Advanced Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles: A Breakthrough for Commercial Scale-Up of Complex Heterocycles
Introduction to Patent CN114920707B
The pharmaceutical and agrochemical industries continuously demand efficient, scalable routes to fluorinated heterocycles, particularly 1,2,4-triazoles, which serve as privileged scaffolds in modern drug design. Patent CN114920707B, published in April 2023, discloses a groundbreaking preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds that fundamentally shifts the paradigm of C1 source utilization. This technology leverages the ubiquitous solvent N,N-dimethylformamide (DMF) not merely as a medium, but as an active carbon synthon, enabling the construction of the triazole ring through an iodine-promoted tandem cyclization. The significance of this innovation lies in its ability to bypass traditional, costly reagents while operating under remarkably mild, open-flask conditions. For R&D directors and process chemists, this represents a tangible opportunity to streamline the synthesis of key intermediates found in blockbuster medications such as Factor IXa inhibitors and GlyT1 inhibitors, where the trifluoromethyl group is essential for enhancing metabolic stability and bioavailability.
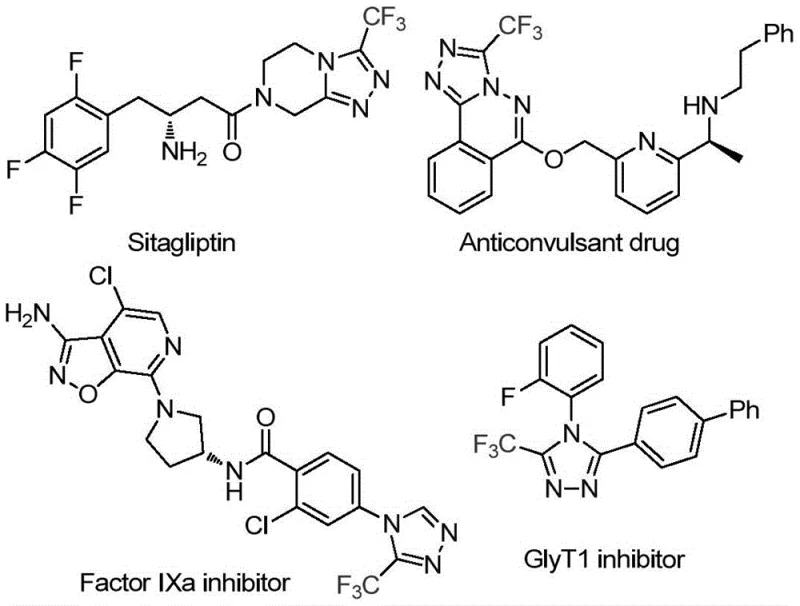
By integrating this methodology into existing production workflows, manufacturers can achieve substantial improvements in atom economy and operational simplicity. The patent details a robust protocol that tolerates a wide array of functional groups, ensuring that the synthetic utility extends far beyond simple model substrates to complex, medicinally relevant molecules. This report analyzes the technical merits of this invention, providing a comprehensive evaluation of its mechanistic underpinnings and its potential impact on supply chain resilience and cost structures for global chemical enterprises seeking reliable pharmaceutical intermediate suppliers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,4-triazoles has been fraught with synthetic challenges that hinder large-scale adoption. Traditional pathways often rely on the condensation of hydrazides with nitriles or amidines, requiring harsh reaction conditions such as high temperatures, strong acids or bases, and strictly anhydrous environments to prevent hydrolysis of sensitive intermediates. Furthermore, many established protocols necessitate the use of expensive transition metal catalysts or specialized fluorinating agents that introduce significant cost burdens and complicate downstream purification due to heavy metal residue concerns. The requirement for inert atmospheres, such as nitrogen or argon shielding, adds another layer of operational complexity and equipment cost, making these processes less attractive for ton-scale manufacturing. Additionally, the introduction of the trifluoromethyl group often requires pre-functionalized starting materials that are themselves difficult to source or synthesize, leading to longer linear sequences and lower overall yields.
The Novel Approach
In stark contrast, the methodology described in CN114920707B offers a streamlined, one-pot solution that elegantly circumvents these bottlenecks. The core innovation involves the use of molecular iodine as a promoter in conjunction with DMF, which acts dually as the solvent and the carbon donor. As illustrated in the general reaction scheme below, the process combines trifluoroethyliminohydrazide with DMF under air at moderate temperatures (110-130°C). This approach eliminates the need for exotic reagents or rigorous exclusion of moisture and oxygen, significantly lowering the barrier to entry for production facilities. The reaction tolerates a broad spectrum of substituents on the aromatic ring, including alkyl, alkoxy, halogen, and trifluoromethyl groups, demonstrating exceptional versatility.
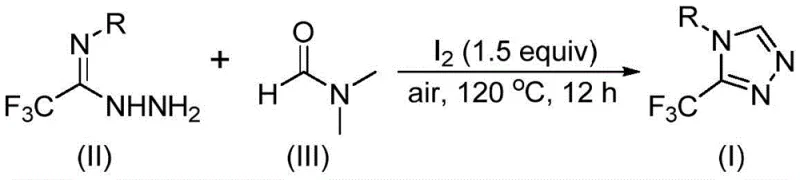
Moreover, the post-treatment process is remarkably straightforward, involving simple filtration and standard column chromatography, which facilitates rapid isolation of high-purity products. This simplicity translates directly into reduced processing time and lower energy consumption, addressing key pain points for procurement managers focused on cost reduction in API manufacturing. The ability to utilize a commodity chemical like DMF as a reactant rather than just a solvent further enhances the economic viability of this route, positioning it as a superior alternative to legacy methods for generating these valuable heterocyclic cores.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
Understanding the mechanistic pathway is crucial for R&D teams aiming to optimize this process for specific substrates. The reaction proceeds through a fascinating dual-pathway mechanism depending on which part of the DMF molecule participates as the carbon source. In the first pathway, the formyl group of DMF acts as the C1 unit. It undergoes a condensation reaction with the trifluoroethyliminohydrazide to generate a hydrazone intermediate. This is followed by an intramolecular cyclization event that eliminates a molecule of dimethylamine, ultimately furnishing the 3-trifluoromethyl-substituted 1,2,4-triazole product. This pathway highlights the electrophilic nature of the DMF carbonyl carbon when activated, allowing for efficient bond formation without external activating agents.
Alternatively, the N-methyl group of DMF can serve as the carbon source in a more complex sequence initiated by molecular iodine. Here, iodine activates the DMF, potentially forming an amine salt species which subsequently eliminates hydrogen iodide. This activated species then undergoes nucleophilic addition with the trifluoroethyliminohydrazide. Following the elimination of N-methylformamide, an azadiene intermediate is generated. The cycle concludes with an intramolecular nucleophilic addition and an oxidative aromatization step to yield the final triazole ring. This mechanistic flexibility ensures high conversion rates across diverse substrates. From an impurity control perspective, the mild oxidative conditions provided by air and iodine minimize the formation of over-oxidized byproducts or polymeric tars often seen in harsher cyclization protocols, thereby simplifying the purification burden and ensuring consistent quality for high-purity 1,2,4-triazole derivatives.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The implementation of this synthesis route is designed for ease of execution in standard laboratory and pilot plant settings. The protocol utilizes readily available starting materials and avoids the need for specialized glovebox techniques or high-pressure reactors. By adhering to the optimized molar ratios and temperature profiles outlined in the patent, chemists can reliably access a library of trifluoromethylated triazoles with varying substitution patterns. The detailed standardized synthesis steps, including precise stoichiometry and workup procedures, are provided in the guide below to facilitate immediate technology transfer and process validation.
- Charge a reaction vessel with molecular iodine (1.5 equivalents), trifluoroethyliminohydrazide substrate, and excess DMF solvent under an air atmosphere.
- Heat the reaction mixture to a temperature range of 110-130°C and maintain stirring for 10 to 15 hours to ensure complete conversion.
- Upon completion, perform standard post-treatment including filtration, washing, drying, and silica gel column chromatography to isolate the pure triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers compelling strategic advantages that extend beyond mere chemical novelty. The shift towards using DMF as a reactant fundamentally alters the cost structure of producing these intermediates. By replacing expensive, specialized C1 donors with a bulk commodity solvent, the raw material costs are drastically simplified. This reduction in input complexity directly correlates to improved margin potential and reduced exposure to price volatility in the specialty chemical market. Furthermore, the elimination of transition metal catalysts removes the necessity for costly metal scavenging steps and rigorous testing for heavy metal residues, which are critical regulatory hurdles in pharmaceutical production.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the substitution of high-cost reagents with low-cost, high-volume solvents. Since DMF is produced on a massive industrial scale, its pricing is stable and predictable compared to niche fluorinating agents. Additionally, the reaction operates under air atmosphere, negating the capital expenditure and operational costs associated with maintaining inert gas lines and monitoring oxygen levels. The simplified workup procedure reduces solvent usage for extraction and purification, leading to substantial cost savings in waste disposal and solvent recovery systems.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of universally available starting materials. Trifluoroethyliminohydrazide and molecular iodine are standard catalog items with multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions means that production is less susceptible to disruptions caused by environmental controls or equipment failures related to sensitive atmosphere management. This reliability ensures consistent delivery schedules for downstream API manufacturers, mitigating the risk of production delays.
- Scalability and Environmental Compliance: The scalability of this process is excellent due to its operation in a homogeneous liquid phase with manageable exotherms. The absence of hazardous gases or pyrophoric reagents simplifies safety protocols for scale-up from kilogram to multi-ton batches. From an environmental standpoint, the atom economy is improved by incorporating the solvent into the product, reducing the overall E-factor of the process. The reduced generation of heavy metal waste and the use of common solvents facilitate easier compliance with increasingly stringent environmental regulations regarding effluent treatment and discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on operational parameters and expected outcomes for potential adopters.
Q: What is the primary advantage of using DMF in this synthesis?
A: DMF serves a dual role as both the reaction solvent and the carbon source (providing either the formyl or N-methyl group), which eliminates the need for separate, expensive C1 building blocks and simplifies the reaction setup.
Q: Does this process require inert gas protection?
A: No, the reaction proceeds efficiently under an air atmosphere, removing the operational complexity and cost associated with maintaining strict anhydrous or anaerobic conditions typically required for sensitive heterocycle synthesis.
Q: What is the substrate scope for this methodology?
A: The method demonstrates broad compatibility with various substituted aryl groups, including those with electron-donating (alkyl, alkoxy) and electron-withdrawing (halogen, trifluoromethyl) substituents, yielding products with moderate to good efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the one described in CN114920707B for the production of high-value pharmaceutical intermediates. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 3-trifluoromethyl-1,2,4-triazole derivative conforms to the highest industry standards required for drug substance manufacturing.
We invite global partners to collaborate with us to leverage this cost-effective and scalable technology for their specific pipeline needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our capabilities can enhance your supply chain security and drive down your overall cost of goods sold.
