Advanced Synthesis of Fluorinated Norcantharidin Derivatives for High-Potency Anticancer Applications
Introduction to Novel Fluorinated Norcantharidin Derivatives
The pharmaceutical landscape is continuously evolving towards high-potency, low-toxicity anticancer agents, and patent CN111362962A represents a significant breakthrough in this domain by introducing a novel class of norcantharidin carboxylate tetrafluorobenzyl esters. This intellectual property details a sophisticated molecular engineering strategy where the core norcantharidin scaffold is chemically modified to overcome the historical limitations of cantharidin-based therapies, specifically their narrow therapeutic index and severe systemic toxicity. By strategically incorporating a carboxyl group and multiple fluorine atoms into the molecular architecture, the inventors have created compounds that exhibit markedly improved water solubility, enhanced metabolic stability, and superior pharmacokinetic profiles compared to the parent molecule. The general structural formula of these innovative intermediates is depicted below, highlighting the critical ester linkage and the tetrafluorophenyl moiety that drives the enhanced biological activity.
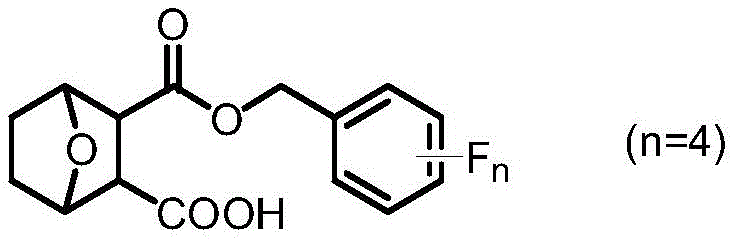
For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, understanding the precise structural nuances of Formula I is essential, as it dictates both the synthetic complexity and the potential therapeutic value. The presence of the fluorine atoms is not merely decorative; it fundamentally alters the electron density and lipophilicity of the molecule, facilitating better membrane permeability and receptor binding affinity, which are crucial parameters for effective anticancer drug design. Furthermore, the synthetic methodology described in the patent offers a robust pathway for generating these complex structures from commodity chemicals, suggesting a viable route for cost reduction in API manufacturing without compromising on purity or efficacy standards required for clinical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to utilizing cantharidin and its derivatives in oncology have been severely hampered by the compound's inherent toxicity, which causes significant irritation to the human urinary and digestive systems, thereby limiting the maximum tolerable dose and overall therapeutic efficacy. While norcantharidin (NCTD) was developed as a less toxic alternative by replacing methyl groups with hydrogen, it still suffers from a short residence time in the human body and a narrow safety margin where slight overdosing leads to adverse reactions. Conventional synthesis methods often struggle to introduce functional groups that can simultaneously improve solubility and metabolic stability without requiring prohibitively expensive reagents or complex multi-step protection-deprotection sequences that drive up production costs. Additionally, the lack of fluorine incorporation in earlier generations of these drugs meant missed opportunities for enhancing bioavailability and binding specificity, which are now recognized as critical factors in modern medicinal chemistry.
The Novel Approach
The novel approach outlined in the patent circumvents these historical bottlenecks by employing a direct and efficient esterification strategy that couples norcantharidin carboxylic acid derivatives with various isomers of tetrafluorobenzyl alcohol. As illustrated in the reaction scheme below, the process utilizes norcantharidin (Compound 4) and reacts it with side-chain tetrafluorobenzyl alcohol (Compound 5) in the presence of an organic base within a suitable solvent system. This method is remarkably straightforward, avoiding the need for harsh reaction conditions or exotic catalysts, which significantly simplifies the operational complexity for large-scale manufacturing teams. The versatility of this approach is evident in its ability to accommodate different fluorine substitution patterns (2,3,4,5-; 2,3,4,6-; or 2,3,5,6-), allowing medicinal chemists to fine-tune the physicochemical properties of the final drug candidate to optimize its performance against specific tumor types.
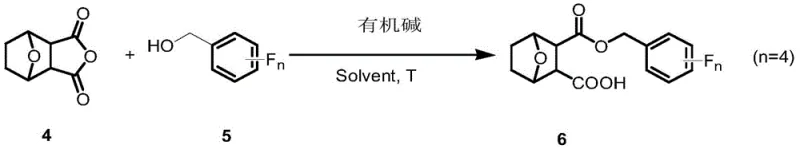
From a supply chain perspective, this novel synthetic route is highly advantageous because it relies on starting materials that are commercially available and relatively inexpensive, such as furan and maleic anhydride for the precursor synthesis. The reaction conditions, typically involving mild heating around 60°C in solvents like dichloromethane, are easily manageable in standard stainless steel reactors, reducing the capital expenditure required for specialized high-pressure or cryogenic equipment. This accessibility translates directly into enhanced supply chain reliability, as manufacturers are not dependent on single-source suppliers for obscure reagents, thereby mitigating the risk of production delays due to raw material shortages. The ability to produce these high-value intermediates through a streamlined process positions this technology as a compelling option for companies aiming to secure a stable supply of next-generation anticancer ingredients.
Mechanistic Insights into Diels-Alder Cycloaddition and Catalytic Esterification
To fully appreciate the technical robustness of this synthesis, one must examine the mechanistic underpinnings of the precursor formation, which begins with a classic Diels-Alder cycloaddition between furan and maleic anhydride. This [4+2] cycloaddition is a highly atom-economical reaction that constructs the bicyclic oxabridge framework characteristic of the norcantharidin skeleton with excellent stereocontrol, forming the 5-ene-norcantharidin intermediate (Compound 3). Following this, a catalytic hydrogenation step using palladium on carbon (Pd/C) saturates the double bond to yield the stable norcantharidin core (Compound 4), a transformation that is critical for establishing the rigid geometry required for biological activity. The subsequent esterification step leverages the nucleophilic catalysis of 4-dimethylaminopyridine (DMAP), which activates the carboxylic acid moiety of the norcantharidin derivative towards attack by the fluorinated benzyl alcohol, ensuring high conversion rates even with sterically hindered substrates.
Impurity control in this process is managed through the high regioselectivity of the Diels-Alder reaction and the specific choice of fluorinated alcohol isomers, which minimizes the formation of positional isomers that could complicate downstream purification. The use of organic bases like triethylamine or DMAP helps to scavenge protons generated during the esterification, driving the equilibrium towards the product side and preventing acid-catalyzed degradation of the sensitive anhydride-derived core. For quality assurance teams, understanding these mechanistic details is vital for establishing critical process parameters (CPPs) such as temperature control during the exothermic cycloaddition and the precise stoichiometry of the coupling reagents to ensure consistent batch-to-batch reproducibility. The resulting products demonstrate distinct NMR signatures, particularly in the fluorine NMR spectra, which serve as powerful analytical tools for verifying the identity and purity of the specific tetrafluoro-isomer produced in each run.
How to Synthesize Norcantharidin Carboxylate Tetrafluorobenzyl Ester Efficiently
The synthesis of these potent anticancer intermediates follows a logical progression from commodity feedstocks to high-value fine chemicals, beginning with the preparation of the norcantharidin core followed by the crucial coupling step. The process is designed to be operationally simple, utilizing standard laboratory glassware or industrial reactors without the need for specialized high-pressure equipment beyond standard hydrogenation setups. Detailed procedural instructions regarding reagent grades, solvent drying, and specific workup protocols are essential for achieving the reported yields and purity levels, ensuring that the final material meets the stringent specifications required for preclinical and clinical evaluation. The following guide outlines the standardized synthesis steps derived from the patent examples, providing a clear roadmap for technical teams looking to replicate or scale this chemistry.
- Preparation of Norcantharidin (Compound 4) via Diels-Alder reaction of furan and maleic anhydride followed by catalytic hydrogenation.
- Esterification reaction between Norcantharidin and specific tetrafluorobenzyl alcohol isomers using an organic base catalyst.
- Purification of the final tetrafluorobenzyl ester product through aqueous workup and flash column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits centered around raw material security and process simplicity. The primary starting materials, furan and maleic anhydride, are bulk petrochemical derivatives produced on a massive global scale, ensuring a stable and competitive pricing structure that is insulated from the volatility often seen in niche pharmaceutical building blocks. This reliance on commodity chemicals significantly de-risks the supply chain, as multiple qualified vendors exist worldwide, preventing bottlenecks that could arise from dependency on a single source for complex chiral pools or rare earth catalysts. Furthermore, the elimination of transition metal catalysts in the final coupling step (using organic bases instead) removes the need for expensive and time-consuming heavy metal scavenging processes, which are a major cost driver in API manufacturing.
- Cost Reduction in Manufacturing: The synthetic pathway achieves cost optimization primarily through the use of inexpensive, readily available starting materials and the avoidance of complex chiral resolution steps, as the Diels-Alder reaction provides the necessary stereochemistry inherently. By utilizing organic base catalysis rather than precious metal complexes for the esterification, the process eliminates the cost associated with metal removal and validation, leading to substantial savings in both reagent consumption and waste treatment. The moderate reaction temperatures and ambient pressure conditions further reduce energy consumption and equipment wear, contributing to a lower overall cost of goods sold (COGS) for the final intermediate.
- Enhanced Supply Chain Reliability: The robustness of the chemistry ensures high supply continuity, as the reaction tolerances are wide enough to accommodate minor variations in raw material quality without catastrophic failure of the batch. The modular nature of the synthesis allows for the independent production of the norcantharidin core and the fluorinated alcohol side chains, enabling parallel processing strategies that can drastically shorten lead times for high-purity pharmaceutical intermediates. This flexibility is crucial for responding to sudden spikes in demand from downstream drug developers, ensuring that production schedules can be accelerated without compromising on the rigorous quality standards expected in the oncology sector.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory optimization to kilogram and ton-scale commercial production using standard unit operations like filtration, distillation, and crystallization. From an environmental perspective, the atom economy of the Diels-Alder step is excellent, and the use of common organic solvents like dichloromethane or ethyl acetate allows for efficient recovery and recycling systems to be implemented, minimizing the environmental footprint. The absence of toxic heavy metals in the final steps simplifies wastewater treatment compliance, making this a greener and more sustainable manufacturing option for environmentally conscious pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these fluorinated norcantharidin derivatives, based on the detailed disclosures within the patent literature. Understanding these aspects is critical for stakeholders evaluating the feasibility of integrating this technology into their existing drug development pipelines or manufacturing portfolios. The answers provided reflect the specific experimental data and mechanistic insights documented in the source material, offering a transparent view of the technology's capabilities and limitations.
Q: What are the key structural advantages of norcantharidin tetrafluorobenzyl esters?
A: The introduction of a carboxyl group significantly improves water solubility and stability compared to parent cantharidin, while the fluorine-containing group enhances pharmacokinetic potency and biological activity against liver cancer cells.
Q: Is the synthesis route scalable for commercial production?
A: Yes, the method utilizes readily available raw materials like furan and maleic anhydride, and employs standard organic synthesis techniques such as Diels-Alder reactions and esterification, which are highly amenable to scale-up.
Q: How does the toxicity profile compare to traditional cantharidin?
A: These derivatives are designed to reduce the severe toxicity associated with natural cantharidin, particularly regarding urinary and digestive system irritation, while maintaining or enhancing antitumor efficacy.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Norcantharidin Derivative Supplier
As a premier CDMO partner, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure necessary to translate this patented laboratory synthesis into a robust commercial reality for global pharmaceutical clients. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of norcantharidin carboxylate tetrafluorobenzyl ester meets the exacting standards required for oncology drug development.
We invite interested partners to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. By collaborating with us, you gain access to specific COA data and comprehensive route feasibility assessments that will empower your decision-making process and accelerate your time to market. Let us leverage our deep domain knowledge in fluorine chemistry and anticancer intermediates to support your mission of delivering life-saving therapies to patients worldwide.
