Advanced Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Next-Generation Pharmaceutical Applications
Advanced Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Next-Generation Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust and scalable methodologies for constructing fluorinated heterocycles, which serve as critical pharmacophores in modern drug design. Patent CN114920707B discloses a groundbreaking preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds, addressing the long-standing challenges of complexity and cost in heterocyclic synthesis. This technology leverages the ubiquitous solvent N,N-dimethylformamide (DMF) not merely as a medium, but as an active carbon source, enabling a streamlined tandem cyclization promoted by molecular iodine. The significance of this chemical scaffold cannot be overstated, as trifluoromethyl-substituted triazoles are central to the bioactivity of numerous high-value therapeutics, ranging from antidiabetic agents like Sitagliptin to potent Factor IXa inhibitors and GlyT1 inhibitors used in neurological disorders.
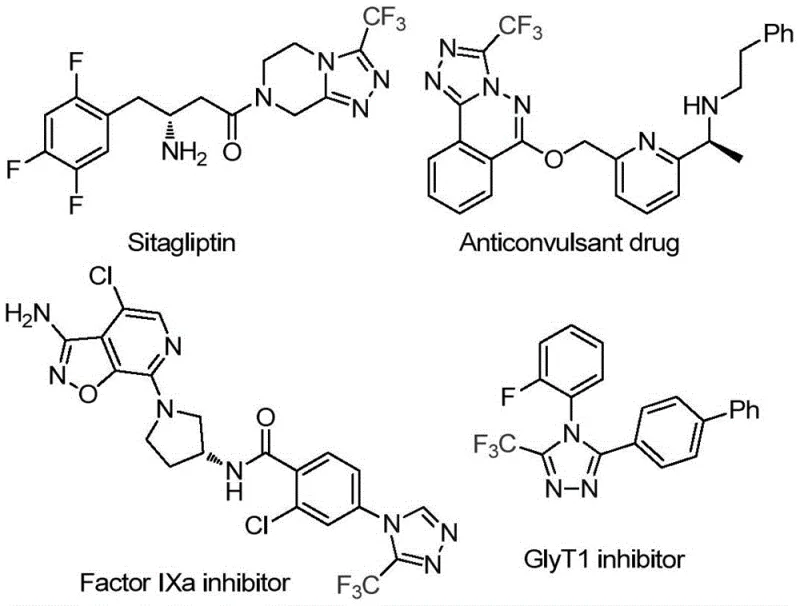
For R&D directors and process chemists, the ability to access these privileged structures through a simplified, one-pot protocol represents a significant strategic advantage. The method eliminates the need for specialized C1 building blocks, instead utilizing the solvent itself to construct the triazole ring. This innovation aligns perfectly with the industry's shift towards atom-economical and operationally simple processes. By operating under air atmosphere and avoiding stringent anhydrous conditions, this patent offers a pathway to high-purity intermediates that is both technically feasible and commercially attractive for large-scale manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of 1,2,4-triazole derivatives has relied on multi-step sequences involving the condensation of hydrazides with nitriles or amidines, often requiring harsh reaction conditions and expensive catalysts. Conventional routes frequently necessitate the use of specialized carbon sources that are costly and difficult to handle on a kilogram scale, leading to inflated production costs and supply chain vulnerabilities. Furthermore, many existing protocols demand strictly anhydrous and oxygen-free environments, requiring specialized equipment such as gloveboxes or Schlenk lines, which significantly hampers throughput and increases capital expenditure for manufacturing facilities. The purification of intermediates in these traditional pathways is often cumbersome, involving complex workups to remove metal catalysts or toxic byproducts, thereby reducing the overall yield and environmental sustainability of the process.
The Novel Approach
In stark contrast, the methodology described in CN114920707B revolutionizes the synthetic landscape by employing DMF as a dual-function reagent. This approach utilizes molecular iodine as a mild yet effective promoter to activate the DMF solvent, facilitating a tandem cyclization that constructs the triazole core in a single operational step. The reaction proceeds efficiently at temperatures between 110°C and 130°C under ambient air, removing the logistical burden of inert gas protection. This simplicity translates directly into operational excellence, as the starting materials—trifluoroethyliminohydrazide and DMF—are commodity chemicals with stable supply chains. The versatility of this method allows for the introduction of diverse functional groups on the aromatic ring, enabling the rapid generation of analog libraries for structure-activity relationship (SAR) studies without the need for process re-optimization.
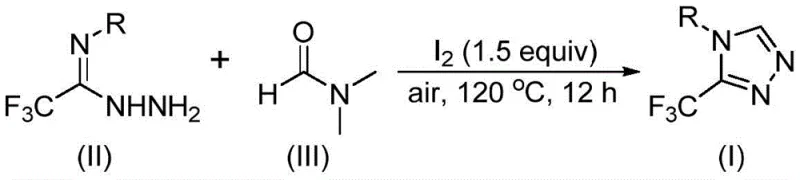
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The mechanistic elegance of this transformation lies in the ambident nature of DMF, which can participate in the reaction via two distinct pathways depending on which carbon fragment is utilized. In the first pathway, the formyl group of DMF acts as the carbon source, undergoing condensation with the trifluoroethyliminohydrazide to form a hydrazone intermediate. This species then undergoes intramolecular cyclization followed by the elimination of dimethylamine to yield the desired 3-trifluoromethyl-1,2,4-triazole product. Alternatively, the N-methyl group of DMF can serve as the carbon donor; in this scenario, iodine activates the solvent to generate an amine salt, which subsequently reacts with the hydrazide via nucleophilic addition. This pathway involves the elimination of N-methylformamide to generate an azadiene intermediate, which then undergoes oxidative aromatization to finalize the heterocyclic ring system.
Understanding these mechanistic nuances is crucial for impurity control and process optimization. The use of molecular iodine (preferably 1.5 equivalents) ensures efficient activation of the solvent while minimizing side reactions that could lead to polymeric byproducts or incomplete conversion. The reaction tolerance is remarkably high, accommodating electron-withdrawing groups such as trifluoromethyl and halogens, as well as electron-donating groups like methoxy and alkyl chains, without significant degradation in yield. This robustness suggests that the transition states involved are stabilized effectively by the reaction conditions, allowing for a broad substrate scope that is essential for the synthesis of complex pharmaceutical intermediates where specific substitution patterns are required for biological activity.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical implementation of this synthesis is designed for ease of execution in both laboratory and pilot plant settings. The protocol requires minimal setup, utilizing standard glassware and heating mantles without the need for specialized pressure vessels. The reaction mixture typically consists of the trifluoroethyliminohydrazide substrate, molecular iodine, and excess DMF, which serves as both reactant and solvent to drive the equilibrium towards product formation. Following the reaction period of 10 to 15 hours, the workup is straightforward, involving filtration to remove inorganic salts followed by standard chromatographic purification. This streamlined workflow significantly reduces the technician time and solvent consumption associated with more convoluted synthetic routes.
- Combine molecular iodine (1.5 equivalents) and trifluoroethyliminohydrazide substrate in a reaction vessel containing DMF solvent.
- Heat the reaction mixture to 110-130°C under an air atmosphere and maintain stirring for 10-15 hours to ensure complete conversion.
- Upon completion, perform standard post-treatment including filtration, silica gel mixing, and column chromatography to isolate the high-purity triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented technology offers compelling advantages that directly impact the bottom line and operational resilience. The reliance on DMF, a solvent produced on a massive global scale, ensures that raw material availability is never a bottleneck, unlike specialized reagents that may have limited suppliers or long lead times. The elimination of noble metal catalysts removes the need for expensive metal scavenging steps and reduces the risk of heavy metal contamination in the final API, simplifying regulatory compliance and quality control workflows. Additionally, the ability to run the reaction under air atmosphere significantly lowers utility costs associated with nitrogen or argon purging, contributing to a leaner and more sustainable manufacturing process.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the consolidation of roles, where DMF acts as both solvent and reagent, effectively reducing the bill of materials. By avoiding the purchase of discrete C1 synthons and expensive transition metal catalysts, the direct material costs are substantially lowered. Furthermore, the simplified workup procedure reduces solvent usage for extraction and washing, leading to significant savings in waste disposal and solvent recovery costs. The high reaction efficiency observed across various substrates implies that less starting material is wasted, maximizing the return on investment for every batch produced.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of commodity chemicals that are readily available from multiple global vendors, mitigating the risk of single-source dependency. The robustness of the reaction conditions means that production is less susceptible to delays caused by equipment failures related to inert atmosphere maintenance. This reliability allows for more accurate forecasting and inventory management, ensuring that critical pharmaceutical intermediates can be delivered to downstream customers without interruption. The scalability of the process from gram to kilogram scales ensures that supply can easily ramp up to meet market demand as drug candidates progress through clinical trials.
- Scalability and Environmental Compliance: The environmental profile of this synthesis is favorable due to the avoidance of toxic heavy metals and the use of a single primary solvent, which simplifies waste stream management. The reaction generates dimethylamine or N-methylformamide as byproducts, which are manageable within standard industrial waste treatment protocols. The absence of stringent anhydrous requirements reduces the energy footprint associated with solvent drying and reactor conditioning. These factors collectively support a greener manufacturing strategy that aligns with increasingly strict environmental regulations and corporate sustainability goals, making the process attractive for long-term commercial adoption.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and specifications outlined in the patent documentation, providing clarity on reaction scope, safety, and scalability. Understanding these details is essential for process engineers and project managers evaluating the feasibility of adopting this route for their specific pipeline projects.
Q: What is the primary advantage of using DMF in this triazole synthesis?
A: DMF serves a dual role as both the reaction solvent and the essential carbon source (providing either the formyl or methyl group), which drastically simplifies the reagent list and reduces raw material costs compared to traditional methods requiring separate C1 donors.
Q: Does this process require inert gas protection?
A: No, one of the key operational benefits of this patented method is that it proceeds efficiently under an air atmosphere, eliminating the need for expensive anhydrous or anaerobic conditions typically required for sensitive heterocyclic formations.
Q: What types of substituents are tolerated on the aromatic ring?
A: The method demonstrates broad substrate scope, successfully accommodating various functional groups including alkyl, alkoxy, alkylthio, halogens (fluorine, chlorine), and trifluoromethyl groups at ortho, meta, or para positions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of life-saving medications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-trifluoromethyl-1,2,4-triazole meets the exacting standards required by the global pharmaceutical industry. Our commitment to technical excellence allows us to navigate complex synthetic challenges, delivering consistent quality and reliability to our partners worldwide.
We invite you to collaborate with us to leverage this innovative synthesis technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your timeline and optimize your budget. Let us be your trusted partner in bringing advanced therapeutic solutions to market faster and more efficiently.
