of forming unwanted regioisomers, ensuring a much cleaner reaction profile and simplifying downstream purification.
Mechanistic Insights into Copper-Catalyzed Amination
To fully appreciate the technical superiority of this method, one must delve into the mechanistic underpinnings of the copper-catalyzed amination. The reaction proceeds through a classic organometallic cycle involving coordination, oxidative addition, and reductive elimination. Initially, the ammonia molecule coordinates with the copper(I) catalyst to form an active amido-copper species. This nucleophilic complex then undergoes oxidative addition with the carbon-bromine bond of the thiophene substrate. This step is critical and is facilitated by the electron-rich nature of the copper center, which activates the aryl halide bond. Following this, the system undergoes reductive elimination, releasing the desired aminothiophene product and regenerating the copper catalyst for the next cycle. This catalytic turnover is highly efficient, allowing for the use of relatively low catalyst loadings while maintaining high conversion rates. The choice of ligand-free conditions or simple salt catalysts like Cu2O further enhances the economic viability by eliminating the need for expensive, specialized phosphine ligands often required in palladium-catalyzed counterparts.
A pivotal aspect of this mechanism is the role of the solvent, which acts as more than just a medium for the reaction. The patent data explicitly highlights the superiority of dioxane over other common polar aprotic solvents like acetonitrile or tetrahydrofuran (THF). Mechanistically, dioxane appears to optimize the solubility of both the organic thiophene substrate and the inorganic ammonia source, creating a homogeneous or near-homogeneous phase that maximizes collision frequency between reactants. In comparative experiments, switching to acetonitrile resulted in a precipitous drop in yield from approximately 70% to 40%. This suggests that in acetonitrile, the catalyst may be less stable or the substrate less accessible, leading to competing side reactions or incomplete conversion. By maintaining the reaction in dioxane, the system ensures that the oxidative addition step proceeds rapidly and that the catalyst remains active throughout the 1-4 hour reaction window, ultimately delivering products with purity levels exceeding 97%.
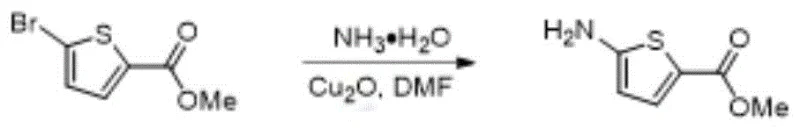
How to Synthesize 5-Amino-2-thiophenecarboxylic Acid Methyl Ester Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and safety. The process is designed to be robust, accommodating various substituents on the thiophene ring while maintaining high efficiency. For technical teams looking to replicate the results for high-purity aminothiophene production, the key lies in the precise control of the ammonia concentration and the exclusion of oxygen. The following guide outlines the standardized operational procedure derived from the most successful embodiments of this technology.
- Prepare the reaction mixture by combining the bromothiophene substrate, cuprous oxide catalyst, and dioxane solvent under nitrogen protection.
- Add the ammonia-dioxane solution dropwise and maintain the temperature between 80-110°C with stirring for 1-4 hours.
- Quench the reaction with ethyl acetate, filter through silica gel, and purify the filtrate via extraction and column chromatography to obtain the high-purity aminothiophene product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed amination technology offers tangible strategic benefits that extend beyond mere chemical elegance. The primary advantage is the substantial simplification of the manufacturing workflow. By collapsing a multi-step nitration-reduction sequence into a single catalytic step, the overall process time is drastically reduced, and the number of unit operations is minimized. This consolidation directly translates to lower capital expenditure (CAPEX) requirements for reactor volume and reduced operational expenditure (OPEX) related to labor and utilities. Furthermore, the elimination of hazardous nitration chemistry significantly lowers the regulatory burden and insurance costs associated with handling explosive intermediates, making the supply chain more resilient and compliant with increasingly stringent environmental, health, and safety (EHS) regulations.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound due to the removal of expensive and wasteful reagents. Traditional methods consume large quantities of fuming nitric acid, acetic anhydride, and metal powders for reduction, all of which generate costly waste streams that require treatment. In contrast, the new method utilizes inexpensive cuprous oxide and ammonia, with dioxane serving as a recoverable solvent. The higher yields (consistently above 68% and up to 80%) mean that less raw material is wasted per kilogram of product, directly improving the material cost basis. Additionally, the high purity of the crude product reduces the load on purification columns, saving on silica gel and eluent costs, which are often hidden but significant expenses in fine chemical production.
- Enhanced Supply Chain Reliability: Reliability in the supply of critical intermediates is paramount for downstream API manufacturers. The robustness of this catalytic system ensures consistent batch-to-batch quality, with purity specifications reliably meeting or exceeding 97%. The use of stable, commercially available starting materials like bromothiophene esters mitigates the risk of supply disruptions often associated with specialized nitro-compounds. Moreover, the milder reaction conditions reduce the likelihood of thermal runaways or equipment corrosion, leading to fewer unplanned shutdowns and maintenance delays. This stability allows suppliers to offer more predictable lead times, a crucial factor for clients managing just-in-time inventory systems for their own production lines.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the ton-scale often exposes hidden inefficiencies, but this method is inherently scalable. The exotherm of the amination reaction is manageable compared to the violent exotherms of nitration, allowing for safer operation in large reactors. From an environmental perspective, the atom economy is superior, and the E-factor (mass of waste per mass of product) is significantly lower. The absence of heavy metal sludge from iron reductions simplifies wastewater treatment, aligning with green chemistry principles. This environmental compatibility not only reduces disposal costs but also enhances the sustainability profile of the final product, a growing requirement for multinational corporations aiming to meet their carbon neutrality and ESG (Environmental, Social, and Governance) goals.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology for their specific applications, we have compiled answers to common inquiries regarding the process parameters and scope. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, ensuring accuracy and relevance for industrial implementation.
Q: Why is dioxane preferred over acetonitrile for this amination reaction?
A: Patent data indicates that dioxane significantly improves the solubility of thiophene compounds and facilitates better interaction with the ammonia source, resulting in yields exceeding 68% compared to roughly 40% when using acetonitrile.
Q: What are the safety advantages of this method over traditional nitration?
A: This copper-catalyzed route eliminates the need for hazardous fuming nitric acid and high-risk reduction steps, operating under milder temperatures (80-110°C) and avoiding the formation of dangerous nitro-isomer byproducts.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the process demonstrates robust yields (68-80%) and high purity (>97%) across multiple substrates, utilizing common catalysts like Cu2O and standard solvents, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aminothiophene Supplier
The technological advancements detailed in CN116023359A represent a significant leap forward in the synthesis of heterocyclic amines, yet translating patent potential into commercial reality requires experienced partnership. NINGBO INNO PHARMCHEM stands ready to leverage this cutting-edge chemistry to support your drug development pipelines. As a premier CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of aminothiophene intermediate meets the exacting standards required for global pharmaceutical registration.
We invite you to explore how this efficient synthesis route can optimize your supply chain and reduce your overall project costs. Our technical team is prepared to conduct a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data for our aminothiophene portfolio and to discuss route feasibility assessments for your custom synthesis projects. Let us collaborate to bring your next-generation therapeutics to market faster and more efficiently.
