Scalable Synthesis of Fluorinated Norcantharidin Derivatives for Next-Generation Oncology Therapeutics
The pharmaceutical industry is constantly seeking novel scaffolds that balance potent biological activity with manageable toxicity profiles, particularly in the realm of oncology. Patent CN111362962B introduces a significant advancement in this field by disclosing a series of norcantharidin carboxylate tetrafluorobenzyl esters. These compounds represent a strategic structural modification of the classic norcantharidin core, integrating both a carboxyl group and fluorine-containing moieties to optimize pharmacokinetic properties. The general structure, as depicted in the patent, highlights the versatility of this chemical space, allowing for various isomeric substitutions on the benzyl ring.
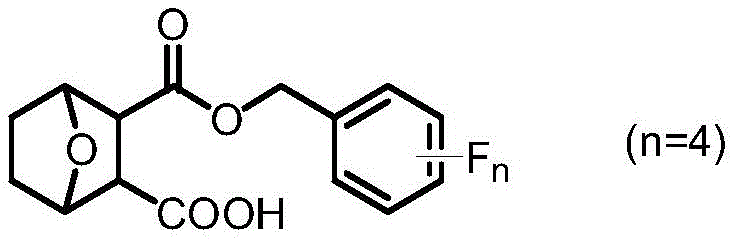
For R&D directors evaluating new lead compounds, the introduction of the tetrafluorobenzyl group is particularly compelling. Fluorine atoms possess high electronegativity and a small atomic radius, which can dramatically influence the binding affinity and metabolic stability of drug candidates. This patent specifically targets liver cancer applications, addressing a critical unmet medical need where current therapies often face resistance or severe side effects. As a reliable pharmaceutical intermediate supplier, understanding the synthetic accessibility of such complex fluorinated molecules is paramount for translating bench-scale discoveries into viable clinical candidates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional cantharidin-based therapies have long been hindered by their narrow therapeutic index. While cantharidin exhibits strong anti-tumor activity, its severe toxicity towards the urinary and digestive systems limits its clinical utility. Even norcantharidin, a less toxic synthetic derivative, suffers from a short residence time in the human body, requiring precise dosing to avoid adverse reactions. Furthermore, conventional synthesis methods for modifying the norcantharidin scaffold often involve harsh conditions or multi-step sequences that degrade the sensitive anhydride ring, leading to low overall yields and difficult purification processes. These factors collectively increase the cost of goods and complicate the supply chain for large-scale manufacturing.
The Novel Approach
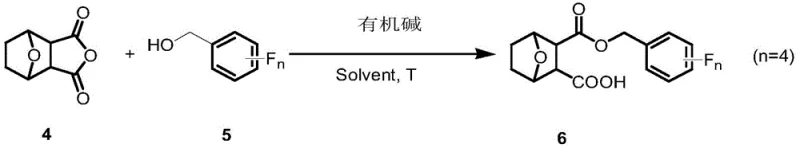
The methodology described in CN111362962B offers a streamlined solution by leveraging a direct ring-opening esterification strategy. Instead of complex protection-deprotection sequences, the process utilizes the inherent reactivity of the norcantharidin anhydride. By reacting norcantharidin directly with tetrafluorobenzyl alcohols in the presence of an organic base, the synthesis achieves selective mono-esterification. This approach not only preserves the integrity of the remaining carboxylic acid group, which enhances water solubility, but also incorporates the bioisosteric fluorine motif in a single efficient step. This represents a significant leap forward in cost reduction in API manufacturing, as it minimizes unit operations and waste generation.
Mechanistic Insights into Anhydride Ring-Opening Esterification
The core chemical transformation in this patent involves the nucleophilic attack of a tetrafluorobenzyl alcohol on one of the carbonyl carbons of the norcantharidin anhydride ring. This reaction is catalyzed by organic bases such as 4-dimethylaminopyridine (4-DMAP), which acts as a potent nucleophilic catalyst. The mechanism proceeds through the formation of an acyl-pyridinium intermediate, which is highly susceptible to attack by the alcohol. This pathway is favored over non-catalyzed thermal reactions because it operates under milder conditions, typically around 60°C, preventing the degradation of the sensitive bicyclic framework. The selectivity for mono-esterification is crucial, as it generates the desired carboxylate functionality that improves the drug-like properties of the final molecule.
From an impurity control perspective, this mechanism offers distinct advantages. The use of specific isomers of tetrafluorobenzyl alcohol (such as 2,3,4,5- or 2,3,4,6-substituted variants) allows for fine-tuning of the steric and electronic environment around the ester linkage. This precision reduces the formation of regioisomeric by-products that are common in less controlled esterification processes. Additionally, the reaction solvent system, preferably dichloromethane (DCM), ensures good solubility of both the polar anhydride and the fluorinated alcohol, facilitating homogeneous reaction kinetics. For procurement managers, understanding that the reaction relies on standard organic bases and common solvents means that sourcing raw materials is straightforward and does not require exotic or hazardous reagents that could disrupt supply continuity.
How to Synthesize Norcantharidin Tetrafluorobenzyl Esters Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing high-purity intermediates suitable for preclinical evaluation. The process begins with the preparation of the norcantharidin core, followed by the critical esterification step. Detailed operational parameters, including stoichiometry and temperature control, are essential for maximizing yield and minimizing impurities. The following guide summarizes the standardized synthesis steps derived from the patent examples, ensuring reproducibility for commercial scale-up of complex pharmaceutical intermediates.
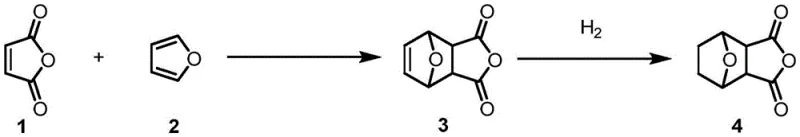
- Prepare the norcantharidin core (Compound 4) via Diels-Alder reaction of furan and maleic anhydride followed by catalytic hydrogenation.
- React norcantharidin with specific tetrafluorobenzyl alcohol isomers (Compound 5) in an organic solvent like DCM.
- Catalyze the ring-opening esterification using an organic base such as 4-DMAP at 60°C to yield the target mono-ester mono-acid derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain heads and procurement managers, the viability of a new chemical entity often hinges on the reliability and cost-efficiency of its production route. The synthesis method described in this patent excels in these areas by utilizing commodity feedstocks and avoiding complex catalytic systems. The starting materials, including furan and maleic anhydride for the core, and various fluorinated benzyl alcohols for the side chain, are widely available from global chemical suppliers. This abundance mitigates the risk of raw material shortages and price volatility, ensuring a stable supply chain for long-term project planning.
- Cost Reduction in Manufacturing: The synthetic route eliminates the need for expensive transition metal catalysts or cryogenic conditions, which are often cost drivers in fine chemical synthesis. By operating at moderate temperatures (60°C) and using recyclable organic solvents, the process significantly lowers energy consumption and waste disposal costs. Furthermore, the high atom economy of the direct esterification step means that fewer raw materials are wasted, directly contributing to substantial cost savings in the overall manufacturing budget without compromising product quality.
- Enhanced Supply Chain Reliability: The reliance on established chemical transformations, such as the Diels-Alder reaction for the core and standard esterification for functionalization, ensures that the process is robust and easily transferable between manufacturing sites. The use of common reagents like DMAP and DCM means that there is no dependency on single-source suppliers for specialized catalysts. This diversification of the supply base enhances resilience against market disruptions, allowing for consistent delivery schedules and reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory synthesis to multi-kilogram pilot production. The absence of heavy metals simplifies the purification process and reduces the environmental burden associated with metal residue removal. This aligns with modern green chemistry principles and regulatory expectations, facilitating smoother regulatory filings. The ability to produce these compounds with stringent purity specifications using standard equipment makes them an attractive option for CDMOs looking to expand their oncology portfolio.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these fluorinated norcantharidin derivatives. These insights are derived directly from the technical disclosures within the patent, providing clarity on the feasibility and advantages of this synthetic approach for potential partners and licensees.
Q: What is the primary advantage of introducing fluorine into norcantharidin derivatives?
A: Introducing fluorine atoms significantly alters the physical and chemical properties of the parent molecule, enhancing metabolic stability and pharmacokinetic potency while maintaining or improving anti-tumor efficacy against liver cancer cells.
Q: Are the raw materials for this synthesis commercially available?
A: Yes, the synthesis utilizes readily available commodity chemicals such as furan, maleic anhydride, and various tetrafluorobenzyl alcohols, ensuring a robust and reliable supply chain for commercial production.
Q: How does this method improve upon traditional cantharidin therapies?
A: Unlike native cantharidin which suffers from severe toxicity, these novel derivatives feature a carboxyl group that improves water solubility and stability, reducing systemic toxicity while targeting hepatoma cells more effectively.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Norcantharidin Tetrafluorobenzyl Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of fluorinated norcantharidin derivatives in the fight against liver cancer. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from discovery to market. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify identity and assay.
We invite you to collaborate with us to leverage this innovative technology for your drug development pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your timeline and optimize your budget for next-generation anti-cancer therapeutics.
